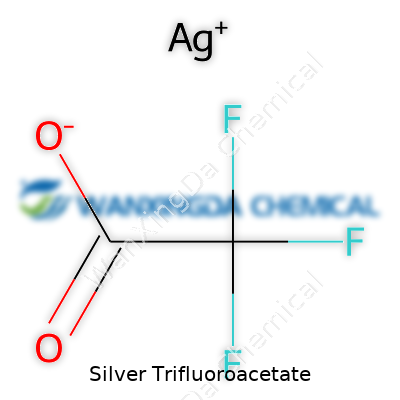
Silver Trifluoroacetate: A Deep Dive into Its Role and Relevance
Historical Development
Silver compounds have been around for centuries, but the turn toward complex organometallic chemistry brought Silver Trifluoroacetate into focus in the late 20th century. Researchers, searching for new reagents to widen the toolkit of organic synthesis, stumbled onto the unique chemical dynamics brought by adding trifluoroacetate to silver. This compound stood out in labs not only for its reactivity but for the way it played with both organic and inorganic chemistry, creating bonds and breaking them apart with an efficiency that was hard to ignore. Laboratories working on fluorinated organics especially pushed Silver Trifluoroacetate into wider use, as its role in introducing trifluoromethyl groups began to matter more for the pharmaceutical and agrochemical sectors. What started as an experimental material in small-scale syntheses gained traction as innovation and industry needs grew.
Product Overview
Silver Trifluoroacetate does not appear in most homes or even in most industrial warehouses. It holds a spot in research facilities and specialized chemical plants, packaged with care due to both its potency and cost. The compound’s unique value lies in its ability to swap trifluoroacetate ligands with other functional groups, opening up transformation pathways that prove challenging with less reactive silver salts. From my work with organic synthesis and metal-catalyzed reactions, I've seen that chemists appreciate reagents that save time, yields, or steps in syntheses—the sort of qualities this silver salt can deliver.
Physical & Chemical Properties
Silver Trifluoroacetate appears as a white to greyish solid, with a notably pungent odor due to its trifluorinated nature. Most chemical workers know silver salts require a bit more care with light, so they keep these bottles in amber glass to avoid degradation. It dissolves in polar solvents like water, acetonitrile, and even some alcohols, which offers flexibility. The compound tends to decompose at elevated temperatures, sometimes releasing aggressive vapors, so fume hoods are the norm during any manipulation. This reactivity has roots in the delicate balance of silver’s easy reduction to metallic silver and the electron-withdrawing trifluoroacetate, both of which make it a favorite for redox-driven organic transformations.
Technical Specifications & Labeling
Nobody can walk into a stockroom expecting all silver salts to carry the same punch. Silver Trifluoroacetate typically arrives in tightly sealed vials with strict labeling: purity, batch number, moisture content, and (for research-grade material) trace metals. Careful handling is part of the ritual. The labels usually warn of light sensitivity and the need to store cool and dry. From experience, good labeling matters most when accidents or mix-ups risk costly experimental loss.
Preparation Method
Labs prepare Silver Trifluoroacetate by reacting silver oxide or carbonate with trifluoroacetic acid. The reaction brings together the strong acid with a mild base, forming water and a salt as the main byproducts. Filtering off the spent reagents leaves a solution, which can be slowly evaporated to yield shiny silver trifluoroacetate crystals. Some operations scale this up for industry, but most workers stick with small-batch syntheses due to cost and safety concerns.
Chemical Reactions & Modifications
This reagent stands up in a few roles that make it valuable. In my lab days, I saw it used to mediate halogen exchange reactions: swapping a bromide for a trifluoromethyl group or activating an arenesulfonate for coupling. Chemists also use it to generate carbocations from weak leaving groups or to produce organosilver intermediates that unlock novel carbon-carbon bond formations. Modifications involve playing with the trifluoroacetate anion: tweaking the acid or solvent profile can push different selectivity or yield, depending on what biochemists and engineers are after. Its lability—the ease with which the trifluoroacetate leaves—lets scientists tune reactivity without major hardware or process changes.
Synonyms & Product Names
Outside academic literature, Silver Trifluoroacetate often shows up as silver trifluoroacetate, silver(I) trifluoroacetate, and AgTFA. Catalog IDs or local shorthand may pop up, but these technical names guide researchers to the same glass vial of reactive white powder. Anyone walking into a university stockroom or browsing a catalog for “AgO2CCF3” lands on the same high-purity compound.
