Potassium Perfluorobutanesulfonate: The Push and Pull of Science, Safety, and Solutions
A Look Back: Development and Discovery
People talk about potassium perfluorobutanesulfonate like it’s a fresh badge earned by the modern chemical world, but its roots stretch back to the decades when researchers tried to build something better than what was already on their shelves. Perfluorinated compounds came out of the need for chemicals that held up under heavy stress from heat, acids, and bases. By the time chemists rolled out potassium perfluorobutanesulfonate, industry was on the hunt for replacements that didn’t build up in the body as relentlessly as earlier long-chain fluorinated cousins. The shift wasn’t about sudden enlightenment, but more about hitting roadblocks with old chemicals in wastewater, soil, and even the bloodstream. The story isn’t just about how to make something strong and useful – it’s also about the weight of what comes after the job is done.
Getting to Know the Compound
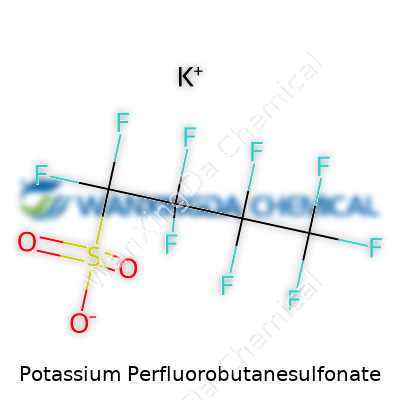
Sitting on the shelf, potassium perfluorobutanesulfonate feels like any white, crystalline powder: easy to pick up, a little slippery, not much to suggest the debates it stirs up. The structure itself features a perfluorobutyl group—a chain where fluorine atoms crowd out anything else—and a sulfonate tail partnered with potassium. This unique set-up gives the compound its resistance to chemical attack and earns it a spot in places where other molecules might buckle. Chemists saw this and set their sights on it, knowing it meant longer wear in tough environments but also recognizing the challenges that come from such stubborn stability.
Properties, Specs, and Labels
Physical properties tell a straightforward story: solid at room temperature, soluble in water, resistant to many forms of breakdown. But behind that, labeling and specifications reflect something deeper. Regulators and manufacturers tag it with warnings about environmental accumulation and persistence, showing a growing concern even as its applications expand. Technical documents don’t glaze over the point that safety measures are mandatory, not just “recommended.”
Making Potassium Perfluorobutanesulfonate
You can look up patents, old textbooks, or published research, and there’s a similar method running through—start with perfluorobutyl sulfonyl fluoride, bring in potassium carbonate, and steer it through a sulfonation reaction. Simple enough in a controlled setting, but it’s clear this isn’t amateur hour. Everything about its production needs tight oversight, from ventilation in the lab to how waste material gets collected and managed. These aren’t just hoops to jump through. They make the difference between everyday chemical progress and the kind of environmental build-up that keeps popping up in headlines.
Reactions and Tweaks in the Lab
This compound isn't flashy when it reacts, but it's tough, and chemists value just how much they can lean on it in catalysis, polymer modification, and surface treatments. Tweaking its sulfonate group sparks new properties, and its strong carbon-fluorine backbone keeps degradation at bay. The drive to alter the chain length or swap the potassium ion for other partners hasn’t stopped. Researchers keep hunting for the perfect blend of flexibility and environmental performance, hoping slight modifications can reduce the risks tied to widespread use.
Other Names in the Field
Anyone who’s sifted through material safety data sheets knows the chemical gets called several things: potassium nonafluorobutanesulfonate, PFBS-K, or the less catchy IUPAC names. This overlap in names can steer projects off track if you're not careful, especially since regulations—or calls for discontinuation—sometimes target certain synonyms but leave others in a gray area. Real experience in the lab shows that clear labeling beats clever naming tricks if you want to keep standards high and mistakes low.
