Potassium Trifluoroacetate: Chemical Reliability in a Rapidly Shifting World
The Chemical That Kept Quietly Growing
Potassium trifluoroacetate sits quietly on the registry of modern reagents, but its history goes back longer than most chemists think. Discovery traces to research in the mid-20th century, chasing new routes in organofluorine chemistry just as global industries started to hunger for precise, reliable catalysts and sources of the elusive trifluoromethyl group. I remember walking through a laboratory where one senior researcher remarked, almost off-hand, how potassium trifluoroacetate once belonged only to esoteric organic synthesis and then, almost overnight, found its place in major production lines for pharmaceuticals and agrochemicals. That’s a hint of why small-molecule chemistry tends to surprise us; the reagents nobody noticed in the ‘60s grow into cornerstones of today’s research pipelines.
Looking at the Product up Close
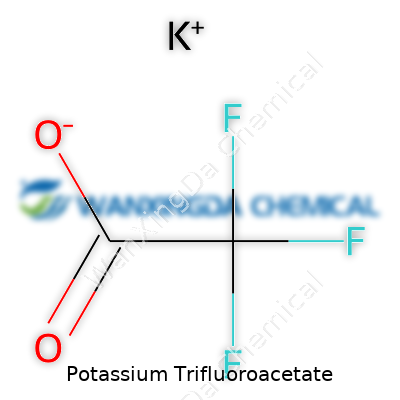
Potassium trifluoroacetate pours out as a white, crystalline solid, usually with a slightly sharp, acidic tang wafting from an open container. Its chemical formula CF3CO2K makes it a salt of trifluoroacetic acid, replacing hydrogen with potassium. That single shift turns a volatile, corrosive acid into a stable reagent that handles shipping, storage, and lab life with far less drama. I have seen researchers looking for consistency in source material appreciate the way this salt refuses to clump up in humid conditions and how it dissolves cleanly in water or organic solvents, giving them flexibility to design reactions that need a dose of trifluoromethyl power—without dragging in sickening odors or corrosion trouble. Boiling and melting points, solubility, and density rounds out the basic technical knowledge every chemist uses, and the numbers here reflect stability and broad compatibility, not just lab convenience.
Bottle and Label: What Matters in Real Life
Proper labeling for potassium trifluoroacetate often reflects international standards, and anyone buying in bulk or for institution-grade research expects clear designations with molecular weight, assay, batch number, and hazard information up front. Lab veterans look for certifications showing compliance with safety and purity, since errors in labeling or careless packaging spell operational delays, not to mention potential danger during large batch scaling. In practice, these protocols gave peace of mind each time a student or technician grabbed a container off the shelf, cutting down on accidents and expensive missteps.
Making the Salt: How Potassium Trifluoroacetate Gets Prepared
Industrial and lab recipes typically run the reaction of trifluoroacetic acid with potassium carbonate or potassium hydroxide, generating water and carbon dioxide as byproducts. Simplicity, predictability, and scalability lead to a process easy to repeat, both for small research quantities and for industrial-grade supply. From personal experience in an academic setting, the straightforward synthesis allowed even undergraduates to participate safely in standard preparations, as long as ventilation and gloves were prioritized. This simplicity reduces cost at scale and opens doors for customizations, since chemists sometimes tune the counterion composition or solvent system based on downstream plans. Modifications stay minimal since the salt itself usually acts as the carrier for the highly prized trifluoromethyl group.
The Chemistry of Reactions and Pathways Forward
Potassium trifluoroacetate steps up as a source of trifluoroacetate ions in cross-coupling reactions and as a mild base or nucleophile in select transformations. Decarboxylative trifluoromethylation stands out, especially in palladium or copper-catalyzed protocols, unlocking routes to compounds that, only decades ago, stayed out of reach for working chemists. In my years collaborating with medicinal chemistry colleagues, I saw how the precise reactivity of trifluoromethyl groups sped up lead optimization, improving biological stability and membrane permeability in drug candidates. The advantage also extends to crop protection chemicals, where fluorine boosts resistance to degradation in the field. Researchers keep tweaking conditions and metal systems, hoping to coax out even cleaner and more efficient transformations for commercial scale, since each saved step translates into less waste and lower cost.
