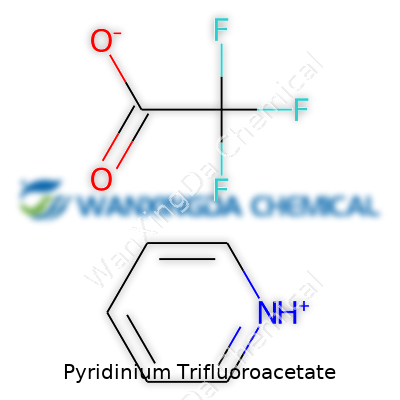
Pyridinium Trifluoroacetate: More Than A Chemical Reagent
Historical Development
Pyridinium trifluoroacetate came about during a time when chemists were looking for new ways to manipulate molecules that weren’t easy to access with traditional acids and bases. Organic synthesis really started to take off in the mid-20th century, and curiosity about tuning reactivity gave chemists a reason to tinker with unconventional salts. Somewhere along the line, the combination of pyridine and trifluoroacetic acid landed on lab benches, not because anyone was guessing, but because pyridine was already well known as a basic scaffold, and trifluoroacetate brought serious electron-withdrawing power to the table. The pairing offered a nice balance of acidity without the stickiness or unpredictability seen with some other reagents. Over the decades, its reputation settled in labs as a go-to partner for mediating reactions that could otherwise spiral out of control under harsher acid conditions.
Product Overview
Walking down the aisles of any chemical storeroom, you’ll find pyridinium trifluoroacetate sitting alongside other organic salts. It’s rarely in the spotlight compared to its more common relatives, but those who know its quirks and tricks rarely swap it out. Chemists value it for reliable, consistent performance in a variety of settings: catalyzing esterifications, tweaking alkylations, or nudging selectivity in peptide synthesis. When someone talks about using it, there’s a sense of trust—almost like a seasoned cook grabbing a favorite spice that never disappoints. Yet, it doesn’t scream for attention or redefine entire fields; it’s part of the backbone of routine organic manipulation, loyal to tasks demanding mild acid strength and minimal byproduct formation.
Physical & Chemical Properties
You can spot pyridinium trifluoroacetate as a colorless to slightly yellow crystalline solid, sometimes showing up as a powder. It absorbs moisture, so you’ll see it sealed tight in bottles, guarding its purity. It dissolves well in many common lab solvents—think acetonitrile or methanol—giving it flexibility for all sorts of synthetic schemes. As far as stability, it holds up well under normal storage, not decomposing unless really pushed thermally or with strong bases. The presence of trifluoroacetate anion doesn’t just alter its acidity; it tamps down nucleophilicity and boosts oxidative stability compared to straight mineral acid salts. That matters in multi-step syntheses where you can’t afford reactive intermediates tearing everything apart.
Technical Specifications & Labeling
Bottles of pyridinium trifluoroacetate wear precise labels with molecular formula and weight, along with purity ratings that tell you what kind of process you can trust them in. For HPLC-grade syntheses or pharmaceutical routes, you want the highest purity, and reputable suppliers run the extra mile to confirm absence of trace metals and organic contaminants. You won’t see garish warnings, but fine print carries enough information about handling, shelf life, and incompatibilities—nobody wants an accidental reaction with a strong oxidizer or a careless spill near water-sensitive chemistry. Lab labels also increasingly highlight environmental impact and recommended disposal paths, since sustainable practices aren’t just buzzwords—they’re policy in modern research.
Preparation Method
Making pyridinium trifluoroacetate boils down to straightforward acid-base chemistry, but it manages to feel like a small magic trick the first time you see it. A measured dash of trifluoroacetic acid meets an equal molar amount of pyridine—sometimes at room temperature, sometimes gently warmed if solvents are reluctant. The moment they shake hands, a salt forms, precipitating from solution or crystallizing as solvents boil away. Large manufacturers often purify the result by recrystallization to strip away stray acid, unreacted base, or water. There’s no need for fancy catalysts or elaborate purification rigs, and as a bonus, yields are consistently high. Chemists trust this method because it’s predictable, and they don’t have to watch over it like a hawk.
