The Story Behind Potassium Vinyltrifluoroborate: Chemistry’s Unsung Building Block
Historical Development: Chasing Stable Reagents
Back in the mid-twentieth century, chemists intensified their search for stable boron-based reagents. Many contemporary reactions—like Suzuki-Miyaura cross-coupling—depended on air- and moisture-sensitive organoboron compounds. It wasn’t until the late 1990s and early 2000s that potassium organotrifluoroborates, including potassium vinyltrifluoroborate, truly caught the scientific imagination. These solid, air-stable salts bridged a frustrating gap for bench chemists everywhere, replacing volatile boronic acids prone to degradation with crystalline compounds that could sit unguarded on a shelf. Compared to their predecessors, they brought convenience and safety into molecular construction, especially for anyone tired of glove box gymnastics. Their introduction marked a shift toward more realistic workflows in both industrial and academic labs, reflecting the practical needs of day-to-day research.
Product Overview: A Workhorse for Today’s Synthetic Chemist
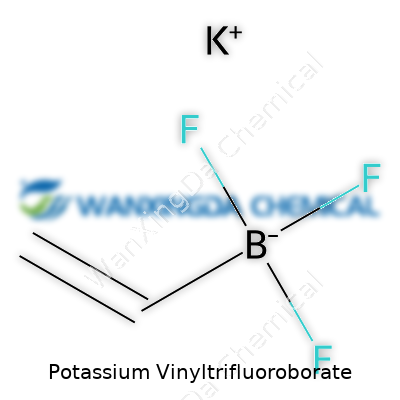
Potassium vinyltrifluoroborate doesn’t look impressive at first glance: just a white to off-white powder or crystalline solid. But its structure packs a punch. The molecule contains a vinyl group linked to a boron atom and three fluorines, all balanced by a potassium cation. Each part plays a crucial role. The vinyl group opens doors for addition and coupling reactions, the boron acts as a handle for Suzuki-type chemistry, and fluorines bring stability. Every major reagent distributor stocks it, a sign of how essential this compound has become in synthesis over the past two decades.
Physical and Chemical Properties: Stability Meets Function
No one can deny the practical appeal of potassium vinyltrifluoroborate. Solid at room temperature and soluble in polar solvents, it doesn’t fume, smell, or stain your workbench. The trifluoroborate component keeps the boron both stable and reactive; the potassium ion makes the compound readily handled, weighed, and measured. Unlike the unstable vinylboronic acids, it survives on bench tops for weeks or longer, even in humid environments. It resists hydrolysis, which saves time and headaches by reducing failed reactions due to decomposition. From an operational standpoint, this property streamlines workflow and helps guarantee reproducibility—critical for anyone working under deadline pressure.
Technical Specifications and Labeling: More Than a Name
Purchasing this reagent means grappling with almost comical specificity in chemical labeling. Chemists know it as potassium vinyltrifluoroborate, but one quick look through catalogs or publications will reveal synonyms such as potassium ethenyltrifluoroborate. Labels may cite purity, typical 97% or higher, and suppliers provide details on molecular weight and basic handling procedures, often suggesting storage in a cool, dry place. Documentation tends to mention lot analysis, residual water content, and elemental analysis. For industrial procurement, certificates of analysis help trace origin and batch-related variations, important for regulatory compliance and quality control. Every step matters, especially for scale-up in pharmaceutical or high-value materials manufacturing.
Preparation Method: From Raw Chemicals to Reliable Reagent
Most lab protocols call for the generation of potassium vinyltrifluoroborate from vinylboronic acids. Reacting the acid with potassium hydrogen fluoride or a similar fluoride source allows the exchange of hydroxyls for fluorine, forming a robust trifluoroborate salt. Purification follows, via crystallization or extraction with suitable solvents, to yield the final solid compound free from unreacted starting materials. Many groups have published variations, tweaking the fluoride source and solvent to boost yield or avoid hazardous byproducts. On a practical level, synthesizing these reagents doesn’t demand exotic equipment—good news for academic settings where budgets run tight and time even tighter.
