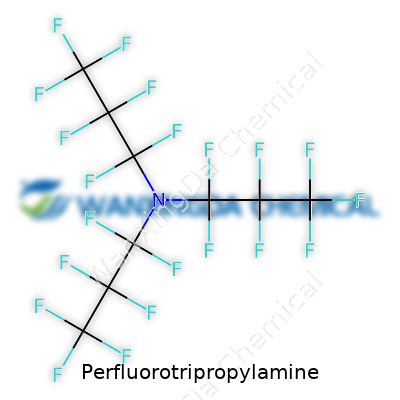
Perfluorotripropylamine: A Commentary on Its Journey and Importance
Tracing the Roots: Historical Development
The chemical world has always ridden the line between innovation and risk management, laying foundations that often stretch into uncharted territory. Perfluorotripropylamine represents a story of ingenuity set against the sprawling backdrop of post-war chemistry. As scientists uncovered the remarkable stability and unique properties of perfluorinated compounds, new doors opened for materials with extreme resilience. These molecules came to the fore during the rush for advanced electronics and aerospace technologies, where resistance to heat and chemical attack could mean the difference between mission success and disaster. Adopting trifluoropropyl groups in amines signaled an intent to engineer substances purpose-built for unforgiving environments. This approach didn’t aim for the mainstream but for niche applications where compromise was a luxury engineers couldn’t afford.
Taking a Close Look: Product Overview
Some chemicals feel built for obscurity, but Perfluorotripropylamine has quietly influenced specialized sectors. It is a perfluoroalkylamine, crafted by fully substituting all hydrogens in a propylamine chain with fluorines, allowing the molecule to shrug off many common reactive chemicals. For those working in electronics manufacturing or the higher reaches of aviation technology, these substances solve stubborn problems—they stay liquid even at chilly temperatures, resist evaporation, and refuse to interact with almost anything short of extreme conditions. In the lab, they provide fluids that won’t break down under pressure, making them outlast rival substances and justify often hefty investment.
Getting to Know the Properties
Fluorinated molecules like this amine carve out reputations through a rare blend of traits. Boiling points and thermal stabilities stand out, especially where standard organic solvents throw in the towel. Their chemical inertness proves legendary; drop them in an acid bath or expose them to strong bases, and they hold their ground. In practice, this stability means that spills and waste, if left unchecked, may accumulate over years, shaping policy and risk management in research labs and factories alike.
Guidelines and Specifications in the Real World
Perfluorotripropylamine finds itself scrutinized not because it’s flashy, but because lapses in handling can have consequences far beyond the walls of a single facility. Tech specs vary, yet the demand for rigor never wavers—purity levels and labeling must keep step with regulatory pressures and user expectations. Often, producers outline requirements for storage in corrosion-resistant containers, shielding the compound from sunlight and extreme temperatures, and documenting every movement from creation to disposal.
Preparation Method—From Concept to Chemistry
Building a robust molecule like Perfluorotripropylamine means tackling a synthesis that calls for fluorination reactions at scale. Historically, direct fluorination wasn’t a friendly process; unplanned results and equipment corrosion made it risky. Over time, chemists leaned toward electrochemical fluorination of precursors, developing better ways to contain byproducts and refine yields. Lab conversations often circle back to process safety—stories of mishaps in the early days still echo, reminding everyone of the need for respect and careful planning around reactive fluorine gas.
Chemical Reactions and Modifications
Most organic chemists quickly learn that perfluorinated amines don’t act like their more conventional cousins. Routine modifications become exercises in frustration. Attempts to tweak the structure or attach functional groups stumble because these molecules resist attack from most reagents. The very stability that makes them valuable also limits what scientists can do in the way of post-production alteration. Researchers often focus on blending or suspending these amines, rather than hoping to dramatically change their backbone through conventional chemical means.
Names and Synonyms: Avoiding Confusion
Chemistry can get confusing fast. Names multiply—perfluorinated propylamine, tri-n-propylamine, and a list of registry numbers that only specialists keep straight. Anyone ordering compounds for research or industrial use knows this pain: a shipment with a wrong label doesn’t just slow down progress, it can shut down a production line. This is no small issue when supply chains run lean and regulatory inspections assume full accountability for each drum or flask hitting the loading dock.