Chemical Reactions & Modifications
Pyridinium trifluoroacetate finds its footing as a mild acid in synthetic chemistry. People reach for it to drive reactions that would go off the rails with mineral acids or where you need to avoid water that comes with hydrochloric or sulfuric acid solutions. It shines during deprotection steps and in certain coupling reactions, especially in the construction of peptides or modified nucleosides, where acid sensitivity can kill a synthesis. Its role as a proton source comes through with minimal side reactions, partly because the trifluoroacetate is weakly coordinating, and you skip a lot of unwanted rearrangement or polymerization fate. For those who tune their processes, the pyridinium cation itself can sometimes act as a nucleophilic partner, opening doors to clever alkylation or acylation tricks. Studies in the literature document its utility for activating oximes and other intermediates.
Synonyms & Product Names
Pyridinium trifluoroacetate wears different names from vendor to vendor. Some call it 1-H-pyridinium, 1-carboxy-1,1,1-trifluoromethane; others stick with acronyms or abbreviations like PyTFA. It pops up in catalogues under alternate salt names, but experienced chemists quickly scan for the telltale pyridine and trifluoroacetate pairing and disregard the branding. This multitude of names occasionally causes mix-ups in communications between labs or regulatory filings, so clear chemical naming and structure diagrams are a must on documentation—no room for vague shorthand when purity and safety are on the line.
Safety & Operational Standards
Nobody walks into a lab and scoops up a handful of pyridinium trifluoroacetate without thinking about gloves and splash goggles. Even though you don’t see it corroding surfaces instantly like hydrochloric acid, it remains an irritant and has its own risks. Inhalation can make throats sore and skin contact leaves redness, so ventilation hoods and good ventilation always go hand in hand with its use. Laboratories set protocols for safe storage, keeping it away from strong alkalis and reducing agents, since prevention is better than cleanup. Regulatory agencies require clear hazard communication, and lab managers drill safe transfer and spill response again and again. Environmentally, disposal regulations urge water dilution and proper waste categorization so the fluorinated moiety doesn’t end up contributing to persistent organic pollutants in waterways—a rising concern as green chemistry gets teeth.
Application Area
Walk into any synthetic organic chemistry lab that’s pursuing peptide drugs, natural product modification, or custom materials, and you will find a spot for pyridinium trifluoroacetate. It doesn’t dominate headlines the way big-name catalysts do, but it’s involved in transition-metal-free couplings and gentle deprotection protocols that leave sensitive molecules intact. Pharmaceutical chemists favor its mildness in the late stages of synthesis, especially when expensive intermediates are on the line and nobody wants a project ruined by a runaway reaction. Biochemists sometimes use it to tweak protein surface charge or signal peptide cleavage. Materials scientists rely on it to mediate functional group installation in surface engineering, where harsh acids would wreck delicate nanostructures. Its versatility fits right into the chemist’s toolbox—not a star performer, but consistently reliable across a range of demanding scenarios.
Research & Development
Academic and industrial research keeps pushing pyridinium trifluoroacetate into new spots. Groups searching for greener reaction conditions experiment with it as a substitute for traditional acid catalysts. Others use it to probe reaction pathways, since it introduces less noise in kinetic studies than heterogeneous or strongly acidic media. Analytical chemists started to study its role in mass spectrometry sample prep, leveraging its volatility and lack of background reactivity. Patent applications often list pyridinium trifluoroacetate among permissive acids for facilitating complex molecular assembly, sometimes as a backup if stronger acids create impurities. As green chemistry ideals take hold, researchers also look at how its fluorinated anion might affect environmental fate, nudging future designs toward less persistent analogs.
