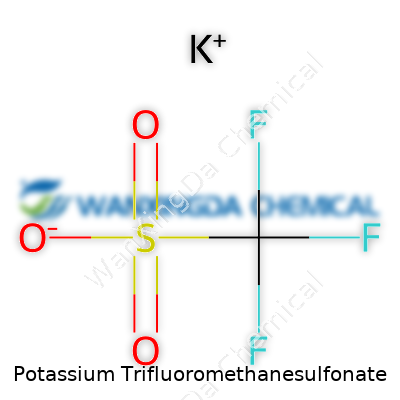
Looking at Potassium Trifluoromethanesulfonate from Every Angle
Historical Development: A Shift Toward Cleaner Chemistry
Chemistry moves forward on the back of practical tools—useful reagents, versatile intermediates, and robust catalysts. Potassium trifluoromethanesulfonate, or potassium triflate, didn't appear out of thin air. It showed up in the lab as fluorine-based chemistry matured, especially in the second half of the twentieth century. Earlier, labs stuck with traditional inorganic salts like potassium chloride or nitrate, but as synthetic ambitions grew bolder, the chemists needed something more. The hunt for stable yet highly reactive sources of potassium led researchers to triflate, marrying the chemical muscle of trifluoromethanesulfonic acid with the reliability of potassium salts. Now, potassium triflate stands as a staple in organic, medicinal, and even industrial chemistry, signaling a shift from the older world of harsh acids and complicated reaction workups to cleaner, often milder transformations.
Product Overview and Practical Details That Matter
Anyone working with potassium triflate quickly realizes it supports a blend of performance and consistency. This white crystalline powder resists caking, stores well under dry air, and dissolves with surprising ease in water and polar organic solvents. Chemists value these features not for their own sake, but because triflate salts tend to speed up reactions and simplify workups—critical when time, money, and material all matter. The potassium ion plays a secondary but important role: it enters a crowded field of metal cations but avoids introducing secondary complications, like heavy metal contamination or unwelcome redox activity. Over time, this unassuming salt has become far more than a curiosity—it finds its way onto packing lists for everything from pharmaceutical scale-ups to advanced materials research.
Physical and Chemical Properties: Why Chemists Keep Coming Back
Physical and chemical properties speak louder than brand names or shiny packages. Potassium triflate has a molecular formula of CF3SO3K and a molar mass that fits neatly into most calculations. It’s got a melting point that lets you dry it with confidence—no need to fret about decomposition at moderate heat. In solution, potassium triflate holds its own as an inert counterion, rarely interfering with the actual chemistry taking place. Chemists working on Suzuki couplings, SN1 substitutions, or cationic polymerizations reach for potassium triflate because its anion barely interacts with hard nucleophiles, stabilizing everything from carbocations to metal complexes without joining the reaction itself. Water and acetonitrile dissolve it readily, and that versatility widens the toolbelt for anyone handling both organic and inorganic transformations in a single workflow.
Technical Specs and Labeling: What Actually Ends Up on the Bottle
Walk into any storeroom, and you’ll see potassium triflate labeled with its molecular weight, its CAS number, and purity grades—most labs buy 98% or better. Labeling looks dull, but these dry details let researchers order the right material, store it safely, and avoid mishaps. Potassium triflate isn’t light-sensitive, but it does appreciate dry conditions, and most vendors put shelf life and storage tips right on the label. That might sound mundane, but after running experiments for days or weeks, one mixup on the bottle can mean lost work or even wasted grants. Reliable labeling isn’t bureaucracy—it’s insurance for the chemist’s time and sanity.
Preparation: How Do We Get from Theory to Useful Powder?