Safety & Operational Standards
Safety doesn’t just come from reading a datasheet. Handling Silver Trifluoroacetate means gloves, goggles, and attention to ventilation. It causes skin and eye irritation if mishandled, and its dust proves especially corrosive over time. Spills stain benches and skin dark brown, a warning that silver ions are never entirely benign. Most chemists store it away from strong acids—except for the one that built it—and away from light. Disposal rules require sending it to licensed chemical waste handlers. The rise of newer occupational standards for lab workers has made this culture of caution routine. Reliable fume hoods, chemical carts, and segregated storage cut down the risk for those who work with it daily.
Application Area
Silver Trifluoroacetate steps into synthetic organic chemistry as a go-to for demanding fluorine chemistry and cross-coupling reactions. Pharmaceutical labs hunting for new drug candidates—especially ones with fluorocarbon motifs—reach for Silver Trifluoroacetate to introduce the trifluoromethyl group, which boosts metabolic stability in promising molecules. Agrochemical researchers lean on its reactivity for pesticide synthesis, often where traditional halogenation or diazotization routes stall out. Some materials science efforts also rely on the compound to deposit thin metal films or manipulate crystalline architectures in advanced sensor technology. Its place in research, compared with mass production, remains dominant, owing to its price tag and specialized utility.
Research & Development
Scientists haven't finished finding new uses for Silver Trifluoroacetate. With advances in catalysis, solvent engineering, and green chemistry, researchers keep searching for ways to coax more selective or less toxic reactions using this reagent. Recent literature points to emerging reactions in photoredox chemistry, where light turns Silver Trifluoroacetate into a uniquely active catalyst, making transformations once considered expensive or unpredictable more reliable. Collaboration between academic labs and industry stretches the boundaries—experimenting with microreactors to minimize waste, or pairing Silver Trifluoroacetate with milder conditions to unlock new reaction pathways. My own brush with late-stage functionalization projects showed me how precious silver salts could, in clever hands, solve problems the textbooks barely sketch out.
Toxicity Research
Regulatory bodies mark silver compounds for scrutiny not just on human contact but for aquatic and environmental impact. The trifluoroacetate anion remains persistent, with potential to bioaccumulate. Toxicity studies peg Silver Trifluoroacetate at moderate risk: harmful if swallowed or inhaled, and distinctly uncomfortable if splashed onto eyes or skin. Chronic exposure may lead to argyria—skin discoloration due to silver deposition. Recent research pays close attention to environmental persistence, as concerns over trifluorinated organics mount. Labs now run routine monitoring of waste streams, looking for traces of either the silver or the trifluoroacetate, driven both by stricter laws and a growing ethical commitment to safety. For anyone working long term with this material, health surveillance—checking for early allergy or skin changes—forms a sensible part of the job.
Future Prospects
Silver Trifluoroacetate sits at a crossroads familiar to many specialty reagents. On one hand, fluorinated chemistry rolls on, powered by medical innovation, advanced diagnostics, and a hunger for materials that endure under stress. On the other, scrutiny over environmental safety and cost keeps the compound in special-use territory. Academic and industry researchers now look for ways to recycle silver, recover trifluoroacetate, or craft “greener” analogs without sacrificing performance. There’s excitement in merging this time-tested reagent with modern process intensification—continuous flow syntheses, hybrid catalysis, and digital monitoring—to squeeze out more value and less waste. As regulatory oversight sharpens and innovation unlocks new synthetic tricks, Silver Trifluoroacetate’s future depends on just how well the scientific community balances practical performance with environmental and social responsibility.
Silver trifluoroacetate doesn’t get much attention outside of chemistry circles, but its impact inside the lab goes a long way. Most folks walk by a chemistry building at a university or research company without guessing the tools inside are shaping more than just grade sheets. Us working with weird-looking bottles and powders occasionally get wind of a new compound that quietly changes how we tackle big problems, from fighting disease to building better batteries. Silver trifluoroacetate joins this club, and it’s worth understanding why.
Fueling Research and Innovation
Chemists prize silver trifluoroacetate for its solid performance as a reagent. You don’t see it on supermarket shelves, but behind the scenes, it acts like a trusted teammate in making carbon-carbon bonds or swapping out hydrogen atoms for fluorine in complex molecules. In pharmaceutical research, drug makers often chase compounds that resist breakdown inside the body. Fluorine atoms help with that. Silver trifluoroacetate stands out because it makes adding fluorine to molecules faster, cleaner, and in fewer steps. These time savings have a bigger ripple effect: Faster syntheses mean labs can make more test compounds, giving doctors and patients a better shot at new treatments for tough diseases like cancer or viral infections.