Toxicity Research
Scientists take toxicity seriously, even for chemicals that seem mild. Studies on pyridinium trifluoroacetate track how the body deals with exposure—acute and chronic—by inhalation, ingestion, and skin contact. Tests show mild to moderate local irritation, but concerns linger about the trifluoroacetate component, as bioaccumulation and slow degradation can raise flags. Most acute toxicity reports center around animal models and cell cultures, and strict exposure guidelines govern how much makes it into waste streams. Occupational health reviews remind chemists not to take gloves and masks for granted, even with substances lacking dramatic warning labels. Environmental groups push for deeper studies on what happens when it’s released beyond the lab, especially as fluorinated organics come under scrutiny. Researchers respond by advocating for collection, containment, and disposal that keeps it from leaving the closed loop of responsible chemical management.
Future Prospects
Pyridinium trifluoroacetate’s future looks tied to how society balances chemical utility against environmental stewardship. As demand grows for selective, controllable chemistry, the call for mild and predictable reagents stays strong. Chemists want to keep using compounds like this, but only if they can confidently show that every bit stays accounted for, not leaking out into broader ecosystems. Calls for biodegradable or easily remediated functional analogs keep rising, inviting researchers to tinker with the pyridinium framework and search out alternative counter-ions that don’t stick around in nature. The willingness of the chemical industry and academic labs to invest in life-cycle assessments, recycling programs, and safer process development will shape which direction this salt heads over the next decades. What stands out to anyone who’s put time in the lab is just how much trust and discipline flow into every flask or vial; people use pyridinium trifluoroacetate not because it’s the only tool, but because it gets the job done with a minimum of drama—provided everyone keeps their eyes open for what happens after the reaction ends.
A Little-Known Player in the Lab
Working with complex organic molecules takes patience, skill, and, sometimes, a little help from chemistry's less-glamorous contributors. Pyridinium trifluoroacetate (PTFA) doesn’t get the spotlight, but its role matters if you care about clean reactions and scientific progress. This salt lands on reagent shelves because it solves specific problems in synthetic chemistry without fuss or drama. It helps move reactions forward where simpler choices would stall or muddy the outcome.
The Real Use: Deprotection and Acylation Reactions
Anyone who has wrestled with protecting groups in organic synthesis knows that taking them off cleanly is where things often go sideways. PTFA shines in this niche. Chemists use it for deprotecting tert-butoxycarbonyl (Boc) groups from amines—crucial for building complex drug molecules or assembling precise building blocks. Boc-protected amines don’t give up their shields easily. PTFA steps in and strips the group away, usually avoiding the side reactions that harsher acids can cause.
PTFA also helps in acylation and esterification reactions. It nudges reaction partners into the right state, so they bond efficiently and predictably. Because of its balance—strong enough to do the job, gentle enough not to trash the rest of the molecule—it’s gained a steady spot in synthetic labs. From my own time struggling to get pure peptides, using PTFA instead of trifluoroacetic acid spared me the joy of extra purification steps and unreliable yields.
Why Not Just Use Stronger Acids?
In theory, stronger acids like trifluoroacetic acid should wipe out protecting groups every time. The problem is, they might also eat away at other parts of the molecule. That’s a recipe for disappointment in both research and industry labs. PTFA avoids these pitfalls by combining pyridinium (a weak base) with trifluoroacetate (the strong acid part), creating a reagent with enough bite but a much lower chance of unexpected side reactions. This makes a difference when chemists try to build molecules with multiple delicate features—one mistake can mess up a week’s work.
Impact on Research and Industry
Pharmaceutical companies and academic groups depend on reliable reagents because a small gain in efficiency can save real money and time across big projects. PTFA fills this role without the regulatory headaches or safety hazards that come with some corrosive acids. It works at room temperature, in common solvents, and doesn’t require handling under anhydrous or inert conditions. That accessibility levels the playing field, especially for startups or university labs running on lean budgets.
Challenges and Directions Forward
Despite the benefits, PTFA isn’t perfect. Waste disposal and environmental impact of trifluoroacetate-containing chemicals add pressure to find greener options. Some teams are experimenting with alternatives that use milder acids or recyclable reagents. Still, until those become widespread, PTFA continues to bridge the gap between harsh, old-school acids and the gentle touch modern chemists want.