Aliases: Identity Beyond a Single Name
Potassium trifluoroacetate shows up in the literature under several aliases—potassium trifluoroacetate, trifluoroacetic acid, potassium salt, potassium trifluoroethanoate—with each variant a nod to the same chemical backbone but reflecting regional preferences or naming conventions based on supplier or research tradition. People working in regulatory agencies or reviewing patent filings often need to track these synonyms to get a complete understanding of the landscape. When searching archives or ordering material from global sources, navigating these aliases often brings unexpected results, especially for those less familiar with the web of systematic and trade names.
Working Safe: Real Concerns, Real Solutions
Comfort with potassium trifluoroacetate often stems from its reputation as stable and easy to handle. While not aggressively dangerous, the compound deserves respect for its irritant potential, especially on contact with skin, eyes, or in cases of inhalation of the dust. Those of us who have prepared or handled compounds in less controlled settings recognize the sharp, dry feeling on exposed skin—a stern reminder to use gloves and masks. Safety data sheets recommend standard lab protocols: avoid ingestion, wash with water upon contact, and store away from strong acids or oxidizers. In laboratories, education and discipline set the standard, creating habits that translate away from the bench as well. Well-run labs put safety reminders up high, but culture also matters; seeing everyone, from professors to first-year students, suit up for apparently “tame” chemicals keeps the routines strong for all compounds.
Where Potassium Trifluoroacetate Goes to Work
Major growth comes from pharmaceutical, agrochemical, and advanced materials fields. In drug design, the trifluoromethyl group improves metabolic stability and bioavailability; potassium trifluoroacetate provides chemists a tool to install this group cleanly and reliably. In the synthesis of specialty fluoropolymers, electronics, and liquid crystals, its role as a trifluoroacetate ion source carves out space in materials science research, both for niche polymers and emerging battery technologies. Many companies leverage its mildness to introduce precision effects in organic compounds—tricking out molecules to better survive tough industrial processes or last longer in storage.
Tracking the Cutting Edge: New Research and Opportunities
Recent breakthroughs focus on extending the reactivity landscape of potassium trifluoroacetate. Work in photoredox catalysis and new decarboxylative cross-coupling protocols aims to lower the temperature and solvent requirements, making sustainable and scalable manufacturing more than just a buzzword. Colleagues in green chemistry circles regularly discuss how tweaking the ligand and metal partners in these transformations could unlock routes to trifluoromethylated building blocks using less energy and generating less waste. There’s a growing appetite for entire reaction cascades using flow chemistry or continuous manufacturing. Academic groups and startups look for partnerships to translate bench advances into industrial scale with fewer re-optimizations—a shift that could reshape the entire business of specialty chemical supply.
Studying Safety: Toxicity and the Need for Vigilance
Research into the toxicity of potassium trifluoroacetate reports low acute toxicity compared to many industrial salts, but concerns remain about chronic exposure and environmental release. Stories still circulate about fluorinated compounds building up in water tables or soil, raising red flags for large-scale operations even when human toxicity sits lower on the immediate risk spectrum. In academic safety meetings, reminders circle back to the importance of contained waste handling and water treatment. Regulatory shifts in parts of North America and Europe monitor fluorinated compound runoff more tightly, so vigilance becomes a daily routine instead of just a compliance checkbox.