Environmental Impact and Industrial Scale
Factories making chemicals at larger scales run into all sorts of headaches. Waste products can pile up, and each extra step in a process raises energy costs. Fluorination, when handled with older chemicals, can create toxic byproducts and dangerous conditions for workers. Labs using silver trifluoroacetate flag it for being a little more manageable and predictable during reactions. Fewer accidents, less mess, and savings on the back end help keep production costs in check. If demand for fluorinated materials grows—think nonstick coatings or specialty plastics—industries count on these kinds of chemicals for speed and safety.
Concerns and Responsibilities
Working with powerful reagents like silver trifluoroacetate brings up questions around health and long-term risks. As with any silver compound, improper disposal or long-term buildup in the environment causes concern. There’s also the matter of cost. Silver-based chemicals never go cheap, so reliance on them raises questions of sustainability. Bigger labs recycle silver. Still, there’s room for improvement in how we reuse precious metals and reduce waste.
Innovation shouldn’t come at the expense of responsibility. Better training on handling and disposal, creative recycling techniques, and searching for alternatives could limit the downsides. As an example, my own experience in a university lab taught me to treat every new bottle as both an opportunity and a risk. Deciding which chemicals to use went beyond the lab bench; we checked if the supplier sourced metals ethically and mapped out a disposal plan before popping any seal.
The Road Ahead
Silver trifluoroacetate’s story shows how a humble powder makes big waves in science and industry. Its promise lies in streamlining tough reactions and supporting new discoveries, while its challenges remind us to stay alert to questions of safety, cost, and stewardship. People tend to overlook the day-to-day realities behind chemistry breakthroughs, but these decisions push science forward just as much as bold theories or clever inventions.
Chemistry That’s Simple on Paper, But Speaks Volumes in the Lab
Most chemists run into all sorts of formulas, but few stand out the way Silver Trifluoroacetate does. It’s not just another string of letters and numbers—though its formula, Ag(OCOCF3), rolls off the tongue for anyone used to reading chemical shorthand. Silver paired up with the trifluoroacetate anion creates a compound that slips into a surprising range of reactions. If you’ve ever watched silver nitrate transform a solution, you get a taste for how dramatic these silver salts can be.
Chemistry Teachers and the Allure of Useful Reagents
During my own university days, I remember the sense of caution you pick up every time a silver compound hits the benchtop. Silver Trifluoroacetate isn’t as famous as its nitrate cousin, but in organic synthesis, it offers a flexibility you might not expect from something so simple. Chemists appreciate its ability to introduce trifluoromethyl groups and drive halide abstractions, something that plays a pretty important role when building molecules for pharmaceuticals or materials research.
Research articles and patents stack up naming Silver Trifluoroacetate as that key ingredient. It does more than just swap halides; it makes transformations gentler on delicate molecules, which opens up more possibilities for advanced chemistry. Try telling a medicinal chemist they need to synthesize a tricky target molecule—odds are good they’ll reach for this reagent before going down a list of more aggressive alternatives.
Costs, Waste, and Practical Concerns
Now, nobody working in the lab ignores the downside—silver is expensive, and this particular salt doesn’t come cheap, especially as prices for precious metals fluctuate. Handling costs stack up quickly for any department. On top of that, disposal and environmental responsibility weigh on every decision. Unlike simple organic compounds that break down easily, silver-based waste can linger and must be managed with care.
Silver runs the risk of polluting lab wastewater or ending up in landfills where it shouldn’t, creating a growing push to recover and recycle it. That’s a constraint many researchers are wrestling with today, especially in university labs where funding jars rattle empty by the semester’s end. Moving forward, teaching students how to recover and purify silver from spent reagents isn’t just a cost-saving measure but an important lesson in sustainable lab work.
Alternatives in the Pipeline
No one in science expects a single reagent to rule forever. Interest keeps rising in greener, less toxic alternatives. Some labs experiment with copper salts or entirely organocatalytic routes, though the efficiency and selectivity Silver Trifluoroacetate delivers can be tough to match. Investment in greener chemistry means looking for ways to get the same results with less financial and environmental strain. Innovation, backed by research, slowly chips away at the need for such specialty compounds.
Silver Trifluoroacetate isn't just a set of letters to memorize for an exam—it tracks the evolution of chemistry itself. From bench-top challenges in waste management to the creative power it provides in organic chemistry, it stands as a reminder that behind every chemical formula, there’s a bigger story about cost, stewardship, and scientific progress.