Bottom Line
Pyridinium trifluoroacetate doesn’t make headlines, but the work it supports influences new treatments and scientific understanding. Focusing on reliability and selectivity, it lets chemists work smarter and cleaner, which, in my view, makes all the difference in the kind of science that ultimately shapes the world outside the lab.
Knowing What’s Inside
Sometimes chemistry can feel like a secret language. It’s a lot clearer once you break down a formula and realize every symbol tells a story about the way atoms connect and chemicals behave. Pyridinium trifluoroacetate may sound like something you'd stumble across in a research journal, but it’s simpler than it looks at first glance. If you look at the name, you can spot its structure: there’s pyridinium, the protonated form of pyridine, teamed up with trifluoroacetate. Put together, the chemical formula is C5H6N+ CF3COO−. Sometimes it’s also written as (C5H6N)(CF3COO) to show both ions.
The Importance of Getting the Formula Right
There’s a lot riding on precision in chemistry labs. It’s not just about filling space in a notebook. Many reactions—especially those used in drug synthesis or material science—rely on predictable results. If the formula for pyridinium trifluoroacetate gets confused with something else, downstream impacts could range from wasted material to failed experiments. From my own days in an undergraduate organic chemistry lab, I remember a student mixing up similar salts—ruining a full afternoon of work for an entire bench. That memory stuck with me. In chemistry, shortcuts on accuracy rarely pay off.
What Makes This Compound Useful?
Pyridinium trifluoroacetate enjoys popularity with synthetic chemists for a reason. The pyridinium ion acts as a mild acid, while the trifluoroacetate anion stabilizes the salt and brings a strong electron-withdrawing punch. In practical terms, this compound gets used to help certain reactions move along, especially transformations that hinge on subtle acid-base tweaks. The robust nature of the trifluoroacetate anion allows it to survive in fairly tough conditions, broadening the toolset for folks who want results that don’t fall apart on them.
Transparency and Fact-Based Knowledge
There’s a constant push for trustworthy information in science. Following evidence, citing chemical databases, and respecting published research means no one has to play guessing games with formulas. PubChem and other respected resources confirm the formula for pyridinium trifluoroacetate as C5H6N+ CF3COO−. Sticking with established facts builds confidence for everyone, whether you're a graduate student or a seasoned R&D professional.
Looking for Solutions to Common Traps
Mistakes in formula notation pop up as often in textbooks as in hurried emails. One way to cut down on these errors: encourage double-checking. Nothing reveals a mistake faster than explaining your thinking to a colleague. Software tools like ChemDraw help, but they don’t replace an attentive eye for detail. In teaching settings, emphasizing how each part of a chemical name guides you toward its formula builds long-lasting habits. Taking a minute to check chemical databases before mixing solutions has saved many projects—and kept people safe.
Why Familiarity Matters
The world of chemistry rewards those who don’t just learn formulas but understand their logic. Recognizing names like pyridinium trifluoroacetate and confidently translating them into a chemical formula keeps science moving in the right direction. After all, small details in a formula can have big consequences in the real world, both in the lab and beyond.
Real Lab Lessons
Anyone who’s spent more than a few sweaty hours working in chemistry knows the headache that comes with storing sensitive reagents. Pyridinium trifluoroacetate isn’t a household name, but it comes up often in synthetic labs thanks to its role in organic transformations, particularly peptide coupling and other acid-driven reactions. Getting casual with storage can turn a working system into a hassle in a hurry.
Why Conditions Matter
Chemical stability forms the backbone of reliable experimental results. Pyridinium trifluoroacetate holds up well under the right circumstances, but its composition — pyridinium cation paired with trifluoroacetate — calls for attention. Exposure to moisture can prompt hydrolysis or introduce impurities; those unseen changes ruin purity and performance. Contamination by water, solvents, or even simple atmospheric gases isn’t rare in busy labs, especially where discipline slips or containers get reused without much thought.