Staying Safe, Staying Smart
Workplaces that handle potassium perfluorobutanesulfonate stick to strict routines. You see people wearing gloves, goggles, and masks—not just for their health, but to keep material out of drains and off their hands. The compound isn’t as acutely toxic as some persistent organic pollutants, but the science points to long-term buildup in water and living things. This is a real sticking point for community health and for those setting operational standards. Proper disposal, routine checks for spills, and written protocols separate safe use from the kind of headline-making mistakes that set back whole industries.
Real-World Uses
Waterproofing agents, firefighting foams, specialty coatings—these areas bring in potassium perfluorobutanesulfonate for consistent, tested performance. The firefighting world, in particular, relied on perfluorinated chemicals for decades, grateful for the way these molecules could smother flames that water alone never could. Over time, research showed the same traits that made it good at lingering on surfaces made it a concern for lingering in soil and water. Everyday consumers may not see this chemical listed by name, but the stain-resistant shoes, paper, and textiles in homes and offices tell the real story of how far and wide these compounds have spread.
The Research Race: Development and Toxicity Testing
I’ve watched labs move from simple performance tests toward complex toxicity research. Early studies didn’t see much acute danger, but this changed as toxicologists checked for subtler effects—how much builds up in fish, what happens in human blood, whether the compound might trigger changes after years of exposure. Toxicity research widened the lens, focusing not just on obvious harm but on things like developmental effects, hormone disruption, and the persistence of these molecules long after application. Facing this data, some companies scramble to swap out older chemicals for those that at least move faster through natural systems. Others call for even tougher rules.
Looking Ahead: Where Do We Go from Here?
Trust in the future of potassium perfluorobutanesulfonate depends more on transparency and innovation than on promises of “safe” chemistry. More and more, researchers and policymakers look for genuine alternatives—compounds that work when needed but don’t collect in oceans and bodies. Responsible research keeps the focus on tracking real-world contamination, pushing for methods that break down persistent pollutants, and staying honest about what these chemicals do once they leave the factory. Stronger standards can make the difference, but so can investing in cleanup technology, green chemistry education, and full disclosure to consumers. Long-term, I believe the chemicals that stick around in our air, water, and bloodstreams belong under the same tough spotlight, no matter how useful they seem in the short run.
The Backbone of Specialty Chemicals
Potassium perfluorobutanesulfonate, often shortened to PFBS potassium salt, shows up in places most folks never think about. I’ve crossed paths with it in industrial labs, where it pulls more weight than its unassuming name lets on. This compound belongs to a class called per- and polyfluoroalkyl substances (PFAS), which is the same family behind the infamous “forever chemicals.” PFBS stands out because of its shorter carbon chain and because some experts argue it carries less risk than its chemical cousins. That said, the public still deserves honesty about what it does in our world.
What Industries Use PFBS Potassium Salt?
From what I’ve seen and read, industries rely on potassium perfluorobutanesulfonate in specialty cleaning agents and as a surfactant to keep things stable when surface tension could cause problems. For example, it keeps electronics from suffering static discharge during manufacturing, where one zap could ruin a costly microchip. PFBS can help keep precision work possible. In fact, cleaning the inside of a hospital scanner calls for agents tough enough to cut through sticky messes but gentle enough to leave no harmful residue. More than once, PFBS-based cleaners come up on purchase orders at medical facilities, mainly because they’re strong but rinse away easily.
Besides electronics and healthcare, PFBS shows up in firefighting foam and industrial coatings. The world has begun shifting away from long-chain PFAS over health concerns, which put a spotlight on shorter-chain options like PFBS. It delivers with chemical stability and resistance to heat — two big priorities if someone’s keeping an oil rig or a military aircraft hangar safe from fire. Firefighting crews need foam that won’t break down until every hotspot’s out, and PFBS has filled the gap since legacy chemicals got phased out.
Health and Environmental Trade-Offs
Knowing about PFBS’s role means knowing about its risks. Scientific studies raise important flags about how PFAS linger in water and soil. PFBS spreads less and sticks around for less time compared to old PFAS chemicals, but it doesn’t disappear overnight. Small doses turn up in drinking water near some manufacturing sites, and scientists worry about long-term build-up. I’ve heard city water managers voice frustration that testing standards haven’t caught up yet, even as communities want answers about what’s in their tap.