Working With a Sensitive Chemical
Silver trifluoroacetate sounds like one of those lab chemicals few outside professional settings have touched. For those of us who have handled these materials, the importance of careful storage comes pretty quickly. This isn’t table salt or sugar — there’s reactivity in every bottle, plus a silver ion that has a knack for tarnishing or causing unexpected reactions. I remember a colleague’s container turning dark within weeks just sitting out. It spurred some serious discussion, and sloppy storage never happened again.
Why the Worry?
The trifluoroacetate part of this compound brings a powerful trifluoromethyl group to the mix. Silver, in this form, is no stranger to light sensitivity and can break down to metallic silver if left exposed on a bench under bright lab lighting. Any moisture in the air – be it humidity creeping into the lab on a July afternoon or a little condensation from opening a cold bottle – spells trouble. Left uncapped or even loosely capped, the material clumps or oxidizes. There goes purity and often, usefulness.
Practical Storage Choices
I’ve seen the best results when people store bottles in tightly sealed containers, away from light, and definitely in a dry place. We’re not talking about a desk drawer or a random spot on an open shelf. Secure, labeled chemical cabinets built to keep out moisture and light keep things stable the longest. Some labs put a desiccant packet in storage boxes to absorb any stray moisture. That extra step saves money in the long run, stopping degradation in its tracks.
Temperature matters, too. Standard practice among chemists—myself included—leans towards a refrigerator or temperature-controlled storeroom for anything sensitive. Yet, freezing isn’t always the answer. Freezers can introduce condensation when those bottles come back to room temperature. There’s wisdom in steady, moderately cool storage instead.
The Cost of Cutting Corners
A bottle gone bad isn’t just a minor headache. It's wasted budget, lost material, and potential risk if someone tries to use degraded compound in a reaction. Silver compounds stand out for their stubborn staining ability, too. Once you’ve tried to wipe up a silver salt spill, you remember it. The stain lingers. Mishandling can slip hazardous chemicals out of control, putting people at risk.
Keeping Safety Real
Labs that take storage seriously have solid training. Labeling dates, worker names, and condition checks bring accountability. People learn to spot crystal changes or discoloration before it turns into a bigger problem. Good habits start at home, or in the classroom, but really sink in once someone faces the fallout from a ruined reagent.
Solutions? Give staff the right tools. Chemical-grade containers, lockable dry storage, and clear protocols make a difference. Give everyone in the lab responsibility for the chemicals they take out—and bring back. Chemistry, like cooking, works best with fresh ingredients, and fresh ingredients stick around longest with honest attention to how they’re kept. Investing in safe storage cuts headaches and keeps valuable chemicals, like silver trifluoroacetate, ready for work.
Taking a Closer Look at the Risks
Silver trifluoroacetate isn’t a household name, though it finds a place in chemistry labs, research facilities, and some niche industrial corners. On the surface, it sounds like a simple silver salt. Its job is to act as a reagent in a range of syntheses, especially when chemists are building complex organic molecules. If you’re not working at the bench, it’s easy to overlook why the hazard question matters. But for those handling it, risks deserve real attention.
What Science Says: Toxicity and Exposure
Anybody who’s spent time in a lab understands that chemicals with “trifluoro” tacked onto the name tend to register on the caution scale. Silver trifluoroacetate carries both silver and fluorinated components, and this mix isn’t exactly benign.
Silver itself can build up in the body with repeated, high-level exposure and cause a condition called argyria – a permanent bluish discoloration of the skin. This doesn’t mean touching a little will turn you into the Blue Man Group. Still, accidents with silver compounds are rarely brushed off as harmless. The trifluoroacetate part, built on the trifluoroacetic acid backbone, brings its own edge. Trifluoroacetic acid is well documented for its corrosivity. It burns skin, damages eyes, and when inhaled, eats at the lungs.
One researcher I know spilt a few grams a decade ago. Gloves, lab coats, and goggles blocked direct skin contact, but fumes hung in the air. The whole crew had to evacuate the room and ventilate for an hour, just in case. It set off a lively debate about whether chemical frugality is worth it if safety corners are cut. No one forgot that lesson.
What Happens Outside the Lab?
Most people never cross its path, but disposal from research sites sparks another thread of concern. Silver trifluoroacetate can contaminate water systems and soil if not handled by professionals. Both fluorine and silver ions stick around and can stress ecosystems. There’s hard evidence that even trace amounts of silver can disrupt aquatic life, causing problems for fish and microorganisms in rivers and streams. The trifluoro group is persistent too—Unwelcome company for anyone who values a clean water supply or healthy soil.
