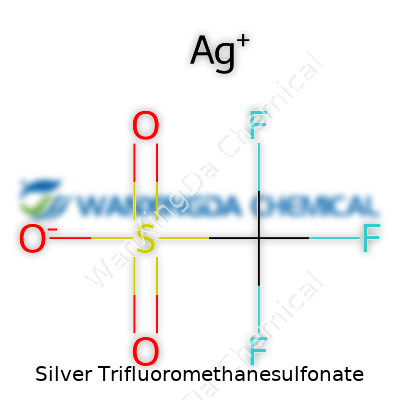
Silver Trifluoromethanesulfonate: Old Roots, New Roads
History in a Bottle
Silver trifluoromethanesulfonate didn’t pop up overnight. The story begins in the 1960s, back around the time chemists started chasing better counterions for advanced reactions. Researchers were hunting for salts that break away clean in polar solvents, especially as organic chemistry grew bolder. Silver triflate answered this need with its wide-open flexibility, and it gave organic chemists a much-needed arrow in the quiver. Over the decades, its distinct character made it a staple in research and experimental synthesis, always hovering near the edge of big discoveries.
What’s in the Bottle and What It Can Do
You’ll spot silver trifluoromethanesulfonate looking like a fluffy, white powder—hardly impressive at first glance, but don’t let that fool you. Its structure couldn’t be simpler on paper: AgCF3SO3. Toss it in a flask and you unlock more than just its appearance. It dissolves nicely in polar solvents like acetonitrile and dimethyl sulfoxide but shies away from ordinary water. This salt combines the oxidative muscle of silver with the non-nucleophilic background of the triflate, giving it some surprising powers in synthesis. Chemists count on its stable, anhydrous nature and love that it doesn’t bring along extra baggage, making it a reliable pick for delicate work.
Preparation and Chemistry at the Bench
Crafting silver triflate isn’t rocket science, though it’s not kitchen chemistry. Most labs marry silver oxide with trifluoromethanesulfonic acid, squeeze out a little water, then filter and dry the result under vacuum. The purer the starting ingredients, the less hassle down the road. The final salt proves itself as more than just a source of silver—swap out halides or activate soft nucleophiles, and watch the reactions flow cleaner thanks to that non-sticky triflate. Ag-triflate frequently steals the show in carbocation chemistry, cross-coupling, and even some modern medicinal syntheses. I’ve seen colleagues coax tough molecules into line using this salt, especially when standard silver salts stalled or made a mess.
Names Across the Globe
Ask around a research lab, someone might call it Silver Triflate, Silver Trifluoromethanesulfonate, or by its handier formula AgOTf. These aliases all line up with databases and journals worldwide. Such synonyms mainly help researchers avoid mix-ups, but no matter the label, the flask’s contents tell the story.
Safe Handling in the Real World
Silver triflate isn’t the nastiest thing on the shelf, yet its safety calls for respect. No one needs a silver salt dusting their skin or eyes. Gloves, dust masks, and a solid fume hood cut down the risk to nearly zero. If you’ve ever spilled a heavy-metal compound, you know cleanup means more than a wet rag. Thanks to triflate’s stubborn chemistry, you sidestep fire hazards or environmental nightmares seen with quite a few other organometallics. The biggest hazards come from chronic exposure or careless discard; silver builds up in the body and can haunt waterways if tossed down the drain. That reality nudges every responsible chemist to keep disposal tight and avoid shortcuts.
Where Silver Triflate Moves the Needle
The toolkit of a research chemist keeps growing, but silver triflate keeps its spot near the center. Synthesis of fine chemicals, especially those tough carbon skeletons in drug trials, draws heavily on its abilities. Move into carbohydrate chemistry and it’s right there, helping to nudge along glycosylations where stubborn sugars refuse to cooperate. Electron-rich complexes for catalysis and electronics often start with a swipe of silver triflate in the clean-up step. Even new frontiers like battery chemistry and conductive polymers have toyed with its role. I’ve watched colleagues in the pharmaceutical industry spin up multi-step syntheses that floundered until AgOTf came off the shelf.