The Environmental Protection Agency and similar authorities in other countries have started calling for stricter limits on PFAS, with PFBS often under review. These organizations point to evidence from animal and cell experiments, even though open questions remain about direct links to human illness. Consumers want alternatives, but the push for safer chemistry demands transparency and investment from companies that use these substances.
Choices for Industry and Community
I talk to engineers who want to shift from PFAS altogether but feel hemmed in by regulations and the lack of proven substitutes. Industry wide, the search for “drop-in” replacements moves slowly but steadily. Labs experiment with biodegradable surfactants and greener coating materials each year—a few years back, a manufacturer of electronics ditched PFAS for a plant-based cleaner, after field tests showed the new stuff worked but needed more frequent application.
Solutions do not come cheap or easy. But as the science evolves and communities demand action, companies have started opening their playbooks. This kind of accountability—alongside new breakthroughs—will push the next generation of safer, sustainable products forward. Potassium perfluorobutanesulfonate sits at a crossroads, familiar across supply chains, but it also carries the weighty responsibility of helping both people and the planet thrive.
Potassium Perfluorobutanesulfonate, known in some labs as PFBS potassium salt, has started turning up in all sorts of places—water testing, electronics, stain-resistant coatings. Whenever something with “perfluorinated” in the name lands on a chemical order sheet or local inventory, my ears perk up. Too many fluorine atoms have led to headaches in the past. The question isn’t only whether it works. The real question: what risks come along with handling it?
Practicing Safety, Not Guesswork
Every chemist or technician gets handed an SDS at some point, and for PFBS potassium salt, that document dishes out the typical safety lingo—the warnings about skin and eye irritation, dust inhalation risk, and long-term unknowns. Can I trust the SDS alone? Not really. So much of the long-term effect data for these newer, short-chain PFAS chemicals still lags behind industry use. PFBS has often been presented as a “safer” alternative for the infamous PFOS, yet water boards and toxicologists continue to urge caution, especially after studies found PFBS present in drinking water and the tissues of wildlife around the world.
The chemical seems easy to handle in terms of acute exposure. Scratches and goggles do the job for immediate splashes or dust. Even then, the powders drift off the spatula or cling unnoticed to gloves and glassware. After running a few standard lab protocols with it, I noticed colleagues reaching for extra gloves or even taping their cuffs—habits borrowed from years dealing with persistent PFAS. That kind of routine doesn’t come from the material safety sheet, but from a growing sense that invisible risks might not show up for years.
What We Know—and Don’t—About PFBS Exposure
Regulatory chatter in the US and EU keeps getting louder. PFBS might clear out of human bodies quicker than long-chain relatives like PFOA or PFOS, but “quicker” still means weeks—not hours or days. Peer-reviewed work links PFBS exposure to thyroid changes and developmental effects in animals. I haven’t seen human cases yet, but scientists spent decades chasing the health fallout from other PFAS. That’s a hard lesson to forget.
Waste is where PFBS raises red flags for me, especially in places with weak hazardous disposal protocols. Landfills won’t break down a molecule this tough. It builds up, finds water. Though PFBS does not stick in the body like PFOS, the constant trickle into the environment adds up. No one wants their lab to contribute to the next big environmental cleanup headline.
What Handling PFBS Responsibly Looks Like
So what does good practice mean? None of the old rules go out the window: close that bottle, work under a hood, wipe down the bench, double-bag contaminated tips and gloves. Staff deserve more training—more than just a read-through of the SDS or ten minutes in a safety seminar. Spills and dusting from transferring powders deserve special drills, maybe even air monitoring where heavy use happens. DIY disposal by drain or trash shouldn’t be an option, especially where municipal treatment plants can’t catch PFAS. Chemical hygiene officers need to update protocols as new scientific findings hit journals, not wait for federal agencies to catch up.
PFBS in the workplace highlights a bigger problem: having to make safety calls before the science weighs in fully. My approach leans toward caution, not just personal risk, but also for the communities and ecosystems that end up wearing every lax habit unchecked in the lab.