The Importance of Oversight and Best Practices
I’ve seen regulations tighten over the years, but enforcement doesn’t always keep pace with new chemicals entering labs. Workers deserve straightforward information about what they’re handling and practical advice, not just lists of rules. Using fume hoods, sealing waste, and storing reagents in clearly marked containers go a long way. Shortcuts on safety never turn out to be real timesavers; too many “close calls” prove that point.
A safer future relies on a culture where asking hard questions about chemical safety isn’t an exception but the rule. Companies, universities, and research groups can revisit their protocols and invest in better training. Substituting less hazardous reagents, where possible, also deserves more focus.
Personal Perspective on Chemical Responsibility
Chemistry holds power to drive progress, but also demands respect for the risks woven into every bottle and beaker. Silver trifluoroacetate’s hazards won’t become front-page news, yet the lessons from its careful use add up across the scientific community. Prioritizing people’s health and a better environment makes sense, not just for the experts on the front lines, but for the rest of us who trust the safety net they build.
A Look at Silver Trifluoroacetate’s Value Beyond the Textbook
Silver trifluoroacetate doesn’t live in the spotlight like gold or platinum, but this compound quietly shapes modern chemistry labs and a slice of today’s industrial processes. I came across it for the first time in a university organic chemistry lab. The instructor joked that, if we spilled it, we better wipe it up fast—not just because of cost, but because silver compounds stain like nobody’s business. That little moment stuck with me as I dug deeper into its chemistry and started to notice how often silver trifluoroacetate showed up on the edges of innovation.
Chasing Efficiency in Organic Synthesis
Ask anyone working in organic synthesis, and they might share a story about using silver trifluoroacetate for halide abstraction, especially with aryl iodides or bromides. The compound offers a quicker, cleaner route to making reactive intermediates, like carbocations or cationic metal complexes, by gently grabbing the halide atoms from organic molecules. This opens the door for reactions that might get bogged down with traditional reagents, helping researchers create new carbon–carbon bonds, especially in pharmaceutical discovery work.
The search for efficiency in making fine chemicals often relies on these side characters. With silver trifluoroacetate, you can avoid the fussiness of moisture-sensitive materials and harsh conditions tied to some older reagents. That means fewer failed syntheses and less time spent cleaning glassware—an unglamorous perk anyone who’s spent days in a lab will appreciate.
Catalysis and the Push for Cleaner Reactions
Chemical catalysis drives both research and real-world production. Here, silver trifluoroacetate brings out the best in certain palladium-catalyzed reactions, helping activate substrates in cross-coupling processes. Cross-coupling matters a lot, since the method forms the backbone for making agrochemicals, active drug components, and advanced plastics. Reliable sources have pointed to these reactions delivering higher yields and saving steps—real value for any chemist or industry trying to scale up from grams to kilograms.
Crafting oxidizing conditions takes finesse, since you want to push a reaction forward without tearing everything apart. Silver trifluoroacetate plays a steady hand as a mild oxidant, suited for selectively oxidizing organic molecules. In industry, this helps save on remediation costs and streamlines workflows by reducing dangerous byproducts.
Electronics and Advanced Materials
Outside organic chemistry, think about how electronics manufacturers fight for every bit of purity and reliability in their materials. Silver trifluoroacetate acts as a source of high-purity silver. You might not see its stamp on a finished sensor or conductive ink, but its chemistry makes possible the deposition of thin, even layers of silver, whether for circuit boards or flexible displays. Early experiments with printable electronics and new types of sensors have used this compound to make surfaces more conductive or responsive.
Meeting Modern Demands, Facing Challenges
Every chemical comes with baggage. Cost and safety concerns limit silver trifluoroacetate’s use outside strict research or high-value manufacturing. Researchers who work with precious metals know why every milligram matters. Regulatory hurdles mean labs and factories need to handle silver waste with care. Responsible chemical management—reuse, recycling, and developing greener substitutes—makes up as much of the conversation as pushing what silver trifluoroacetate can actually do.
A Compound with Quiet Influence
Silver trifluoroacetate represents the sort of ingredient that turns complicated chemistry into something practical. Even if you never handle it yourself, trust that somewhere in the chain of scientific progress, it’s helped move things forward. Creating better, safer, and more efficient uses for it will depend on investments in both new science and responsible stewardship. That approach matches the pace and priorities of today’s research and industry, grounding chemistry’s quiet helpers in the realities of modern production and discovery.