Safety and Operations: The Human Factor
Safety concerns with perfluorinated compounds stretch well beyond the acute toxicity often flagged in chemical handling. Long-term exposure, bioaccumulation, and environmental persistence top the list. Personal protective equipment isn’t window dressing but a real shield against unknowns in the risk profile. Stories circulate inside industry circles about careless disposal or mishandled spills—those become cautionary tales as much for liability as for real human and environmental cost. The ongoing move toward more transparent labeling and a “right to know” attitude helps, but underlying gaps remain, especially wherever rapid-scale production runs up against legacy habits or budget pressure.
Application Areas: Where Utility Meets Necessity
Electronic cooling systems, aerospace hydraulics, and specialist lubricants—their continued evolution draws heavily from molecules like Perfluorotripropylamine. Low flammability and chemical inertness play vital roles in remote satellites, deep-sea sensors, and high-voltage transformers. These are not optional characteristics for users; they represent base requirements, carving out a space for perfluorinated amines even when cost or procurement hurdles would chase lesser substances off the field.
Progress in Research and Development
My experience suggests research in perfluorinated chemistries is as much about responding to pressure as it is about pushing boundaries. Concerns over environmental persistence and toxicity now drive almost half the new inquiries. With mounting regulatory attention focused on “forever chemicals,” R&D teams balance innovation with responsibility. Solutions often look toward harnessing benefits while hunting down viable paths for safe degradation, recycling, or containment, even if tradeoffs sometimes look steep.
Toxicity Research: Hard Lessons and New Directions
Toxicity studies linked with perfluorinated amines kick up hard debates in scientific and policy circles. Persistence in the environment puts even trace compounds under the spotlight, and bioaccumulation adds real urgency. Experiments in animal models highlight risks beyond immediate poisoning—long-term changes in liver function and developmental impacts on offspring raise alarms. Regulatory responses haven’t always run ahead of the curve; many policies follow public awareness and evolving data sets. Personal reflection on these issues comes easy here: few working in the chemical field would now brush aside the call for tighter oversight and cross-disciplinary research on long-term exposure risks.
Looking Ahead: Future Prospects and Responsibility
Global movements now eye the balance between chemical innovation and environmental health with greater skepticism. The future of Perfluorotripropylamine hangs on more than technical excellence. Researchers and industry leaders watch as bans, phaseouts, and new guidelines gather pace—adaptation stands as the watchword. Alternatives that aim for shorter lifespans or more natural breakdown routes are gaining traction, even when performance superiority isn’t automatic. Remaining relevant no longer leans solely on scientific achievement; earning trust and regulatory approval now ride along every step, shaping both new research agendas and practical business decisions into the coming decade.
Perfluorotripropylamine, or PFTPA for short, doesn’t show up in daily conversation for most folks. Yet, whether we realize it or not, it has touched some important slices of our modern life. As a fluorinated amine, it stands out for its chemical stability and resistance to heat. That’s not just chemistry talk—it echoes in the places where the pressures of industry and technology stretch regular materials past their limits.
Cooling Down High-Tech Electronics
Scientists have leaned on PFTPA in the cooling world, especially when talking about supercomputers or data center servers. These aren’t desktops at home — they churn through enormous amounts of data, run hot, and squeeze hardware in ways the average laptop never feels. Here, standard water-based coolants fall short. PFTPA doesn’t break down at high temperatures, and it doesn’t make electronics rust, which makes it a solid fit for “immersion cooling.” Basically, they dunk heat-producing hardware straight into this specialized liquid. That keeps servers from frying themselves and lets companies pack more power in smaller spaces. Less heat means fewer outages, longer life for massive computer investments, and maybe even lower electricity bills over time.
A Place in Medicine
Specialty chemicals like PFTPA sometimes take an unexpected path into healthcare. In the past, researchers have looked at these compounds for use in artificial blood and as a component in liquid breathing research. The idea is wild—using a liquid that carries gases well enough that lungs can absorb oxygen and release carbon dioxide. Real-world adoption for humans hasn’t panned out on a large scale, often due to tricky side effects and big regulatory hurdles. Animals have seen more use in experimental settings, though. The chase for new solutions in emergency medicine and critical care always sparks curiosity, and unique molecules like PFTPA sometimes get a seat at the table for a while before newer science takes over.