Chemical Reactions and Modifications: The Synthetic Toolbox Expands
The main draw of potassium vinyltrifluoroborate lies in its compatibility with palladium-catalyzed cross-coupling reactions. Suzuki-Miyaura coupling leads the pack, letting chemists stitch together vinyl fragments with aromatic or heteroaromatic partners in high yields and minimal fuss. The stability of the trifluoroborate group allows longer reaction times and elevated temperatures, making it friendly to demanding conditions. Some groups have used these salts in Petasis reactions or as vinylating agents for the construction of complex pharmaceuticals. The vinyl group itself undergoes further modifications—hydrogenation, epoxidation, and cycloaddition open additional synthetic avenues. This flexibility explains the growing stack of research papers featuring this unassuming white powder.
Synonyms and Product Names: Nomenclature Chaos
Depending on where one shops or which publication lands on your desk, potassium vinyltrifluoroborate pops up under a variety of aliases. Chemists see names such as potassium ethenyltrifluoroborate, vinyltrifluoroborate, or even shortened codes in patent filings. Each supplier pushes their own naming standard, and students still new to the bench might double-check the chemical structure to avoid confusion. These overlapping names reflect the quirky, sometimes fragmented evolution of chemical sourcing and academic publishing, which rarely privileges clarity over tradition.
Safety and Operational Standards: Risks and Best Practices
Solid potassium vinyltrifluoroborate doesn’t bring the fire hazards or acute toxicity associated with more aggressive reagents, but its handling still requires respect. Dust can irritate the eyes or respiratory tract, especially in confined spaces or during weighing. Long-term toxicity remains low, judged from routine use and researcher experience, but exposure to chemical powders always invites caution. Wearing gloves, goggles, and sometimes working inside a fume hood keeps direct contact to a minimum. Modern safety data sheets urge against ingestion or inhalation, though adverse events seem rare compared to more dangerous analogues. Waste disposal presents no exceptional challenge, with standard protocols sufficing. Such practical safety profiles encourage broader adoption, even in undergraduate settings, where accidents from highly reactive chemicals once discouraged hands-on exploration.
Application Area: Reaching Across Research Fields
Pharmaceutical chemists, materials scientists, agrochemical developers, and academic researchers find reasons to keep potassium vinyltrifluoroborate close at hand. The heart of its appeal is adaptability; by enabling reliable cross-coupling of vinyl fragments, it plays supporting actor in drug discovery, polymer development, and complex natural product synthesis. Some groups use it to attach distinctive labels or functional groups onto organic frameworks for imaging or diagnostics. The widespread interest crosses disciplines; organic synthesis textbooks increasingly feature its preparation and coupling strategies. This accessibility means even small labs or startups, with limited funds or personnel, can incorporate advanced cross-coupling steps into their pipelines without major overhead or capital.
Research and Development: Progress on Many Fronts
Ongoing work tests the boundaries of potassium vinyltrifluoroborate, expanding its applications and fine-tuning its reactivity. Catalysis experts look for faster, greener cross-coupling protocols by adjusting ligands and reaction conditions. Patent literature hints at uses beyond its flagship role: some research teams have developed applications in medicinal chemistry where the direct installation of vinyl groups speeds up candidate evaluation. Others explore automated and continuous-flow processes, which save time and improve scalability for industrial supply chains. The steady publication rate shows lasting interest, suggesting this is far from a niche reagent. For those dreaming of new methodologies or faster routes to known targets, the steady evolution of potassium vinyltrifluoroborate chemistry promises innovation with each passing year.
Toxicity Research: Weighing Practical Risks
Compared to old-school boron chemistry, the trifluoroborate salts generally show low acute and chronic toxicity. Scientific assessments suggest potassium vinyltrifluoroborate doesn’t pose major risks by inhalation, skin contact, or environmental release—though as with any chemical, care matters. Analytical labs check residue levels and environmental persistence, but thus far, evidence points to ready breakdown under typical waste treatment. Most reports focus on worker safety, with emphasis on minimizing dust generation and preventing accidental ingestion. The general consensus sees this compound as a welcome shift toward safer, more user-friendly boron reagents, especially in hands-on teaching environments or high-volume pharmaceutical labs, where risk accumulates through repeated exposure. Regulation lags behind technology at times, but practical experience continues to show these salts are less potent threats than many widely used solvents or catalysts.