Where Potassium Trifluoroacetate Might Go Next
Looking ahead, its role keeps expanding as industries hunt for new ways to incorporate the trifluoromethyl group faster, cheaper, and with less downstream cleanup. Companies want plug-and-play reagents for modular synthesis, cutting down on steps and specialized equipment. Green chemistry will push the field to design more biodegradable or recoverable trifluoroacetate sources. At the same time, regulatory scrutiny on fluoride and persistent organic pollutants guides innovation, urging a balanced approach where reactivity meets responsibility. For those of us who value both safety and scientific progress, seeing open communication between academia, industry, and public advocates around potassium trifluoroacetate’s handling sets a hopeful example. Commitment to smarter synthesis, better safety standards, and honest discussions about environmental footprint will keep this quiet, unassuming reagent firmly in the spotlight for years to come.
Digging Into Its Role in Modern Chemistry
Any time someone mentions potassium trifluoroacetate, the average reader probably pictures a laboratory shelf lined with mysterious bottles. Truth is, this white, crystalline powder plays a bigger part in advancing science than its name lets on. Its main use pops up in the organic chemistry world, especially for making carbon-fluorine bonds — a job that’s trickier than most would expect.
Behind the Bench: What It Does in Real Labs
Potassium trifluoroacetate has gained a reputation in research labs. Synthetic chemists look for shortcuts and clean reactions, and this salt offers just that. It starts as a stable, easy-to-handle alternative to nastier fluorinating chemicals. You see it feature often in trifluoromethylation, a technique that shoves a -CF3 group onto molecules. This trifluoromethyl group rarely shows up in nature, yet it holds real value for pharmaceuticals and agrochemicals, boosting metabolic stability and biological activity. Many blockbuster drugs—ranging from antidepressants to cancer medications—contain at least one fluorine atom. This is where potassium trifluoroacetate makes its mark.
Personal experience around a synthetic chemistry group taught me to appreciate practical advantages in the lab. The usual suspects for introducing fluorine—like trifluoromethyl iodide or even sodium trifluoroacetate—bring headaches around toxicity, cost, or limited accessibility. Potassium trifluoroacetate swaps danger for dependability. It works as a reagent or a source of trifluoromethyl groups, especially under copper catalysis. Instead of gearing up with gas-tight syringes or wrestling with hazardous materials, labs turn to this potassium salt for its low fuss and moderate reactivity.
Why Does It Matter?
Drug development doesn’t slow down for hazardous chemicals. Researchers hunt for greener, safer, and more cost-effective protocols. Potassium trifluoroacetate fits this need, letting teams design new molecules while lowering their risk exposure. Over the years, published methods using this salt have spread in the academic literature. It features in methods for aryl trifluoromethylation, often producing higher yields with fewer byproducts. Green chemistry pushes for less waste and safer working conditions, and this is one reagent that already fits the bill.
There’s also the speed that comes with a stable solid. No need to chill everything with dry ice or scramble to use up a volatile chemical before it breaks down. In practice, potassium trifluoroacetate dissolves readily and survives a standard shelf life. This efficiency lets scientists focus on invention rather than logistics.
Room for Improvement and Next Steps
Potassium trifluoroacetate isn’t perfect. It asks for metal catalysts—often copper—which brings cost and sustainability concerns. Recycling copper and reducing its use remain subjects of current research. Accessibility also depends heavily on established suppliers, which could bottleneck growth if demand spikes. Labs are already tinkering with ways to tweak the underlying reactions, aiming to substitute cheaper or reusable metals and streamline purification.
Scaling up from the benchtop to manufacturing means keeping safety at the center. Teams designing new molecules for industry watch their costs and waste closely. Wider adoption will require more robust ways to recover both copper and trifluoroacetate residues, along with tighter supply chain monitoring. But judging by its steady gain in popularity, potassium trifluoroacetate will likely keep showing up, proving that chemistry can evolve toward both safety and efficiency without killing discovery along the way.
Understanding the Formula
Chemistry sometimes brings us to compounds that look simple on paper but end up playing a big role in research and applied science. Potassium trifluoroacetate falls into this category. Its chemical formula, C2F3KO2, tells a lot in a handful of letters. Each molecule comes packed with two carbon atoms, three fluorine atoms, two oxygens, and the ever-familiar potassium. Anyone who’s handled common salts like potassium chloride might not think much at first—but one look at the structure and things get interesting, fast.