Concerns About Environmental Footprint
Fluorinated chemicals, including PFTPA, tend to stick around. Literally. They don’t break down quickly once released into the environment. There’s growing global debate around the long-term effects of persistent chemicals. I’ve seen news on PFAS—another group of “forever chemicals”—popping up in the water and raising alarms. It reminds us that with any powerful tool, caution matters. Regulatory agencies have started to watch these compounds more closely, pushing scientists and manufacturers to search for safer or more biodegradable alternatives. Large-scale use calls for thoughtful handling, robust disposal processes, and real transparency about long-term risks. This isn’t about stoking fear but keeping public health and clean water in sight while innovation continues.
Finding Balance in Innovation
Using perfluorotripropylamine feels like walking a tightrope. Its benefits in tiny, focused quantities during chip manufacturing or for extreme cooling mark real leaps in efficiency and performance. But the confidence to use it comes with trust—trust that companies will handle waste responsibly and invest in greener chemistry. Consumer pressure has pushed whole industries to reformulate products after learning about legacy pollution. The hope lies with those innovators who put as much work into eco-friendly exits as they do into groundbreaking entries. That mindset could take the shine off future headlines about contamination or health scares, and keep the best of science in service to everyone.
The Realities Behind Perfluorotripropylamine
I’ve always had a healthy skepticism about chemical compounds that sound more like a tongue-twister than something most folks need in daily life. Perfluorotripropylamine, often called FC-3283 in labs, turns up in some surprising places: electronics cooling systems, certain medical procedures, and sometimes as part of research at institutions I trust. The thing is, substances that make it into these high-tech systems usually slide into discussions about safety and toxicity. People hear “perfluoro” and immediately connect it with long-lived pollutants or those tricky PFAS chemicals everyone’s trying to keep out of their water supply.
The Evidence So Far
Experts pay attention when a chemical has a string of fluorines locked into its structure, since those bonds don’t want to break down in nature. That’s the root of the concern. When perfluorotripropylamine pops up, it brings some baggage—persistence in the environment, bioaccumulation worries, and a legacy in the same family as notorious substances like PFOA or PFOS. For these, health agencies found links to liver problems, developmental harm, and even some cancers in heavily exposed people. The EPA and WHO both flag this class of chemicals for extra scrutiny.
Looking at perfluorotripropylamine specifically, hard numbers on its toxicity are strangely tough to pin down. Most safety data sheets put the focus on general hazards: avoid breathing the vapor, don’t let it splash in your eyes or on your skin. Animal studies on some perfluorinated amines hint that inhalation at high levels can irritate lungs and may mess with the nervous system. Long-term effects in humans haven’t been widely documented, partly because the use cases are still quite niche.
Why This Matters to All of Us
When I talk to friends in research or healthcare, nobody wants to gamble with chemical exposure. We’ve seen what happens when industry moves faster than regulation or independent study. Decades of use, then scientists realize a particular substance builds up in blood, or lingers in dust, or turns into something more toxic down the line.
This isn’t just an academic issue. Modern tech often leans on fluorinated chemicals to make devices run cooler, to improve imaging in medicine, and to chase the latest breakthroughs in chemistry. But there’s a trade-off: stability in engineering can mean near-permanence in the environment. Once these substances get outside controlled settings, removing them from soil or water becomes almost impossible at reasonable cost.
Charting a Smarter Path Forward
It makes sense to press for sharper studies on compounds like perfluorotripropylamine—not as a scare tactic, but because the stakes are real. Regulatory bodies move slow. Consumer awareness can speed things up. Companies that depend on these chemicals could invest more in green chemistry so the next generation doesn’t have to pay for our shortcuts.
I came up in a time when “better living through chemistry” meant exciting new products every year. Now, I see that living with those choices means staying informed, asking what’s in our systems, and holding both industry and regulators accountable. If history teaches anything, waiting for problems to show up before we act costs more—financially and in human health—than taking a critical look before that first leak or spill.
Why the Structure Matters
It's easy to overlook the impact of molecules like perfluorotripropylamine unless your work lives close to the world of industrial chemistry or high-tech manufacturing. I stumbled upon this compound working through research on materials that must withstand the harshest possible environments—think spacecraft, nuclear facilities, or advanced electronics.