Future Prospects: Wide Open Frontiers
Everything suggests the story of potassium vinyltrifluoroborate has more chapters to write. Cross-coupling chemistry shows no signs of slowing, and demand for reliable, stable, and practical reagents only grows as new research fields emerge. Green chemistry calls for higher yields, lower waste, and minimal environmental footprints, pressing both manufacturers and synthetic chemists to keep refining protocols for even greater efficiency. Advances in catalysis—think ligand design or bio-inspired methodologies—could unlock reactions so far impossible, making this humble salt the node for new transformations. My own experience tells me this: once a synthetic tool wins broad trust for its safety and performance, labs everywhere hang onto it and push boundaries even further. If science rewards steady reliability and versatility, potassium vinyltrifluoroborate seems poised to remain an essential partner for tomorrow’s chemists, just as it has for the past twenty years.
Everyday Life Depends on Quiet Revolutions
Potassium vinyltrifluoroborate doesn’t show up in daily conversation, but anyone with an interest in medicine, agriculture, or materials has felt its impact. I started noticing how much chemistry shapes our world in college labs, where handfuls of powder could turn into medication or new plastics. Even the smallest changes in molecules matter. Substances like potassium vinyltrifluoroborate make those shifts possible, especially when building new molecules gets tricky with old-fashioned ways.
Bridging Chemical Barriers
This compound stands out for one clear reason: it smooths the path for making strong carbon–carbon bonds in complicated molecules. Many advanced medicines and electronics depend on just that—connecting carbon atoms without the hassle and risk of traditional alkylating agents. Potassium vinyltrifluoroborate gives chemists a tool that stays stable on a shelf, handles moisture without breaking down, and delivers results with less fuss. The Suzuki-Miyaura coupling—a reaction that helped earn a Nobel Prize—owes much of its recent progress to easy-to-handle boron chemicals like this one. Suddenly, research labs and factories can plan longer, tougher syntheses with fewer detours or surprises.
Why Innovation Relies on Smarter Building Blocks
Pharmaceutical makers have been chasing new cancer drugs, antivirals, and antibiotics year after year. Many of today’s most active molecules, from targeted therapies to next-gen pesticides, need tough chemical bonds in places where older methods risk failure or create toxic byproducts. Potassium vinyltrifluoroborate lets scientists swap the messy reagents for a cleaner, safer process. There’s less need for dangerous materials and more chance that a promising compound survives the scale-up stage, all thanks to better ways of stitching molecules together.
Electronics are another place where this chemistry has an edge. Rings, chains, and frameworks with tricky carbon bonds often turn up in new display materials, semiconductors, and energy storage devices. By allowing precise connection of these units, potassium vinyltrifluoroborate helps push research past old boundaries and meets both higher performance and stricter safety rules.
Challenges and Smarter Solutions
Some hurdles remain. Not every plant handles these chemicals with the care they deserve, especially as the call for sustainable practices grows louder. Even the cleanest lab process can create waste. One path forward lies in better recycling and greener solvents, where industry and universities investigate ways to cut down on byproducts. Another angle involves education: with more chemists trained to use these new building blocks, the whole field becomes more nimble. That includes data sharing, collaborations, and bringing in ideas from green chemistry to make sure progress doesn’t mean more pollution.
Learning to handle and apply potassium vinyltrifluoroborate safely and creatively opens doors for more practical medicines, cleaner manufacturing, and less hazardous jobs for the people behind the scenes. Sustained investment in both science and training will keep these benefits reaching beyond the lab, into the hands and homes of everyone relying on progress for a healthier world.