Current Research and Rough Edges
Research into silver triflate continues to peel back new uses. Lab groups keep hunting for greener ways to use and recover silver, pushing past old habits. There’s also plenty of action in catalysis, trying to unlock milder or more selective routes in complex syntheses. Toxicology research hasn’t stood still either; as chemists push up the scale, every bit of exposure matters. Real toxicity in humans rings louder when large volumes and chronic exposure come into play, especially since silver accumulates slowly and quietly in tissue. Lab tests show it doesn’t vaporize under normal conditions, but it will linger on surfaces and in solutions unless cleaned properly. Regulations nudge labs toward best practices, and that discipline pays off with safer workplaces across the world.
The Road Ahead
Silver triflate still has untapped potential in the lab and far beyond, though the price of silver slows down wide adoption. If the cost curve can soften, or if recycling silver ions from spent reactions gets streamlined, every corner of chemical synthesis could open up new doors. Some labs are even tinkering with hybrid salts, looking for similar punch without the expense. With the drive for more sustainable and precise synthesis, the value of smart, well-characterized reagents like AgOTf keeps climbing. For those digging deep into new reaction pathways or crafting tomorrow’s medicines, silver triflate stands as both a proven workhorse and a ticket to new territory, so long as the industry keeps walking the line between safety, cost, and innovation.
Silver trifluoromethanesulfonate, often called AgOTf for short, isn’t something that shows up on the radar of the average person. Yet, for chemists and folks working on the frontlines of innovation, this compound carries a lot of weight. Think of it as a skilled player on a team — not a superstar with a household name, but essential for running the plays that keep progress moving.
Connecting the Dots: Where AgOTf Fits
Walk into a chemistry lab, and you may find a bottle of this white powder quietly sitting among much flashier reagents. Its main appeal comes from its knack for getting things started. In particular, chemists use AgOTf to help kick off reactions that produce new molecules. It handles this by acting as a Lewis acid. What does that mean for folks not buried in textbooks? Picture AgOTf as a catalyst, nudging other chemicals to combine and form new products. This makes it a go-to for creating carbon-carbon and carbon-heteroatom bonds, both key steps in assembling complicated pharmaceuticals or advanced materials.
I remember working on a reaction that simply refused to start, even with the usual suspects. Swapping in AgOTf saved the project. That moment stuck with me — sometimes progress depends on knowing which tool for the job will unlock the next step, even if it isn’t flashy.
Pharmaceuticals and Beyond
One of the big reasons people care about AgOTf comes down to what it makes possible. Drug research often moves slowly or stalls because the reactions can’t deliver the exact molecule needed. AgOTf’s reliability in making certain bonds streamlines experiments and sometimes prevents dead ends. Companies racing to discover better treatments, or universities experimenting with new therapies, turn to compounds like AgOTf because it can mean the difference between a failed and a successful trial.
Another important use pops up in the field of materials science. AgOTf helps add functional groups to organic molecules, tweaking their properties so they can conduct electricity better, or bind to metals in special ways. These tweaks might end up in new solar panels, smarter sensors, or batteries that last longer. For those of us who rely on technology working—even if we don’t always see what’s under the hood—tools like AgOTf matter more than we might think.
Real-World Impact and Safety Matters
Handling AgOTf calls for care. It isn’t toxic in the sense that it will send someone straight to the emergency room, but gloves and fume hoods should always be in the picture. A small group of researchers handles most of it, so oversight is easier. Still, labs need to keep a close eye on procedures to prevent careless mistakes. It helps to remember that every lab accident sets back research that could lead somewhere important.
Supply and cost sometimes present hurdles. Silver is expensive, and global shifts in material prices ripple right into research budgets. Making AgOTf isn’t trivial either, which means labs think twice before using it in large-scale production runs. In my experience, careful planning and sharing tips across teams eases these pain points, though funding agencies could do more to account for these realities too.
Looking Forward
As labs keep chasing better drugs, cleaner energy, and smarter materials, compounds like silver trifluoromethanesulfonate will keep playing a part. The more researchers share their stories about what works — and what doesn’t — the better our collective shot at breakthroughs gets. Sometimes, just knowing the right tool exists can breathe life into an idea and push it further than expected.