Industrial production of potassium triflate reflects chemistry’s balancing act between elegance and scale. The old-school method—neutralizing trifluoromethanesulfonic acid with potassium carbonate or potassium hydroxide—generates the salt in a single step. Remove the byproduct gases and water with vacuum or gentle heat, and you’re left with a solid that gets recrystallized for high purity. This isn’t a glamorous process, but it’s reliable and straightforward. Some labs take it further, tailoring the process with solvents like acetonitrile to boost purity or grinding up smaller batches for specialty synthesis. The key is consistency: batch-to-batch variability ruins reproducibility in research and manufacturing, making refinement ever more crucial as potassium triflate finds new roles in high-precision fields.
Chemical Reactions and Modifications: Fueling New Chemistries
Potassium triflate’s main claim to fame lies in its use as a supporting electrolyte, catalyst base, or reactant in organic synthesis. Chemists use it to make triflate esters and to introduce triflate as a leaving group—one of the most reliable avenues for tricky SN1 or SN2 reactions. In complex molecule construction, potassium triflate opens doors to aryl triflates, which in turn breathe life into cross-coupling reactions. The salt stays mostly inert, but its very passivity lets it shepherd sensitive intermediates, lending stability without mucking up reaction profiles. Tinkering with its concentration, or swapping the potassium for other cations, lets researchers fine-tune outcomes across a range of subfields, from medicinal chemistry to photolithography.
Names and Synonyms: Not Just Another Salt
Names in chemistry matter, and potassium triflate wears several. The IUPAC mouthful, potassium trifluoromethanesulfonate, spells out its structure. Others call it potassium trifluoromethanesulphonate or, more simply, potassium triflate. Synonyms get tossed around in patents or trade journals, so anyone working in the field needs to keep these straight. Mistaking sodium for potassium, or confusing triflate for a sulfate, turns tomorrow’s plans into today’s mistake pile. Clarity in naming helps chemists keep their priorities straight.
Safety and Operational Standards: Protecting Both the Scientist and the Science
Safety doesn't play second fiddle in any well-run lab. Potassium triflate gets categorized as a low-hazard salt, but its handling still calls for gloves, safety goggles, and proper ventilation. Inhalation of fine particulates, eye irritation, or accidental ingestion remain risks if care slips. Labs put faith in their Material Safety Data Sheets—most highlight stable storage, basic spill protocols, and environmental guidelines. Waste triflate solutions should never go down the drain unexamined; routine compliance with hazardous waste standards transforms a well-equipped lab into a responsible neighbor. These procedures seem routine only because they finish the unfinished business of chemistry’s checkered safety past—care on the bench top reduces headaches, legal or medical, for everyone.
Applications That Matter: Science, Industry, and Beyond
Potassium triflate’s story gets interesting at the bench, but its impact radiates outward. In organic synthesis, it provides a route to exploring new pharmaceuticals, agrochemical leads, and specialty polymers. Analytical chemists tap its strong dissociation and excellent solubility for non-aqueous titrations and as a calibration standard in NMR and mass spectrometry. Engineers and materials scientists study its role in dots, wires, or films—any context where inert but highly polarizable anions could tip the balance. Outside the lab, potassium triflate sometimes features in battery research, organic electronics, and clean energy projects. Reliable performance in all these fields testifies to its adaptability and the cross-disciplinary thinking that chemistry prizes.
Research and Development: Pushing Past the Limits
No salt—no matter how trusted—escapes the push for better performance. Teams worldwide look beyond simple triflate salts, seeking modified versions that further minimize background reactions or optimize solubility in green solvents. Some labs tinker with mixed-anion systems hoping to unlock exotic reactivity. Others dive into computational studies, modeling ion pairing and solvent effects to wring every drop of efficiency from each molecule. Funding agencies and industry leaders reward these efforts, recognizing that a tweak in a supporting electrolyte can ripple upward, trimming process costs and environmental impact on a grand scale. Researchers now blend real-world trial and error with machine learning—an approach that spreads across journals faster than old prejudices about what “standard” reagents can do.