Stuff lands at the warehouse dock, and most people think the job is done. But just like food in the fridge, products in storage need real attention. I’ve seen what happens when bags of dry chemical sit under a leaky roof for two weeks—clumps, bad odor, and a call from a furious client. Too often, folks just stack things where there’s a bit of free space. Thinking like that creates tonight’s disaster and tomorrow’s headache.
Clean Spaces Cut Down On Trouble
Contamination sneaks up when you least expect. I worked at a place where someone left pallets near open window vents, telling themselves dust wouldn’t matter. Fast forward, the integrity of half the batch was lost to airborne grit and a strong whiff of diesel from the parking lot. Good storage comes down to one thing: keep it dry, keep it clean, and keep it sealed tight. You protect not just the product, but everyone down the supply chain—from workers to end users.
Temperature Isn’t Just a Number
The products some folks store today are not the same as they were decades ago. I’ve seen surfactants and foodstuff thicken or even spoil after a few summer days stuck near a loading dock window. Temperature swings do more than melt chocolate or soften wax—they can wreck the quality, change consistency, even create safety risks. I remember opening a drum to find a bloated vessel fizzing at the seams because it was stored outside in the sun. It’s easy to say “keep it cool”, but in a busy operation, it takes planning and reliable equipment. Clockwork temperature checks and smart logistics make all the difference.
Simple Handling Rules That Save Money
Training employees how to handle goods safely pays off more than any insurance policy. I’ve learned that overwhelmingly, warehouse accidents come from skipping this step. I saw a worker pull a pallet from the top of a half-broken stack—a cascade of bags flattened the entire aisle, and most of that batch ended up in the dumpster. Clear rules about stacking, weight limits, and keeping walkways open protect both product and people. Forklift mishaps, crushed boxes, punctured containers—these aren’t just costly, they slow everyone down.
Documentation and Regular Checks Keep People Honest
Documenting storage locations, conditions, and checks helps spot problems early. I’ve caught errors that could have cost thousands by checking a simple logbook. For many years, I thought routine checklists were just paperwork. After seeing one missed inspection lead to mold, leaks, and recalls, I now see those logs as insurance. Sometimes, a quick daily walkthrough and initialed checklist do more than expensive sensors ever will.
A Culture of Care Means Less Waste
Waste doesn’t just mean lost cases or returns—it signals a gap in the company’s mindset. I’ve watched companies that treat each product batch like something valuable always outlast those looking to cut corners. They’re rarely left apologizing to customers for short shipments or bad lots. A culture that trusts employees to flag issues early, and encourages pride over rushed work, usually keeps both product and people safe.
In the end, storage and handling aren’t just backend chores—they’re how companies protect reputations and bank accounts. A little extra care makes every load just a bit more certain, and keeps businesses thriving instead of merely surviving.
Chemistry Taking Center Stage
Potassium Perfluorobutanesulfonate stands out in industrial and scientific circles for more than just its unique name. This compound, carrying the chemical formula C4F9KO3S, brings a particular kind of presence to the table, both for what it represents in terms of chemical structure and in its broader story around environmental and human impact. Its molecular weight, clocking in at 348.19 grams per mole, might seem like a dry fact. But it sets the foundation for its physical behavior, how it dissolves, moves, or resists change in water and other solutions.
Transparency and Trust in Chemical Use
Chemists and those monitoring product safety get a pretty clear signal from a molecule’s formula. With this substance, the backbone built from carbon, fluorine, potassium, and sulfur isn’t just for show. Perfluorinated chemicals, as a category, don’t just break down easily. They build reputations for sticking around in the environment and showing up in unexpected places—including living organisms. Because of this, the transparency about what’s in a chemical, down to the last atom and exact weight, becomes essential for public trust.
Practical Uses: Where It Shows Up
Potassium Perfluorobutanesulfonate finds its name printed on lists of substances used in research, especially in the analysis of difficult-to-separate mixtures by advanced chromatography. It doesn’t excel at grabbing headlines like some more infamous per- and polyfluoroalkyl substances, but it shares the same class. Its stability and resilience might solve one problem in a lab, yet create others out in the wider world. Once released, these molecules do not ghost the environment; they persist, outlasting many attempts at clean-up.