Looking at a Chemical Name
Some chemicals roll off the tongue, others twist it into knots. Silver trifluoromethanesulfonate belongs to the second group. Its long name hints at a complex structure. Yet at its core, the conversation is pretty simple and practical—especially if you’ve ever dealt with catalysts or chemical synthesis in a lab. The chemical formula, AgCF3SO3, packs a punch. Understanding why it matters starts with recognizing each part: silver for reactivity, trifluoromethanesulfonate to stabilize and guide reactions in ways that traditional salts can’t.
Experience with Salts Like This
I’ve seen chemists reach for silver triflate, as it’s often called, when they hit a dead end using more common reagents. In my own experiments with organic reactions, silver salts sometimes offer that missing nudge, especially during tough substitution steps or in situations where you need a non-coordinating anion. The trifluoromethanesulfonate part doesn’t stick around and disrupt the chemistry in the same way some other anions might. AgCF3SO3 lets chemists push the envelope, pulling off reactions that otherwise stall or refuse to complete.
Importance in Today’s Labs
Nobody chooses a fancy salt just for the fun of it. Take cross-coupling, a workhorse reaction in making pharmaceuticals and advanced materials. Silver triflate opens doors. It activates halides, forms useful complexes, and sometimes, brings a clean finish where others leave a mess. Companies racing to build new drug molecules lean on this kind of chemistry. AgCF3SO3 keeps the chain moving, allowing new bonds to form without the heavy baggage that clunky, reactive counter-ions introduce. Facts back this up—the presence of the triflate group reduces nucleophilicity and increases thermal stability of intermediates, giving better control in many synthetic steps.
Weighing Drawbacks and Solutions
No silver bullet exists in chemistry, and silver trifluoromethanesulfonate brings costs. Silver isn’t cheap, and this salt doesn’t come in bulk prices. Disposal can’t be brushed off either—silver waste calls for special handling, since nobody wants to introduce heavy metals into soil or water. Lab teams get creative here—recovering silver from spent reagents, swapping in less expensive metals for less critical steps, or fine-tuning reaction conditions to squeeze every atom’s worth out of the material.
Focus on Responsible Chemistry
Chemistry evolves. Demand for sharper, more selective reactions isn’t easing up. Neither is the pressure to minimize waste and cut reliance on rare metals. Teams share protocols, publish green chemistry tweaks, and explore catalysts that limit precious metal use. All this while respecting the rules—the Environmental Protection Agency and similar regulators set strict limits for silver disposal. Smart labs put tracking systems into place and experiment with silver recovery resins or electrolytic traps instead of letting good metal slip away.
Keeping Facts Straight
Missteps happen less often than they used to, partly because chemists share knowledge so rapidly now. The formula—AgCF3SO3—and its uses crop up in journals, workshops, and in the lively chatter of senior researchers guiding newcomers through tricky reaction pathways. The path from name to application hinges on solid evidence. In the spirit of responsible science, folks keep questioning their tools, weighing pros and cons, and always chasing better solutions. Whether scaling up a drug route or exploring an unknown transformation, the facts behind silver trifluoromethanesulfonate stay close at hand.
Why Storage Isn’t Just a Box-Ticking Exercise
Silver trifluoromethanesulfonate, known around the lab as silver triflate, doesn’t attract much attention outside of chemistry circles. Ask anyone who works with it regularly, though, and they’ll tell you storage decisions carry real consequences. Looking back at my time in academic and commercial labs, a single slipup could mean a ruined batch or a dangerous mess.
Chemicals don’t always make headlines, but their storage can come with outsized risks—silver triflate is no exception. Moisture, light, and heat cause it to degrade or react. The backstory here isn’t mysterious: moisture triggers hydrolysis, light sparks decomposition, and fluctuations in temperature push it toward instability. Overlooking storage details creates safety hazards, wastes money, and adds frustration nobody needs.