Getting to the Roots: The Chemical Formula
Potassium vinyltrifluoroborate pops up in chemistry circles thanks to its handy formula: C2H2BF3K. Every piece of this formula tells a story. You have two carbon atoms from the vinyl group, a couple of hydrogen atoms hitching along, one boron, three fluorines, and a potassium ion. This mix pulls chemistry away from dry textbooks and puts it in the hands of people working on new drugs, advanced materials, and experiments that push boundaries.
The Real-World Tale of a Chemical Oddball
Lots of folks outside chemistry might glance at C2H2BF3K and shrug, but this little molecule makes sparks where it counts. For people in medicinal chemistry, it’s a staple for piecing new molecules together. One of the reasons: it’s stable, unlike some older boron reagents that break down too quickly or blow up your experiment. I remember seeing colleagues get frustrated with other reagents that fizzled out too fast. Once potassium vinyltrifluoroborate landed on the scene, their lab work took fewer wrong turns. They could trust this salt wouldn't ruin a week's worth of effort with a pop or fizz. Stability, in the end, saves precious time and money.
Supporting Real Progress
The chemical world doesn’t stay still. Researchers love tools that let them build molecules that never existed before. Doctors want new medicines that attack disease in clever ways. Innovation needs reliable chemistry. Potassium vinyltrifluoroborate isn’t a celebrity, but it’s a workhorse. Its formula gives you trifluoroborate, which teams up beautifully with other chemical partners. The Suzuki-Miyaura reaction, for example, depends on this teamwork. This reaction has changed how scientists create complex organic compounds, especially in cancer research and new electronics.
Environment and Safety: Not Skipping the Tough Stuff
People talk big about green chemistry, and for good reason. Our planet doesn’t have endless patience for dumps and spills. The old way to get certain boron compounds involved solvents and conditions that left scars. Potassium vinyltrifluoroborate acts differently. It usually behaves kindly in water and remains solid under most storage conditions. If you handle it right and don’t dump it down the drain, the risk drops while the usefulness stays high. It won’t fix every environmental problem, but it gives researchers safer choices compared to some of the alternatives. Everyone wins when fewer nasty byproducts show up in the waste barrel.
Facing Challenges and Looking Forward
The chemical community runs into snags, just like any other group. Sourcing pure potassium vinyltrifluoroborate can get tricky. Demand spikes, supplies thin out, and suddenly projects stall. While alternative methods exist to make the compound, consistency across batches isn’t always perfect. It makes sense to invest in better synthesis methods and smart storage to keep quality steady. Some scientists eye new training programs for handling rarer chemicals, aiming to keep mishaps off the evening news.
Building Blocks, Big Changes
No one compound changes everything, but potassium vinyltrifluoroborate proves how small changes in a formula matter. It offers steady performance, reduces some old risks, and helps creative minds chase new cures and materials. Chemistry matters in everyday life, even if we don’t see these molecules at the grocery store. Every bit of progress depends on foundations like C2H2BF3K—the kind of chemical that keeps the wheels turning behind the scenes.
Looking Out for Safety in the Lab
Working hands-on with chemicals teaches you that storage is as important as knowing the properties of each compound. Potassium vinyltrifluoroborate can look harmless sitting on a shelf, but anyone who has handled tightly regulated materials knows the story can change quickly with a bit of carelessness. Experience in academia and industry shows small mistakes—like an unlabeled lid or a damp spot in the wrong corner—raise risks not just for the experiment but for everyone nearby.
The Reality of Dealing with Potassium Vinyltrifluoroborate
Let’s talk about why things can go sideways with this one. Vinyltrifluoroborate salts aren’t explosive under most conditions, but they have quirks that create headaches if handled poorly. Moisture in the air eventually eats away at the purity of many boron compounds, including potassium vinyltrifluoroborate. Anyone in chemical research has seen powders clump up or turn sticky from being left open too long. Not a dramatic event, but it ruins the sample. Worse, spoiled material ends up in waste bins, hammering tight research budgets and turning inventory pressing for a replacement.