The Skeleton of the Molecule
The structure builds off trifluoroacetic acid—swap out a hydrogen for potassium and potassium trifluoroacetate emerges. At the core is a two-carbon backbone, one fully loaded with three tightly bound fluorine atoms (the trifluoromethyl group), and the second part forms a carboxylate group. The potassium ion balances the negative charge left behind by the oxygen atoms, sticking close, but not so close that it forms a covalent bond. It’s a classic example of ionic interaction you learn in basic chemistry, but with all the extra electronegativity packed into those fluorines, the compound gets a unique personality.
Why Structure Matters
This isn’t just molecular trivia. Small tweaks in structure change how a compound behaves. The three fluorines seriously crank up the electron-withdrawing power, which means this salt can act very differently than regular potassium acetate. In synthetic chemistry, that switch gets noticed. The presence of so much fluorine can influence acidity, solubility, and the way molecules react in a lab environment.
The combination of properties has led to potassium trifluoroacetate being favored in a niche set of chemical reactions, especially for chemists looking to bring fluorine into organic compounds—a move that’s important for designing new pharmaceuticals, for instance. Many drugs feature a fluorine atom because it can make drugs more stable or help them reach the target site in the body more effectively. Getting fluorine onto a molecule in a controlled way depends on reliable building blocks, and this salt delivers.
Safety and Practical Use
Lab work always comes with a safety angle. Potassium trifluoroacetate brings with it the standard safety considerations of a strong ionic organic salt. It doesn’t explode. It doesn’t dissolve your gloves, but those fluorines mean you treat it with respect. Everyone in a real lab knows to gear up—gloves, goggles, good ventilation. That’s not paranoia, just preventive wisdom picked up from handling all sorts of chemicals over the years.
Some newer chemists don’t fully appreciate the environmental load that compounds like this can pose. The fluorinated part doesn't break down easily. Over time, these compounds can accumulate. Working toward greener chemistry really means finding routes that use less persistent materials, or set up systems for safe recovery and disposal. This isn’t impossible—teams worldwide already push for processes that recycle solvents, reduce waste, and take care with fluorinated chemicals.
Moving Chemistry Forward
Potassium trifluoroacetate seems unassuming—just some carbon, potassium, oxygen, and a few highly reactive fluorines. But in hands-on organic chemistry, it enables reactions that once looked tough on paper. It shows how attention to core chemistry, a respect for safety, and a push for greener labs drive science forward—a formula as sound as the compound itself.
What’s At Stake with Potassium Trifluoroacetate?
Few things wake up your attention in a lab quite like strong chemicals. Potassium trifluoroacetate packs a punch in organic synthesis labs thanks to its unique reactivity and use in specialized reactions. It’s not on the short list for backyard chemistry or art class, but for researchers, getting the details of its safe storage and handling makes the difference between routine work and a safety incident.
The real importance of this topic jumps out if you’ve spent time around chemical supplies. One day the bottles are lined up, clean and undisturbed. Next, a mishap leads to a powdery mess, a whiff of sharp odor, or a stained glove, all because storage or handling shortcuts crept in. Experience teaches: you respect the risks or they’ll teach you respect the hard way.
Understanding Potassium Trifluoroacetate’s Challenges
Potassium trifluoroacetate isn’t radioactive or wildly flammable, but it brings together strong ionic character with the biting acidity of fluorine. Accidental contact causes irritation and overexposure raises health concerns. Regardless of the hazard level on official signs, too many underestimate these reagents, treating them like everyday salt or baking soda. Mistakes follow from underestimating risk or overestimating one’s luck.
This compound absorbs moisture from the air, clumps fast, and loses its purity if left out. I once worked in a lab where someone “borrowed” a container and forgot to recap it. A few days later, the bottle held a brick instead of a free-flowing powder. Apart from the loss, nobody wanted to figure out what else had drifted in with the humidity.