Toxicity Research: Understanding Risks, Not Guessing
Potassium triflate’s safety record looks solid, but chemists and toxicologists don’t leave well enough alone. Early studies focused on acute toxicity—what happens with accidental exposure during handling. Results suggested low oral and dermal toxicity in lab animals, minimal environmental hazard if cleaned up quickly, and little risk of bioaccumulation. Ongoing research tries to capture long-term effects, degradation pathways, and subtle ecological impacts, especially as large-scale uses in energy storage or manufacturing expand. For now, the absence of grim headlines owes less to luck and more to careful study and straight talking about unknowns—a culture shift that sets real science apart from speculation.
Future Prospects: Chemistry’s Search for Even Better Tools
Potassium triflate stands at an interesting crossroads. As labs chase green chemistry solutions and regulatory agencies tighten up the rules, low-toxicity, non-volatile, and highly functional salts like this one attract more attention. The move toward sustainable industrial synthesis keeps potassium triflate in the running as both a building block and an enabling reagent. Success in developing biodegradable derivatives or salts with tunable properties could push this old favorite into entirely new markets—think energy storage, next-generation catalysts, or single-use medical devices. Real progress means reaching for both incremental improvement and radical innovation. Chemists who trust potassium triflate right now will likely see it either transformed or superseded by something even more elegant down the line, but its durability as a lab standard proves it’s not going anywhere soon.
Some chemicals sound daunting, and potassium trifluoromethanesulfonate sits high on that list. People call it potassium triflate for short, which helps take the edge off. In my time at the lab bench, even the name alone used to catch my eye. But behind the tongue-twister lies a humble but powerful helper that most folks outside a lab never hear about. Digging into its place in research, it stands as a reminder that progress often depends on tiny, obscure players doing a quiet job.
Why Chemists Reach for Potassium Triflate
Potassium triflate enters the scene mostly in organic synthesis. Every day, chemists must find ways to coax molecules into joining, swapping parts, or falling apart in just the right ways. In many of these journeys, you need a counterion—a partner that balances charges and keeps things moving. Potassium triflate holds its own here. Its triflate part, loaded with fluorine, stays out of the action, not sticking or gumming up the works. This non-interference matters when a clean, predictable result is the goal.
People often add potassium triflate to make new types of bonds, like connecting carbon to oxygen or nitrogen. It helps form strong intermediates and lets other chemicals do their job without crowding in or making a mess. I have seen colleagues struggle with other salts—like chlorides or nitrates—leaving behind tough-to-remove residues or unpredictable byproducts. Switching to potassium triflate brought smoother reactions and easier purification. That kind of practical benefit keeps it in the toolkit.
Triflate Salts and Their Growing Role
Research labs aren’t the only customers. In battery and electronics research, triflate salts, including potassium triflate, support the hunt for faster-charging and more reliable devices. Their chemical stability and solubility in organic solvents let engineers push electrolytes to new limits, something ordinary salts can’t always manage. As technology pushes for greener solutions and more efficient energy storage, small actors like potassium triflate back up the effort behind the scenes.
Another arena worth watching is pharmaceutical research. Drug makers often deal with molecules rich in fluorine, given that fluorine atoms can make a big difference in how medicines behave in the body. Fluorinated reagents like triflate salts help build these molecules, opening up new design pathways for curing disease or treating chronic illness. The right supporting chemicals, even if they don’t end up in the final pill, help keep drug pipelines moving and prices down.
Concerns and Smarter Use Going Forward
Rarely does a supporting chemical become a headline risk, and potassium triflate stays out of news for toxicity or environmental impact. Still, lab safety calls for respect: like most chemicals, it needs thoughtful use, safe disposal, and a sensible supply chain. In my own work, getting rid of the saltwater from a reaction used to create a headache, but triflates made cleanup less hazardous. Even with safer profiles, it helps to keep all waste streams under review, especially as labs worldwide adopt new materials at scale.
People talk about innovation as if it bursts from nowhere. In truth, building safer medicines, faster electronics, or cleaner energy so often runs on the invisible work of unassuming chemicals. Potassium trifluoromethanesulfonate plays the part of the silent helper: it doesn’t shout for credit, but it keeps the gears of science turning. With careful use, it could help unlock the next round of discoveries we all depend on.
