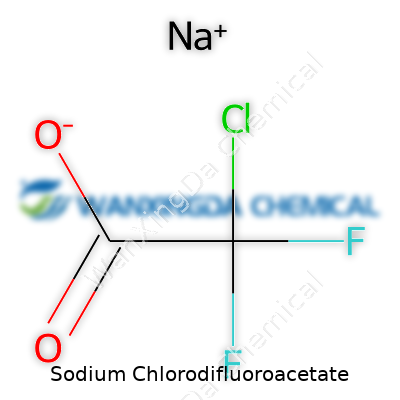
Sodium Chlorodifluoroacetate: Looking Past the Label
Historical Roots Meet Modern Chemistry
Every decade brings new faces to the chemical industry, but some compounds hold a quiet importance that makes them worth a second look. Sodium chlorodifluoroacetate, not a chemical famous outside specialized labs, threads its way through agricultural and pharmaceutical stories alike. It traces back to broader work on halogenated organic acids, a field that’s been rich with discoveries since the last century. People have been tweaking carbon chains with halogens for ages; in this case, fiddling with fluorine and chlorine has delivered a substance with its own quirks and strengths. Back in the day, the race to develop more potent herbicides and more flexible drug intermediates pushed this molecule out of the theoretical realm and into real-world use. Each step in its journey—part chemistry gamble, part engineering problem—shows how deep persistence shapes what ends up on tabletops and in research programs.
What Makes Up Sodium Chlorodifluoroacetate?
This chemical stands out in any bottle it gets poured into. It comes off as a white or off-white powder, easy to dissolve in water and giving off a faint whiff that hints at its halogen friends. It melts and breaks down in a tight window—engineers pay close attention to this, since it matters both for storage and for lab work. The salt’s reactivity opens the door for further chemical tinkering, serving as a bridge in constructing more complicated molecules. Teams drawn to its versatility see it handle well under standard lab protocols, proof that sometimes solid chemistry trumps fancier molecular gymnastics.
Putting the Spotlight on Use Cases
The compound finds a place mostly in the background, making lives easier for synthetic chemists and agronomists. In farming, sodium chlorodifluoroacetate forms a key ingredient for herbicides aimed at knocking out unwanted weeds without torching crops. Its knack for disrupting specific plant biochemical pathways means you don’t get the scorched-earth effect that older generation weedkillers delivered. In the drug world, scientists lean on its difluoroacetate backbone to build drugs step by step, slipping it into synthetic routes that benefit from the stability and reactivity it supplies. The fine balance it maintains between helpful reactivity and manageable hazard hasn’t gone unnoticed. For some, it is technology quietly doing heavy lifting instead of headline grabbing.
Digging Into How It’s Made
Most production stories start with a few basic ingredients and a lot of clever reaction management. Manufacturers often take monochloroacetic acid and swap in fluorine atoms—a process that turns up the pressure on quality controls. Along the way, sodium hydroxide helps create the salt out of the parent acid. The underlying methods trace back to classic halogenation chemistry, yet each scale-up brings in hard lessons about yield, purity, and waste. Industry veterans know that the devil lives in the details—careful temperature and timing determine not only cost, but the odds of nasty by-products surfacing. I’ve seen firsthand how a seemingly modest tweak in a reaction flask can double cleanup time, making this more than a matter of ticking off steps in a textbook.
Reactivity, Adaptation, and Tweaks
Chemical flexibility puts sodium chlorodifluoroacetate a step ahead in certain workflows. The difluoromethylene group packs a punch when grafted onto aromatic or aliphatic chains, introducing new properties farther down the line. Under strong acidic or basic conditions, the salt can decompose, shifting from lab-friendly to something you don’t want splashing on your bench. Savvy chemists exploit these pathways, cutting and patching the molecule into everything from pharmaceutical precursors to designer agrochemicals. Tuning reaction partners and conditions takes experimentation. Every new application gets pushed through a gauntlet of trial syntheses, and failures often teach more than the rare, immediate wins.