Perfluorotripropylamine stands out for its carbon-fluorine bonds. Each hydrogen in typical tripropylamine is replaced by fluorine. That change does more than tweak a molecule’s appearance. It rewires its chemical behavior, making it impressively stubborn against heat, acids, bases, and pretty much any reactive material you toss at it. This profile means you won’t see it breaking down or reacting in messy, unpredictable ways. In my own work, durability at the molecular level almost always pays off in fewer maintenance headaches and more consistent results over months or even years.
Chemical Inertia: A Real World Double-Edged Sword
I learned early on that perfluorinated compounds refuse to mingle with water or oils. This is because the surface falls under the grip of tightly packed fluorine atoms. The molecule won’t catch on fire, and microbes give up trying to use it for food. That sounds great if you're building electronics that must not short out or degrade from moisture or organic attack. With this much resilience, we imagine electronic coolants and some tricky lubricants for specialized gear use it all the time. The flip side? These same properties set off alarm bells among environmental scientists—these molecules hang around, almost never breaking down on their own.
Real Concerns: Health and Environment
I once spoke to a colleague in environmental science who described the frustration of tracking compounds like perfluorotripropylamine in drinking water studies. The molecule’s resistance to breakdown means that wherever it ends up, it tends to stay. While direct evidence pointing to toxicity for this specific compound hasn’t grabbed major headlines, we know from the broader PFAS family that persistence alone raises concern. Many perfluorinated compounds, or “forever chemicals,” show up in unexpected places, from Arctic ice to suburban tap water—raising uncomfortable questions about long-term health and responsibility.
Industry Leans on Stability
Companies have strong reasons for using molecules like this. Think of a coolant that needs to stay stable under both high voltages and extreme temperatures. Here, breakdown or unwanted chemical reactions could endanger a multimillion-dollar operation. Researchers prize that chemical patience in demanding scientific experiments too. In my time collaborating with engineers, I saw first-hand the relief that comes from choosing materials proven to behave the same way, every time, under stress.
Solutions: A Call for Better Stewardship
Regular folks don’t notice these chemicals, but engineers, scientists, and regulators carry the responsibility. Companies need to trace exactly where these molecules go, avoid careless disposal, and design closed-loop processes that reclaim and recycle wherever possible. Advanced filtration techniques and “green chemistry” approaches—where safer, biodegradable alternatives are developed—deserve more funding and attention. Cleaner solutions often demand creative leaps from the laboratory to manufacturing scale, as I witnessed working on filtration projects for specialty solvents. Public pressure and sound regulation help spur this kind of change.
Every new application teaches us something fresh about both the value and cost of chemical stability. If we get smarter about production and clean-up, we can keep reaping the benefits while hacking away at the downside of environmental build-up.
Perfluorotripropylamine isn’t an ordinary chemical you stumble across in everyday life. For those working with it in labs or processing environments, understanding how to manage it safely isn’t just a suggestion—it’s a matter of responsibility. I’ve seen what happens when people treat specialty chemicals like any other basic lab supply. It causes trouble, sometimes even accidents nobody saw coming. If you’re around this compound, you can’t just rely on old habits. You need to engage with it like you mean it.
Why Safe Storage Matters
Ask anyone who has spent their career around fluorinated compounds—they’ll echo the same thing: proper storage keeps everyone safe. This chemical has unique properties that make it useful in specialized industries like electronics cooling and as a component in some advanced materials. But that doesn’t erase its potential hazards if left unattended or exposed to the wrong conditions.
One big risk is reaction with incompatible materials. Storing perfluorotripropylamine near oxidizers or strong acids sets the stage for unwanted chemical reactions. If you work in a small facility, pressed for space, the temptation to pack containers together in any open shelf is strong, but it’s a shortcut I’ve seen go wrong. Even small leaks or spills can escalate if containers sit side by side with reactive chemicals.
Sturdy Containers and Thoughtful Placement
If you take a walk through a well-run chemical storeroom, something stands out: nothing sits randomly. Perfluorotripropylamine calls for tightly sealed, clearly labeled containers. Quality materials for storage matter—the wrong plastics or old seals let vapors escape or break down over time. I once saw a bottle nearly empty itself after a neglected cap warped months after being purchased.