A Dry, Cool Routine Beats Out Wishful Thinking
Moisture control stands out. Labs I’ve worked in usually seal silver triflate tightly in its original bottle or, if that’s damaged, a solid glass vial. Lab culture in some places still treats desiccants—those silica gel packets—as optional. My own mistake with skipping them once taught me otherwise; moisture crept in, and the silver triflate clumped into an unusable chunk. Investing time to add silica gel seems minor, but it saves headaches down the line.
Cool, stable temperatures keep the compound steady. One of my colleagues stored a new batch on a shelf near a heater for convenience—expecting only a short delay before use. No surprise the next day: discolored powder thanks to mild heat exposure. Refrigeration works well, but ultra-cold freezers drive up energy costs for little gain. Best practice means aiming for the same temperature you’d want for most pharmaceuticals: around standard room temperature, a bit cooler if possible, and definitely away from any heat source.
Light Protection: Not Just for Delicate Materials
Silver salts respond poorly to light. Leaving them on the benchtop under bright bulbs changes their character quickly. In one shared facility, open shelving left several bottles exposed—bad luck forced a rushed order for replacements. Simple foil wrapping around the vial or sticking to amber glass bottles makes a real difference here. Even strong room light, not just sunlight, breaks down the compound if given enough time.
Label Everything—Common Sense, Not Just Compliance
Forget about compliance for a moment and focus on practicality: everyone’s had the experience of opening an unlabeled or poorly labeled jar, wondering what lurks inside. Clear labeling with name, hazard warnings, and storage date isn’t just for regulatory checklists. It’s so the next person—the one who didn’t originally receive or seal the batch—knows exactly what they’re handling. Mistakes drop when everyone follows this simple habit.
Toward Tighter Safety and Less Waste
Instances of contamination, spoiled work, or accidental exposure to moisture aren’t rare in busy labs. Strengthening storage routines means reducing waste and avoiding loss of time and money. Auditing storage every few months helps; you find the accidental leaker or the container hidden behind a box of pipette tips. A quick retraining session now and then pays lasting dividends. Reliable access to tightly sealed containers, desiccants, and light-blocking storage, though simple, blocks many problems before they start.
In the real world, solid chemical handling grows from small, disciplined actions repeated every day—not from rulebooks, but from the lessons learned through hands-on frustration and the knowledge that good practice protects both people and the work they care about. Silver triflate deserves that respect.
Looking Beyond the Lab: Hazards of Silver Trifluoromethanesulfonate
Silver trifluoromethanesulfonate turns up mostly in chemistry laboratories and industry settings, where chemists count on it as a powerful catalyst and reagent. At first glance, those glass bottles don’t raise alarms. But anyone who’s handled this compound for a living knows there’s more to the story. Hazardous materials often hide in plain sight, especially ones that sound like a tongue twister.
What Makes It Hazardous?
People tend to underestimate chemicals that don’t eat through gloves on contact or fill the room with choking fumes. Silver trifluoromethanesulfonate doesn’t behave quite that way, but danger still lies beneath the surface. The compound irritates skin and eyes. It can leave you with a nasty burn or cause a lingering cough if inhaled as a fine powder. Breathing in even small amounts over time can lead to chronic respiratory issues. Mix it with water and you won’t see drama on the surface, but fumes can form.
According to published research and hazard summaries from government safety boards, silver compounds disrupt the body’s ability to process certain enzymes and can build up in major organs over months or years. Skin can turn grey-blue—a condition called argyria—after repeated exposure. Trifluoromethanesulfonate, the other half of this molecule, unlocks persistent environmental risks. It doesn’t break down easily, and can find its way into groundwater.
Why People Keep Using It
Chemists stick with silver trifluoromethanesulfonate because it solves tough problems. It opens up synthetic pathways in pharmaceuticals and electronics manufacturing that simpler chemicals can’t manage. This performance comes at a price: close handling means workers must gear up with gloves, masks, and sometimes full respirators. That doesn’t always happen outside well-funded facilities, especially in developing regions or schools with tight budgets.
Health and Environmental Concerns
As someone who has worked with dozens of laboratory reagents, I’ve learned that accidents rarely follow predictable scripts. Bottle labels fade. Fume hoods break. High turnover leads to new staff who haven’t seen the darker side of chemicals like this one. Breaching safety protocols isn’t always dramatic; sometimes it’s slipping off a glove to check your phone or working after hours without supervision.