The Many Names on the Label
Not every researcher calls it sodium chlorodifluoroacetate. Synonym-hunting reveals terms like sodium difluoroacetate chloride or the simpler NaCF2CO2. Sometimes what’s on the drum depends on local nomenclature rules or regulatory quirks, complicating literature searches and regulatory filings. The core chemistry doesn’t change, but the paperwork chase can add real hurdles, especially for small operations. Clear records and communication save headaches, giving research and industry teams a shot at smooth collaboration and safer handling.
Standing Up to Safety Scrutiny
Working with any halogenated organic requires respect for both its properties and its limits. Sodium chlorodifluoroacetate can irritate eyes, skin, and airways, and strict personal protective equipment use clamps down on workplace incidents. Chemical spills, left unchecked, pose both acute hazards and environmental headaches—so spill kits and well-trained staff become a frontline defense, not just regulatory checkmarks. Proper labeling, locked storage, and up-to-date safety drills reduce the odds of careless accidents. In my own lab days, every new bottle brought a review session, and the habit stuck, because even the sharpest teams can slip when routines get dull. Establishing a culture of safety, and keeping standards high enough for repeat audits, means fewer emergencies and more focus on the research itself.
Research Frontiers and Applied Curiosity
Academic groups keep picking at the edges of sodium chlorodifluoroacetate chemistry, looking for routes to new drugs, crop protectants, and specialty chemicals. Each year sees tweaks to synthetic methods for greener, leaner, or higher-yield results. Some attention now falls on how best to recycle by-products, converting what used to get tossed into something useful. Green chemistry initiatives have nudged producers toward lower-emissions processes, not only for cost but for the mounting regulatory pressure facing halogen-flavored syntheses. This trickle-down from university benches to pilot plants gives a sense of interconnection across the industry pipeline. Even in larger companies, teams build on scientific papers they used to breeze past as students. The cycle refreshes with each cohort, keeping the field vibrant.
Questions Around Toxicity and Long-Term Impact
Nobody walks into halogen chemistry blind to potential risks. Toxicology studies hover over sodium chlorodifluoroacetate, especially around questions its breakdown products raise. Some early animal exposure work pointed to central nervous system effects and liver stress at higher doses. Translating those findings to complex real-world exposure isn’t straightforward; there’s always a margin between lab rats and daily handlers. Occupational guidelines have grown tighter over the years, and environmental chemistry teams run routine monitoring of wastewater streams—hoping to catch problems long before they hit wider ecosystems. I’ve seen the growing skepticism toward legacy compounds; a new generation of chemists brings sharper questions and more demanding standards, leaving little room for comfort with old data. This push toward transparency, backed by wider toxicological screens, marks a healthy change, even when it slows down commercial rollouts.
Looking Past the Present
What comes next rarely gets scripted in advance. Teams are shaping sodium chlorodifluoroacetate’s role, nudged by shifting regulatory frameworks and new technological demands. Advances in synthetic biology and green chemistry could push for alternatives, or prompt cleaner production processes for halogenated intermediates. The compound’s future links to the fate of related fluorochemicals now under tighter environmental scrutiny, especially as persistent organic pollutants draw more regulatory fire globally. I’ve talked shop with researchers interested in redesigning this chemical from the ground up—making it more biodegradable, less toxic, while keeping its unique chemistry alive. This blend of caution and creativity defines the next chapter. The drive behind sodium chlorodifluoroacetate’s survival isn’t just about protecting market share or legacy patents. It’s about solving new problems, meeting sharper expectations, and learning from past oversights. For all its technical detail, the true story of this compound carries a lesson in adaptation and responsibility, echoing across the wider chemical world.
Looking Past the Lab Bench
People who haven’t worked in agriculture or pest control probably haven’t heard of sodium chlorodifluoroacetate. The name alone sounds like a dare for your tongue. Stepping into my uncle’s storage shed as a teenager, I’d see drums and bags with long chemical names stacked behind the tractors, and sodium chlorodifluoroacetate wasn’t there by accident. Over time, I realized every odd-sounding compound had a story—sometimes good, sometimes worth a closer look.
What’s Driving Use in Agriculture?