Potassium Trifluoromethanesulfonate’s Unfancy but Vital Role
Not many people talk about potassium trifluoromethanesulfonate over coffee—or anywhere, really—unless they spend their days with beakers and chemistry textbooks. While it sounds like a mouthful, its workhorses qualities deserve a closer look. Chemically, this compound is known by the formula CF3SO3K. The molecular weight clocks in at about 222.19 grams per mole, anchoring its spot among useful potassium salts in modern labs.
More Than a Name: Why It Matters
Scientists and engineers run into this salt often because it brings both stability and utility without fanfare. Potassium trifluoromethanesulfonate doesn’t grab headlines, but its presence changes the outcome in battery research, organic synthesis, and even a few pharmaceutical reactions. A trifluoromethanesulfonate group, also called triflate, shows remarkable stability. That lets researchers use it as a non-coordinating anion. By nature, it won’t tangle itself up with more reactive bits of a molecule, so it creates a neutral playground for other chemistry to happen.
Many learn about potassium trifluoromethanesulfonate—often shorthanded as potassium triflate—in the context of “greener” or more reliable alternatives to older, messier reagents. Not too long ago, chemists leaned heavily on substances with longer preparation times and more dangerous byproducts. Looking for safer, less wasteful options turns up potassium triflate as a logical choice. With its straightforward makeup, it lets projects move forward, often with fewer headaches or regulatory red tape.
Real-World Use: The Quiet Backbone
In the world of organic synthesis, potassium triflate plays its most important card: boosting reactions like certain metal-catalyzed couplings. Researchers aiming to build carbon-carbon bonds for new medicines or materials trust it to avoid unwanted interference. For those who worry about technology needing to catch up on sustainability, potassium triflate points in the right direction. Its chemistry gives practitioners a robust tool that won’t easily break down into toxic leftovers, which keeps work benches—and water tables—cleaner.
Battery makers notice its reliability, too. The triflate anion provides better ionic conductivity in some electrolyte blends than more common anions. Anyone who’s dashed to recharge a dying phone benefits from steady innovation like this. Compounds that can carry ions without fouling up other materials drive everything from electric vehicles to solar power storage. Potassium triflate serves as one of several building blocks in a growing push toward battery chemistry that’s efficient, safe, and less reliant on rare elements.
Making Chemistry Practical and Responsible
My background includes plenty of time with technical manuals, but the real-world lesson holds true: chemistry doesn’t live in a vacuum. Choosing the right reagent—a solid option, not always flashy—often separates practical progress from theoretical footnotes. Potassium trifluoromethanesulfonate, with its formula CF3SO3K and an approachable molecular weight, stands as an example of progress that doesn’t need fireworks to make a difference.
Looking ahead, better awareness and easier access to reliable reagents like potassium triflate could push both academic and industrial chemistry toward safer, smarter workflows. Open discussions between scientists, environmental advocates, and manufacturers can demystify these compounds. Sharing accurate information—from chemical formulas to real application stories—opens doors to brighter, cleaner science that benefits everyone.
Practical Storage Isn’t Just a Checklist
Over the years, working in various labs, one thing has always stood out to me: the importance of respecting chemical storage beyond ticking boxes. Potassium trifluoromethanesulfonate pops up in a slew of organic syntheses, and if people overlook proper storage, trouble follows fast. This compound isn’t explosive or especially toxic, but its reactivity with moisture can ruin reactions and pose problems for lab safety.
Why Dry Spaces Matter
I remember a frustrated grad student who once left a sample out on a humid day, scrambling later to figure out why their catalyst would not kick things off. Potassium trifluoromethanesulfonate absorbs water from the air. That simple fact means a desk drawer won't cut it. Moisture contamination usually ends up spoiling both the material and the chemistry downstream. Many chemists learn this lesson the hard way—watching precious reagents slowly degrade into a sticky mess. Keeping this compound in an airtight container, tucked away with a desiccant, protects shelf life and safeguards results.