Health risks aren’t just theoretical. Data from toxicological studies shows increased rates of skin problems and lung irritation in people who routinely handle these chemicals. Environmental data is murkier, but persistent organic pollutants have long histories of leaching into waterways and soil. Silver, in particular, harms aquatic life even at low concentrations.
What Can Actually Make a Difference?
Instead of handing out thick binders full of safety warnings, practical steps make the most difference. Regular hands-on training that treats new staff as partners in safety rather than just lecturing them. Automatic safety systems—like powder containment hoods and air monitors—flag trouble before symptoms set in. Companies must invest in safer alternatives where possible, or engineer controls that keep the chemical off workers’ hands and clothes in the first place.
As more labs and factories look to “green chemistry” and eco-friendly manufacturing, the conversation shouldn’t only focus on performance. The safest workplaces put health at the center, investing in both technology and training to move away from hazardous legacy chemicals wherever they can. Until safer substitutes exist, the real hazard is treating these risks as just another checkbox.
A Closer Look at the Role in Organic Transformations
Silver trifluoromethanesulfonate, often called silver triflate, catches the eye of any synthetic chemist looking for a versatile reagent. Plenty of chemists have stories about that one transformation that wouldn’t budge, no matter how long they tried. Bring out silver triflate, and suddenly the stubborn reaction yields the pure product. That says a lot about its clout.
Powering Glycosylation and C–C Bond Formation
Carbohydrate chemistry comes up regularly when talking about silver triflate. Ask anyone who’s wrestled with glycosylations—these reactions build the sugar linkages that make up natural products and biological markers. Silver triflate steps up as a gentle, yet reliable, Lewis acid that activates glycosyl donors, pushing otherwise sluggish reactions forward. The result? Cleaner, faster bonds between sugar units, essential for making vaccines and medicines.
It’s not just sugar work. Silver triflate sparks interest in carbon–carbon bond construction. In Friedel–Crafts acylation, for example, it shuffles the acyl group onto aromatic rings with a gentle touch. Compared to iron or aluminum salts, the silver approach leaves less mess, cuts down on side reactions, and helps scale up challenging syntheses.
Engine for Selectivity and Mild Reaction Environments
Chemists aren’t just chasing high yields—they aim for selectivity. Silver triflate’s main trick lies in activating substrates under mild conditions. Palladium-catalyzed cross-couplings rely on it to convert stubborn halides into reactive intermediates. The result is targeted bond construction, leaving background material untouched. Whether assembling pharmaceuticals or fine-tuning agrochemicals, that sort of control makes a difference.
In the lab, the reagent’s surprising stability adds peace of mind. While some silver salts darken and degrade at the hint of light, silver triflate often stays usable longer, so fewer headaches with repeat runs or storage nightmares. That encourages more innovative uses, like catalyzing rearrangements or supporting photoredox processes.
Addressing Drawbacks and Looking Toward Greener Chemistry
No reagent gets by without some baggage. I’ve seen budgets strained by the price tag on silver triflate, and nobody likes dealing with the waste streams it leaves behind. Disposal of silver-containing byproducts, if done carelessly, brings real environmental risks. Some labs have started recapturing silver or opting for catalytic rather than stoichiometric use, shrinking both costs and waste.
Green chemistry folks push for even safer, smaller-scale alternatives. Recycling silver after a batch run cuts down on environmental impact, while newer protocols suggest using silver triflate in lower quantities—sometimes just as a co-catalyst alongside more robust metals. Creative approaches like these point the way for sustainable practice, without losing the edge silver triflate brings in synthesis.
Supporting the Next Wave of Synthesis
Silver trifluoromethanesulfonate holds a spot in the toolbox of anyone working on complex molecules. The way it enables efficient, selective, and sometimes otherwise impossible reactions has earned its reputation. While the push for greener processes grows louder, silver triflate continues to adapt—with improved techniques, better recovery, and new hybrid methods. Its presence reminds chemists that pushing the boundaries in synthesis often means working smarter, not just harder.