Place containers in a cool, dry part of the storeroom. Heat or direct sunlight speeds up chemical changes and can strain seals. This isn’t just about keeping things tidy; it’s about stopping accidents before they can start. A climate-controlled cabinet goes a long way here.
Personal Protection and Good Habits
No matter how careful your colleagues seem, direct skin or eye contact remains a risk. Trusting gloves, lab coats, and decent goggles gives peace of mind that lasts through a long shift. After you’ve finished, wash your hands—sounds simple, but this routine has saved countless people from hours of irritation or worse.
Work areas matter too. Handling perfluorotripropylamine in well-ventilated spaces reduces chance of inhaling fumes. Running a fume hood isn’t overkill—it’s smart. I’ve seen labs go without for short tasks, and small exposures add up.
Solutions in Communication and Training
I’ve learned that sharing knowledge beats written rules alone. Regular training makes the difference between people who glance at guidelines and those who know them inside out. Don’t just rely on a poster or a binder gathering dust—talk through safe practices, repeat basic checks, and share real stories about what happens when things go wrong. This builds a culture where nobody feels uncomfortable double-checking storage or asking about some unfamiliar bottle.
Safe handling of perfluorotripropylamine doesn’t rest on expensive equipment or complicated procedures. It thrives on attention to detail, good habits, and the willingness to ask questions. That’s how you build trust, safety, and reliability around something as potent as a fluorinated amine.
Everyone who has worked in a science lab or dabbled in research knows about the Chemical Abstracts Service, better known as CAS. Sometimes, a name just isn’t enough. Chemicals get lost in translation, especially when they pick up a dozen synonyms or the language slips into abbreviations. The CAS number steps in like a barcode at the grocery store—no confusion, just a unique shot of certainty.
Perfluorotripropylamine has made its way into many conversations about fluorinated compounds. You might find it discussed in environmental reports or when folks talk about "forever chemicals." This compound belongs to the broader family of perfluorinated amines, not a household staple but increasingly on the radar for folks tracking the movement of synthetic chemicals in the environment.
The CAS number for Perfluorotripropylamine is 511-89-7. Search this number in any major chemical registry or regulatory database, and you’ll find the same molecule, free from mix-ups about brand names or spelling quirks. Responsible handling depends on this number—regulators and researchers use it to map out risk assessments, shipping forms, and inventory lists. Big agencies like the EPA or the European Chemicals Agency rely on CAS numbers for everything from policy writing to customs control. I’ve seen this myself in environmental science work, where cross-checking a CAS code often clears up misunderstandings before they spiral into bigger headaches.
Why CAS Numbers Matter Beyond the Lab
Tracking a chemical by name alone is like looking for a library book by describing its cover. Names come and go, but numbers cut through language barriers. The CAS number prevents accidental switches or mistaken identities, especially with compounds that have sibling molecules a molecule or two away in structure but act wildly differently in a biological system. Given how fast new variations show up in the world of synthetic chemicals, sticking to a precise identifier gives people a fighting chance to keep up.
Perfluorotripropylamine doesn’t turn up in every factory, but it’s part of the broad category of perfluoroalkyl substances (PFAS), which have a reputation for hanging around in soil, water, and living tissue. Regulatory agencies keep close track of these because once they are out in the environment, they don’t simply break down or go away. Knowing the CAS number helps community groups and scientists keep records straight in public databases, environmental testing programs, and scientific papers. The last time I collaborated on a contamination study, the importance of precise naming and the clarity of a fixed CAS code stood out—especially when compared with older reports that tangled up several PFAS compounds into a single “type.”
Getting Clarity in Chemical Communication
One obstacle the public faces is the technical fog in chemical safety communication. Manufacturers and agencies should present clear information, and NGOs or citizen scientists need strong tools to keep industry accountable. Using CAS numbers on labels, reports, and in school science classes can cut through that fog. Educators who give students these identifiers are not just teaching a trivia fact—they’re equipping young people to read technical sources, question environmental claims, and access accurate data.
As we run into bigger environmental and health puzzles brought by synthetic chemicals, standardized terms and unique codes like CAS numbers are simple practices that make a world of difference. They cut through corporate jargon, language barriers, and scientific shorthand, handing the same information to everyone, straight up.