Simple Storage Steps Backed by Hard-Won Experience
Dry, airtight containers protect more than just compounds—they guarantee safety and reliability. Glass bottles with solid seals or thick plastic containers do the job. Folks who’ve opened spent jars know the stale, chemical smell that escapes. That odor signals contamination or unwanted reactions. If a lab stays even a bit humid, silica gels tucked inside storage cabinets add layers of protection by soaking up stray moisture before it hits the chemical. I learned this trick from a mentor who managed sensitive materials for decades. He always said a leaky container was like playing dice with lab safety and your results.
Cool storage slows down reactions in almost every chemical, including this one. Some chemicals need refrigerators, but keeping potassium vinyltrifluoroborate away from heat sources—no matter how stable it might seem—protects its quality far longer. Some labs invest in dedicated cabinets for air-and-moisture-sensitive reagents. Others just keep materials away from hot windows or radiators. Either way, keeping things cool is a lesson learned from busted samples and unnecessary orders.
Labeling and Separation: Small Habits That Save Big Troubles
Labels matter more than anyone wants to admit, especially when shelves fill up. Black ink on a yellow sticker stands out, even in low light. Write down dates alongside compound names—you’ll spot old material before it causes problems. Don’t keep potassium vinyltrifluoroborate with acids, oxidizers, or anything reactive. Mixing chemicals by accident tends to happen late on busy days or in crowded spaces. Separation by chemical class takes away one more risk, making for a cleaner, safer workflow.
Learning from Mistakes and Sharing Knowledge
There’s no substitute for first-hand stories. Friends in research tell how a single missed label delayed weeks of work or how overlooked moisture ruined a batch just hours before a presentation. Many costly errors in labs happen from ignoring simple steps like those above. If new team members see experienced technicians taking storage seriously, safe habits start to feel like the natural way to work.
Solutions for Scaling Up
It gets trickier outside the academic world. Anyone scaling up research into production faces real temptations to cut corners. Good process means sticking with proper, dry, sealed storage and making it routine. Regular checks, team training sessions, and setting a culture where people look out for one another matter most. Investing in proper cabinets and tracking tools saves more money and time than many assume at first.
In the end, storing potassium vinyltrifluoroborate right forms the backbone of safety and reliable work. Every safe return home at the end of the day starts with the choice to do things properly—one jar, one label, one closed lid at a time.
The Understated Presence of Potassium Vinyltrifluoroborate in Chemistry
Potassium vinyltrifluoroborate finds its way into modern labs thanks to the rise of organoboron chemistry. It has this knack for helping build carbon-carbon bonds in organic synthesis. As someone who has handled a fair share of unusual reagents, I’ve watched graduate students and postdocs approach new bottles with the usual caution—gloves, goggles, and plenty of questions about what could go wrong if something spills or splashes.
What Keeps Safety Officers Up at Night?
No one wants to downplay laboratory risks. So whenever a new or lesser-known compound comes up, the next step tends to be diving into the Material Safety Data Sheets and chemical supplier pages. Potassium vinyltrifluoroborate isn’t a household name, and its safety information can feel obscure if all you've got is the chemical formula. What matters most is how it actually behaves, especially compared to compounds known for violent reactions or acute toxicity.
Practical experience and reports show this compound doesn’t rush to catch fire, explode, or release toxic fumes at room temperature. Compared to classic bad actors from the chemistry bench—think sodium azide, hydrazine, or phosgene—it sits much lower on the immediate danger scale. According to published literature, it doesn’t show acute toxicity in small exposures, and it isn’t volatile. That means you’re not dealing with invisible clouds of poison if you open the bottle. You won’t get nerve damage from a single whiff, and contact with the solid doesn’t usually burn through gloves on the spot.
Respect Doesn’t Equal Fear
Even so, underestimating any chemical is a fool’s game. Potassium vinyltrifluoroborate contains boron and fluorine atoms, which always nudges chemists to remember old stories about fluoride’s dangers. Swallowing or inhaling any salt in large doses never benefits one’s health. I would never taste it or allow it to become airborne. Dust can irritate the lungs if breathed in, and long-term impacts from regular exposure haven’t been mapped out. Without years of toxicology data, caution stays on my checklist. There’s no prize for bravado around reactive powder.