Weighing Safety Against Utility
The sometimes-uncomfortable trade-off at work here asks whether convenience or performance justifies adding more long-lived chemicals to landfills and water supplies. I’ve seen plenty of academic papers balancing the needs of progress with the needs of people’s health. These chemicals get into our bodies, even if only in microscopic amounts, and health data keeps growing. Public scrutiny rises each time a study draws new links between long-term chemical exposure and what that can mean for everyday folks. Regulators don’t want to play catch-up forever, so formulas and molecular weights now show up online even for obscure substances.
Stepping Toward Accountability
Conversations between communities, researchers, and government folks help keep us on track toward solutions. Some advocate switching to shorter-chain fluorinated alternatives, hoping they're less likely to stick around or build up in nature. Others bank on stronger regulatory frameworks to cut down unnecessary use, especially in throwaway products. I think the most progress happens when users understand exactly what’s in their labs or local water. Sharing full details like molecular formula and weight isn’t just an academic exercise. That information lays the groundwork for holding producers and regulators to account, while driving innovation in safer, more sustainable chemistry.
Potassium Perfluorobutanesulfonate might not ring a bell for your average person on the street, but you’ll find it behind the scenes in firefighting foams, some industrial cleaners, and specialty chemical processes. Its name tells part of the story: it's a perfluoroalkyl substance, or PFAS, a family of chemicals known for sticking around in the environment longer than just about anything else. This "forever chemical" label is no exaggeration—what goes into your soil, water, or air today is going to hang around for decades.
Talking to folks in the field, you hear the same concerns over and over: these compounds build up in soil and water. Fish take them in, then birds and bigger animals eat the fish, and on up the food chain they go. The fact that potassium perfluorobutanesulfonate resists breaking down means that long after spills, leaks, or runoff, small amounts keep entering the food supply. This isn’t abstract—it’s the real world experience of communities living near manufacturing sites or airports, where firefighting foam discharges can be more common.
The human health story gets complicated. Scientists have flagged longer-chain PFAS, like PFOA and PFOS, for years; both have well-established links to certain cancers, immune disruption, and developmental issues. Potassium Perfluorobutanesulfonate sits on the shorter end, with a four-carbon backbone. Some researchers hoped that shorter PFAS would pose fewer problems, but the track record hasn’t delivered clear safety. Studies on animals point to liver toxicity, minor changes in cholesterol, and other subtle health shifts with higher exposures.
Folks often ask, if the dangers haven’t reached crisis level, why worry? The trouble comes down to cumulative effects. PFAS chemicals, short or long, don’t just flush out—they stick inside people, fish, livestock. Drinking water samples from across the globe already register measurable traces. The U.S. Environmental Protection Agency has started setting stricter advisory levels for PFAS in drinking water, and the list keeps growing.
If you spend enough time talking shop with water engineers, you’ll hear frustration about slow-moving rules and the uphill battle of removing these chemicals once they’ve entered the system. Granular activated carbon filters pull out a big chunk, but not all. Smaller utilities or homes run into cost hurdles. The technology’s out there, it just needs money and willpower behind it.
So what’s the next move? Public disclosure makes a difference. Communities deserve straight talk about what’s falling into their water and what it may mean for future generations. Cleanups must aim at the source, not just treat the symptoms. Manufacturers have started researching alternatives that break down easier in the environment, and progressive companies already invest in safer chemistries.
Transparency from companies and regulators helps. Pushing for more research is just as important—particularly independent studies that track real-world health and environmental changes, not just controlled lab settings. Switching out hazardous compounds for ones we understand even less should not become the norm.
Potassium Perfluorobutanesulfonate isn’t the worst player in the PFAS category, but waiting for full disaster before acting on science has never paid off. Solutions start with open eyes, honest science, and a willingness to put public and environmental health before short-term convenience.