Practical Steps That Make a Difference
Chemical safety usually boils down to practical habits and respect for the material. Potassium trifluoroacetate’s quirks mean it belongs in a tightly sealed, chemically compatible container. Polyethylene and certain glass containers hold up well. An open bottle left on a crowded shelf spells trouble: moisture sneaks in, and the compound loses both shelf life and effectiveness.
Store it in a cool, dry spot. That means away from steam, bright light, and sources of accidental water. Clear, direct labeling helps anyone in the lab know what’s in the bottle and when it came in, which really increases safety. The days you discover a decades-old, unidentified powder lurking on a back shelf are the days your heart skips.
Gloves, safety glasses, and a sturdy lab coat go a long way. Don’t skimp, even if the work feels familiar. Gloves slow down skin contact, and eyewear blocks the chance of a splash heading for your eyes. If you handle more than a pinch, a chemical fume hood pulls dust and vapors away from your breathing space.
Supporting Smarter Handling
Ongoing training helps everyone recognize why these controls matter. OSHA and university guidelines offer clear direction because they’re built on lessons from accidents, not rules for rules’ sake. Researchers who join training sessions once a year may roll their eyes, but the moments after a near-miss always drive home the “why” quite clearly.
Disposal wraps up the safety story. Clean up spills with absorbent material, avoid pouring into drains, and seal waste for hazardous disposal. Rushing the job or skipping the paperwork may feel efficient, but it brings headaches in regulatory visits or future clean-ups. Pairing sound practice with a little vigilance keeps labs safe and science on track.
What Actually Matters with Chemical Safety?
I’ve spent years checking chemical labels, reading safety sheets, and hearing the familiar question: “Is it toxic?” Few names raise as many eyebrows as potassium trifluoroacetate. That mouthful of a name isn’t something most people hold in their kitchen cupboard, but it shows up at a surprising rate in the lab and in industrial circles. Curiosity is justified, especially once you see the word “fluoro” in anything chemical. These compounds sound scarier than they are—or sometimes, not scary enough.
Here’s the deal: potassium trifluoroacetate isn’t something the average person faces daily. Researchers and chemical workers run into it more often. Its main gigs include helping along organic syntheses and sneaking into processes where strong bases are needed. Most folks outside a lab won’t even see it written down, much less handle it. Still, the safety picture deserves close attention.
Is Potassium Trifluoroacetate Toxic?
Let’s break it down. Potassium trifluoroacetate is not on the list of chemicals that kill or seriously sicken with a slight whiff. The key issue is the trifluoro group, which connects to worries about long-lived compounds that stick around in bodies or the environment. Many researchers flag “perfluorinated” chemicals as ones to approach with caution. Some of those, especially the bigger molecules like PFOS or PFOA, have earned a bad reputation for environmental persistence and potential links to cancer or hormone disruption. Potassium trifluoroacetate is smaller and doesn’t fit perfectly in with those infamous long-chain compounds.
Direct contact could irritate skin, eyes, or airways if not handled right. Swallowing a bunch would probably bring on a bad stomach day and maybe worse, but you’d need more than a fingerprint’s worth. Long-term toxicity data stays thin, which really makes the call for careful handling stronger. Science hasn’t closed the case on whether this compound quietly causes trouble over time or in low doses.
Real Risks: It’s in How You Use It
I’ve watched people ignore gloves or eye shields around seemingly tame chemicals. A lot of hurt comes from this attitude. Potassium trifluoroacetate, for all its lack of headline-making toxicity, will give you burns if you get it in your eyes or leave it on your skin. It can take moisture from the air, so it turns sticky fast, making spills harder to control. Inhaling powder or vapors is never smart—doing so invites trouble.
There’s a genuine conversation about its breakdown products too. Trifluoroacetate ions don’t go quietly into the night once poured down a drain. Water treatment plants struggle with them, building up low levels in the wild. Nobody wants a repeat of the mishaps seen with Teflon-linked chemicals, so smart labs plan to trap and contain any waste.