Cool, Consistent Temperatures Save Money and Worry
Lab budgets rarely leave room for wasted chemicals. Heat and wild temperature swings speed up decomposition, even for salts like potassium trifluoromethanesulfonate that seem pretty stable on paper. Storing it at room temperature keeps things simple and effective, provided the location stays away from direct sunlight and heat sources. Chemists often rely on dedicated cabinets for this reason. It’s the kind of routine that pays off over months—one less variable in uncertain experiments, one less order to place.
Labeling and Responsibility
It’s easy to overlook the basics in a shared lab space. Over time, unmarked jars multiply like rabbits. Misidentification leads to risk; anyone who has grabbed the wrong salt understands that. Clear labeling stands as an equal partner with careful storage. This isn’t just a formality, it’s about keeping coworkers and students out of harm’s way and protecting time and resources. I’ve witnessed more than one fire drill set off by a storage mix-up with other reactive chemicals.
Modern Habits for Safe Storage
Our best defense against accidents always comes down to proactive habits. In my experience, new researchers pick up good practices much faster by seeing them in action. A locked, labeled container stored with desiccant sends a clear message about discipline. Annual training refreshers help, but real change shows up in daily routines—checking seals, drying agents, and designating shelves for incompatible materials. Regulatory agencies set broad guidelines, but real safety culture flourishes within each team and lab bench.
Moving Forward as a Community
Science leans on collective responsibility. If each person treats potassium trifluoromethanesulfonate—and every other reagent—with care, everyone benefits. Lost product and ruined experiments take a toll no lab budget wants. Good storage practices save more than chemicals—they support reliable results, protect people, and build trust across teams. That mindset matters far more than any checklist ever could.
Chemistry Beyond the Lab: A Real-World Take
Walk into any university lab, and you’ll spot bottles with names that take a whole line to pronounce. Potassium trifluoromethanesulfonate is one of those compounds most folks wouldn’t hear about unless they work with organic synthesis or advanced electronics. With chemicals that sound this intense, the average person might assume danger is just around the corner. But facts matter, and so does our ability to weigh chemical hazards—or the lack of them—with open eyes.
What the Science Says
People trust chemical labels and safety data sheets for good reason. Years of study, and sometimes a few memorable mistakes, have taught us to never downplay a hazard. Potassium trifluoromethanesulfonate, or “potassium triflate,” gets its share of questions in academic and industrial spaces. While information floats around the web suggesting it’s less risky than its name might imply, folks who handle it still follow the same basic safety routines they’d use for any lab chemical. Gloves go on, goggles get tightened, and ventilation fans hum in the background.
Toxicity always draws attention because stories about chemical spills and mysterious illnesses linger in people’s memory. Looking at potassium triflate, current research and safety documentation hint at relatively low acute toxicity. No waves of poisoning cases, no headlines about accidental exposure. If spilled, the stuff doesn’t leap off the bench and attack. It dissolves in water, and labs usually clean it up without much fanfare. That’s not to say care should drop; even “low hazard” chemicals can irritate skin or mucous membranes if they end up where they shouldn’t be.
Personal Experience in the Lab
In the years I spent working with organofluorine reagents, potassium triflate became a quiet companion on synthesis days. I never heard about anyone getting sick from it, but veteran chemists reminded everyone to stay mindful, mostly because complacency invites avoidable mistakes. One time, a colleague accidentally brushed some powder onto a sleeve—routine washing sorted it out, but it reminded everyone to treat every bottle with respect.
The bigger issues crop up when disposal practices get sloppy. Potassium triflate contains fluorinated groups, and anything with fluorine in its structure tends to stick around in the environment. Persistent chemicals don’t break down quickly, so trashing large amounts without thinking could spell trouble over decades. Regulatory focus on “forever chemicals” like PFAS continues to grow for that reason. Safe disposal matters more than ever, especially in research hubs where these types of chemicals flow through weekly.