The Role of Container Choice
Experience taught me the hard way: skimp on the container, lose quality and confidence. Pyrex glass or tightly sealed HDPE bottles work well, especially those with reliable screw caps lined with PTFE. Materials should resist strong acids and keep outside air from creeping in. Glass gives a visual check on clarity — cloudiness or crust along the lid means something unwelcome happened. Rely on fresh containers; a squeeze to save on waste often costs more in ruined stock or wasted time chasing down failed reactions.
Keeping Moisture and Light Out
Humidity brings swift trouble here. Even in air-conditioned labs, spores of water can slide past old seals and unmonitored caps. Keep desiccant packets — fresh ones, not those picked from old shoe boxes — in storage containers. These little bags pull moisture from the air and protect the compound. Limited light exposure supports longevity, too, as photochemical reactions in some salts creep up over months. Cabinets without direct sunlight or transparent doors do the trick. Refrigeration isn’t essential for every case, but cooler storerooms in the 2-8°C range slow down unwanted changes for long-term storage.
Labelling and Segregation
Overlooked details, like honest labeling, separate orderly spaces from chaotic ones. Every bottle wants clear dates, material weights, and initials. Keep incompatible materials apart. Traces of amines or strong bases nearby can lead to slow, barely visible contamination that undercuts batch-to-batch reliability. A small shelf dedicated to acid salts — kept distant from bases and oxidizers — creates a safer workspace and sharper results.
Solutions for Common Slip-Ups
In most cases where a batch failed, the culprit could be traced to improper storage — leaky lids, forgotten desiccant, or just too much handling outside the cabinet. Setting up simple routines pays off: check the seals after every use, swap out desiccants before they clump, and avoid transferring between bottles unless absolutely necessary. These acts seem minor until someone tallies up the cost of losing a critical reagent mid-project.
The Bigger Picture
Good storage habits protect health as well as integrity. Avoiding spills and vapors guards both research and the people running it. Documentation, consistent storage practices, and keeping chemicals in tip-top shape support reproducible science and minimize unexpected downtime. Old habits may die hard, but they’re worth trading in for the confidence that comes from knowing your reagents will behave the way science textbooks say they should.
Pyridinium trifluoroacetate doesn’t kick up much dust in the mainstream. Most people aren't likely to see it outside of a chemistry lab, but for anyone dabbling in organic synthesis, this chemical rings a bell. It's used because it brings unique properties to the table—especially as a catalyst or reagent in certain chemical reactions. The thing is, the devil’s always in the details, and with chemicals like these, you don’t get a flashy label warning you just how potent or risky things can get.
Why Pyridinium Trifluoroacetate Has People Talking
I’ve spent enough time around labs to notice how easily people gloss over the fine print. Pyridinium trifluoroacetate looks tame: a white powder, without much in the way of noxious fumes like some old-school reagents. Yet, its apparent harmlessness is part of the problem. There’s a misconception that lab chemicals need to look sinister to cause real harm. All it takes is a quick look at some material safety data sheets to realize that’s a big mistake. Pyridinium trifluoroacetate is classified as an irritant and, if handled carelessly, can do a number on your skin, eyes, and respiratory system.
Touching it without gloves can lead to redness and that itchy, burning feeling you don’t shake off easily. Accidentally breathing it in might leave your throat irritated or coughing. The bigger concern is that information on long-term health effects from repeated exposure remains spotty. It hasn’t had broad community attention, so you won’t find decades of studies tracking cancer rates or chronic toxicity in people using it every day.
Invisible Risks in Everyday Work
I remember my earlier days in research, where proper handling sometimes took a back seat to finishing a late-night project. Sometimes gloves run out, sometimes goggles fog up, and sometimes shortcuts creep in during busy stretches. That sense of urgency amplifies risk. With pyridinium trifluoroacetate, these lapses multiply the chances of exposure. The trifluoroacetate part brings its own set of hazards, tied to fluoride chemistry—these can cause issues ranging from acute toxicity to longer-term environmental damage.