Practical Solutions for Safer Handling
Controls and training need to match the real-world risks. Solid gloves, safety glasses, and good ventilation keep problems away. I’ve seen labs cut incidents by half just by running short reminders before pulling bottles off a shelf. It’s also smart to rethink disposal—special waste containers do a far better job than flushing anything with “trifluoro” in the name. Clear guidance keeps new researchers out of trouble, and periodic reminders help veterans stay sharp.
The importance of respecting even low-profile chemicals like this one can’t get overstated. It’s easy to get comfortable and then end up with a preventable chemical burn or environmental mishap. Real safety grows from informed habits, not fear or complacency. Potassium trifluoroacetate keeps proving the point: the chemicals themselves rarely jump off the shelf—how we handle them always matters more.
Potassium trifluoroacetate doesn’t end up on grocery lists or mainstream shopping carts. This chemical lands on the radar of people who spend their days in the lab, turning ideas into real solutions and discoveries. Compared to many household or industrial salts, this one plays in a specialized arena, mainly tied to organic synthesis, pharmaceuticals, and a few high-tech niches.
Why People Seek Out Potassium Trifluoroacetate
As someone rooted in experimental chemistry, I’ve watched colleagues puzzle over reagent quality. In the lab, even a trace impurity can reroute weeks of planning, so the hunt for chemicals like potassium trifluoroacetate starts with an obsession for purity. Synthesizing certain compounds or tuning reaction conditions often pushes researchers to seek grades labeled above 98% and, depending on the application, chase the edges of what suppliers can produce, sometimes even paying double for a percent’s difference.
Where the Search Begins
Buyers usually turn to established chemical suppliers, not fly-by-night websites. Reputation matters, and so does traceability. Companies with a strong record of supplying reagents for academic and commercial research—think names rooted in decades of chemical distribution—tend to carry potassium trifluoroacetate in their catalogs. Researchers head straight for suppliers who list lot analysis, quality control data, and documentation that back up stated purity.
Most online sales run through certified distributors serving labs and universities, and their storefronts aren’t open to just anyone. There are obvious reasons: potassium trifluoroacetate isn’t dangerous on its own, but regulations around chemical sourcing in many countries aim to intercept suspicious procurement of specialty chemicals. Some regions set up purchase requirements—documentation, institutional proof, or end use declarations. The hoops may frustrate, but they help keep lab work above board.
Quality often tops out at 97-99%, with high-grade material targeting sensitive syntheses or analytical work. I’ve seen purities drop below that in surplus or bargain listings, but seasoned chemists rarely gamble with unknown sources. Subpar purity can drag down yields, introduce side products, or cause safety issues, especially in pharmaceutical or catalyst research.
The Struggle for Reliable Access
Researchers outside major institutions sometimes hit roadblocks. Small businesses, start-ups, and educational projects face higher prices or minimum order hurdles. Occasionally, a respected supplier runs out of stock or updates import rules, and sourcing the reagent becomes an exercise in negotiation. Cooperative purchasing or tapping into university networks sometimes helps ease these bottlenecks.
Rogue sourcing from unverified online sellers still tempts the desperate, but it’s a shortcut rarely worth the risk. Even the best protocols can’t compensate for contaminated material. In my own work, a single batch of low-grade chemical forced a team to repeat an entire series of tests, wasting weeks. Trustworthy suppliers might cost a little more, but they bring the peace of mind that every scientist needs.
Building a Stronger Supply Chain
Solutions for better access usually focus on transparency and cooperation. More robust online verification processes speed up legitimate purchases, while coordinated databases of reagent quality help catch bad actors early. Research communities have started openly sharing supplier reviews—an effort that nudges standards higher and encourages chemical companies to publish more detailed analysis certificates.
Start with the right supplier, check the paperwork, and don’t let shortcuts tempt you. Purity isn’t just a lab number—it shapes the outcome of every project that depends on it.