How to Make Things Safer
Better labeling and clear training always beat complacency. Students and workers need reminders that chemicals lacking a skull-and-crossbones symbol still deserve basic respect. On top of personal protection, it helps to have disposal bins nearby, plus reminders to review local regulations for fluorinated waste. Wastewater plants were never built to break down every exotic molecule chemists can produce, so locking down lab waste before it leaves the facility closes off potential headaches for communities downstream.
People worry about synthetic chemicals for good reason. Disasters from the past show what happens when industries cut corners or skip due diligence. While potassium trifluoromethanesulfonate does not jump out as highly toxic, that doesn’t mean it should get a free pass. Responsible use and steady education keep risks in check, protecting both workers and the broader environment in the process.
Why Chemists Rely on Potassium Triflate
As someone who has spent a lot of time following the push for new ways to build carbon–carbon and carbon–heteroatom bonds in the lab, I’ve noticed certain reagents keep coming up. Potassium trifluoromethanesulfonate—a mouthful, but most folks call it potassium triflate—has earned its spot in the regular toolkit for a reason. There’s no big mystery to it: chemists want reactions that work cleanly, scale easily, and cut down on unnecessary byproducts. Potassium triflate fits right in.
Bringing Out the Best in Cross-Coupling Reactions
The Suzuki coupling stands out as one of the biggest revolutions in organic synthesis, and I’ve watched it change how academic and industrial labs knock together complex molecules. Potassium triflate serves as a base in many transition metal–catalyzed processes like Suzuki, Heck, and Stille reactions. Why use it over something else? It keeps out water, remains stable under harsh conditions, and resists getting caught up in unwanted side reactions. Having a dry, reliable base makes the difference between chasing down mystery peaks in crude NMRs and getting crisp, clean products.
Living Up to Demanding Reaction Conditions
From a practical standpoint, potassium triflate dissolves in organic solvents such as acetonitrile, DMF, and DMSO. This opens the door for methods that just wouldn’t play nice with water or more aggressive alkali bases. When making aryl triflates from phenols, chemists sometimes turn to potassium triflate as a mild and compatible partner. Those aryl triflates, in turn, feed directly into cross-couplings or other transformations, expanding what chemists can do with simple building blocks.
Staying One Step Ahead in Fluorination Chemistry
The introduction of fluorine into pharmaceuticals takes careful maneuvering. Every atom added has the potential to change a drug’s metabolism, stability, and bioavailability. Potassium triflate often tags along for fluorination steps, playing the role of a supporting electrolyte or activating group. In my experience, tweaking yields on tricky steps sometimes comes down to the right ionic partner, and in fluorine chemistry, triflate salts stand out. They steer the reaction, keep things from gumming up, and help bring those expensive starting materials fully through to the product.
Making Complex Chemistry a Little Smoother
While it’s easy to focus on breakthrough molecules, daily work in the lab often circles back to reliability and efficiency. Potassium triflate rarely hogs the spotlight—it usually supports the process behind the scenes. Its stable nature cuts down accident risk in routine use. Its non-nucleophilic character means side reactions stay in check, which saves frustration and time at the purification bench.
Room to Innovate
Research marches forward. Green chemistry calls for rethinking reagents, including ones as useful as potassium triflate. While traditional processes have leaned on its strengths, more teams are testing alternatives to limit environmental impact. Cheaper, less toxic, or even reusable options could someday match the versatility of potassium triflate. For now, though, its purity, stability, and track record keep it in steady demand wherever smart synthesis counts.
Final Thoughts
The practical payoffs of potassium triflate in organic synthesis go beyond theory. After watching dozens of colleagues rely on it for high-stakes reactions, it’s clear why this reagent holds such loyalty. It makes tough transformations work, cuts waste, and lets researchers focus on what really matters—building up new science, not troubleshooting mistakes.