Environmental release rarely makes headlines, but trifluorinated compounds don’t break down easily. They linger in soil and water, posing challenges to wildlife and ecosystems. These consequences don’t get much limelight amid the rush of chemical innovation, but the planet keeps tabs, even if we forget.
What It Means for Labs and Beyond
Training plays a big role here. Lab safety protocols need to rise above the "just read the MSDS" mentality. There’s value in straightforward signage where the chemical lives, real practice using fume hoods, and routine supply checks for gloves and eye protection. Substitution offers another option—when possible, people should push for safer alternatives or greener methods. In recent years, chemical suppliers and university labs have started to embrace greener chemistry, but it’s not always simple to switch out the tried-and-true for the new and untested.
People demand more transparency before putting new food additives or cosmetics on the market. Yet, thousands of chemists handle reagents every day with only a slim folder of data on what a spill, splash, or routine exposure means long-term. It only takes a careless move to remind someone how real the risks are. If there’s one message worth repeating, it’s that convenience can never outrank caution, especially with substances like pyridinium trifluoroacetate. Being aware and prepared isn’t just about ticking safety boxes—it’s daily due diligence for anyone serious about safe science.
Looking Past the Label
These days, it’s easy to overlook the finer points of chemistry until a reaction goes sideways. Pyridinium trifluoroacetate might sound esoteric, but in a synthetic organic lab, it shows up as a quiet workhorse. I remember the first time someone handed me a flask containing this stuff—there was some hesitation. Blame it on a string of past experiments ruined by impure reagents that could’ve just as well come from the bottom of a coffee pot.
Spotting the Good Stuff
Anyone handling pyridinium trifluoroacetate expects it to come as a white to off-white crystalline powder. Sometimes, it shows up faintly yellow, especially if it’s been exposed to a little moisture or left around for longer than you’d like. Even the best vendors can’t beat the laws of thermodynamics, and a hygroscopic compound like this will soak up water faster than dry rice in soup.
A chemist trusts their eyes as much as the label. Perfect white crystals tend to signal little in the way of contaminants, but a yellow tint should give pause. Many labs still rely on this quick once-over, but nothing replaces TLC, NMR, or a reliable melting point check. Lab benches worldwide are littered with stories of reactions turned to goo, just because someone trusted a supplier's word without testing for themselves.
Purity’s Impact on the Real World
In the field, purity isn’t some academic bragging right. People use pyridinium trifluoroacetate as a proton source in organic synthesis, especially in peptide chemistry, because its trifluoroacetate group helps shuttle protons efficiently. Even trace impurities can sideline a synthetic scheme, introducing byproducts that are almost impossible to get rid of without starting over. No one wants to discover the problem after weeks of wasted effort or watch their column chromatography separate a rainbow of mystery compounds.
Good labs don’t just rely on the stated purity from suppliers. Analytical data—often showing >98% or higher—is double-checked. Most reputable batches clock in at this mark. That means researchers actually confirm what they’re using, ensuring it won’t sabotage the next experiment. Who wants to spend a grant budget on starting material that promises high purity and then doesn’t deliver, only to see a project’s data go up in smoke?
Fixing the Gaps We Know
Adopting a culture of checking, not just trusting, prevents headaches. Labs that run quick checks on their batches, storing this compound tightly capped and away from humidity, sidestep common pitfalls. I’ve seen labs rescue expensive projects by stopping to double-check material that looked a little off, even if manufacturers promise “optical purity” or similar claims.
Open communication between chemists and suppliers also goes surprisingly far. When inconsistencies pop up batch to batch, it’s the researchers pressing for answers who end up with better outcomes in the long run. A little skepticism, paired with concrete analysis, makes for solid science.
Focusing on purity and keeping an eye on appearance matters, not just for getting clean data, but for trust in the whole scientific process. That’s as close to a constant as you’re going to find in chemistry.