Building a Culture of Safety
What’s missing from many chemistry discussions is the focus on mindful lab culture. Early in my career, I thought following the rules and ticking off glove usage was enough. As I saw more, I learned it takes more than paperwork and routine: lab safety depends on open discussion, experienced oversight, and everyone keeping an eye out for hazards that might get forgotten. That low toxicity label sometimes causes a sense of complacency, which does more harm than most realize.
For any organoboron reagent—not just potassium vinyltrifluoroborate—handling should always involve well-ventilated hoods, proper personal protective equipment, and clear protocols for spills. Headaches, skin irritations, or undiagnosed rashes may develop after long-term repeated exposure, even when the compound itself hasn’t earned a strong toxicity ranking. I tell new students not to rely solely on the absence of horror stories. Every new reagent brings its own quirks, and safety updating is never over. Staying humble about small gaps in our knowledge, even for widely used research tools, does more to protect science—and scientists—than memorizing a list of ingredients to avoid.
Organic synthesis has always thrived on clever reagents, and Potassium Vinyltrifluoroborate marks one tool chemists keep reaching for when building complex molecules. In the past decade, new reactions keep popping up in the literature, but some reagents hold steady because they meet the needs of both industrial and academic labs—this one fits that bill. Years ago, during my own time in the lab, handling air-sensitive boronic acids felt like babysitting. Many share that memory. The day I first weighed out some Potassium Vinyltrifluoroborate felt liberating. No special glassware, no worries about it falling apart on the bench, just scoop and add. For most folks, that reliability is not hype but a real necessity for modern synthesis workflows.
The Suzuki–Miyaura Reaction: Fueling Cross-Coupling Advancements
One reason Potassium Vinyltrifluoroborate gets attention centers on its performance in the Suzuki–Miyaura reaction. This cross-coupling strategy lets chemists link up pieces of molecules to make new carbon–carbon bonds, a backbone for pharmaceuticals, agrochemicals, and materials. Potassium Vinyltrifluoroborate brings a vinyl group—the very thing needed to add unsaturation—cleanly into the mix. Compared to more sensitive couplings with vinyl halides or boronic acids, this salt takes the heat and moisture in stride. Yields remain strong even with tricky partners. This makes life easier for those who want to avoid wasted runs or re-running reactions to chase down elusive products.
Building Blocks Beyond Pharmaceuticals
The vinyl group Potassium Vinyltrifluoroborate introduces opens doors beyond medicines. Polymers gain new functions. Performance materials benefit from enhanced chemical stability. Agrochemical researchers push for more targeted crop treatments. Many chemists I’ve known have found it welcome for pushing vinyl groups into aromatic rings or heterocycles, creating cores for dyes, electronic materials, or additives with improved life span and performance. In each of these cases, the ease of storing and handling this crystalline solid clears a big hurdle. Scalability remains a sticking point for much synthetic work, and stable intermediates like this remove one stress factor from process planning.
Potential Drawbacks and Current Challenges
Not every reaction favors Potassium Vinyltrifluoroborate. Some catalyst systems don’t tolerate the trifluoroborate group, demanding tweaks in ligand or solvent choice. In complex molecules, the vinyl unit can run into competing reactivity. Industrial synthesis often dictates high throughput and tight budgets, which places pressure on chemists to refine these protocols. In my own work, a reaction sometimes needed more careful control of water or base, but the reliability of the trifluoroborate salt often outweighed that minor hassle.
Paths Forward: Greener and More Efficient Synthesis
The drive for sustainability means even well-loved reagents face scrutiny. Waste minimization, solvent choice, and greener reaction conditions all matter. Researchers look to ways of reducing metal catalyst usage or moving to milder reaction protocols. Some teams have uncovered methods using less expensive metals or water-based systems. Student-led projects tackling these challenges keep popping up at conferences. The skills learned on these transformations—scaling reactions, controlling selectivity, identifying bottlenecks—translate directly from academia to industry. For anyone in the chemical community, Potassium Vinyltrifluoroborate acts as a practical bridge between bench discoveries and real-world applications, a reminder that simplicity and reliability open the door for creative synthesis.