Sodium chlorodifluoroacetate often finds a role as an active ingredient in certain herbicides and pesticides. Its reputation in controlling weeds and pests means higher crop yield and fewer losses on the family farm. There’s real urgency behind these chemicals. Farmers bank on consistent harvests, and a ruined field can wreck a year’s income. The same logic plays out from massive industrial farms to small patches of specialty produce.
Chemicals like this one help tip the odds in the farmer’s favor. But they’re not magic bullets. Pests get tougher. Insects and weeds can develop resistance, forcing growers to hunt for replacements or blend treatments. The big picture? Every time a new chemical gets pulled into use, someone weighs both the power of what it can do and the risk it brings.
Health and Safety: The Uncomfortable Truth
People in towns near large farms wonder what all these long-named substances do, besides protecting plants. Stories about farm workers getting sick, local waterways supporting fewer fish, or strange odors drifting over backyard fences can’t be shrugged off. Some research points at connections between synthetic agrochemicals and risks to both human health and local ecosystems. With sodium chlorodifluoroacetate, as with many new chemicals, the playbook isn’t always complete. Studies suggest that environments need careful monitoring to make plain any long-term risks.
Pushing for Smarter Solutions
Nobody likes a shouting match between “all chemicals out” and “science knows best.” Real-world fixes usually land in the messy middle. Tighter regulation, stronger guidance for safe handling, and investment in monitoring will help everyone. Farmers don’t want to poison the land their families live on. Regulators need up-to-date science that reflects how these substances really work, not just how they’re supposed to work in theory. Communities want transparency and a say if decisions could affect their drinking water or kids’ health.
Some progress is happening. Farmers try rotating crops, planting pest-resistant strains, and exploring biocontrol. The hope: each step reduces reliance on chemicals with complex names and histories. The journey winds through policies, research, old habits, and new discoveries. Reaching safer, smarter agriculture asks for everyone’s patience and willingness to listen—even when the conversation gets uncomfortable.
The Stakes for the Next Generation
Kids growing up near fields deserve clean water and safe food, just like their city cousins. A long name like sodium chlorodifluoroacetate shouldn’t stand in the way of asking tough questions or making sure families, land, and small towns don’t pay the price for someone else’s shortcut. The future isn’t written yet, and that gives everyone a chance to demand better answers and better options.
Understanding the Chemical and Its Context
Sodium chlorodifluoroacetate isn’t something you stumble across in everyday life. Most people probably haven’t even heard of it unless they work in labs or industries where specialty chemicals play a big part. It’s used as an intermediate in chemical synthesis, including the creation of certain pesticides. That means real people, with names and families, handle this substance in their daily jobs. This brings up the plain question: is it safe to work with?
Sifting Through the Known Risks
I’ve spent enough time in labs to know that safety isn’t just a label slapped on a bottle. Chemicals have personalities: some are downright friendly, while others deserve a wide berth. With sodium chlorodifluoroacetate, things get serious fast. This compound falls into the organofluorine group, known for unique properties but also for the potential to irritate skin, eyes, and respiratory systems. Evidence from studies in the field shows that inhalation can provoke symptoms such as cough, dizziness, or even respiratory distress if not treated with care. Direct skin or eye contact brings its own dangers and can result in burning or stinging.
No chemical works in isolation. Its safety depends on how folks in the field interact with it. PPE—personal protective equipment—isn’t a suggestion; it’s a must. Chemical-resistant gloves, goggles, and well-ventilated workspaces become the silent guardians for anyone handling this material. In my years of lab work, skipping a pair of gloves or forgetting a mask led to close calls, sometimes with far less aggressive substances. One wrong move can spark a chemical reaction or a health scare that lingers long after.
Health and Environmental Concerns
Sodium chlorodifluoroacetate doesn’t just pack a punch for people; the environment doesn't get off easy either. Like many fluorinated chemicals, breakdown in soil or water takes a long time and may generate byproducts with their own baggage. The Environmental Protection Agency keeps a watchful eye on substances with the potential to persist in the environment. If spilled, this chemical doesn’t just disappear, which raises the stakes for transport, storage, and cleanup operations.
Questions about safety aren’t just academic—they affect the way industries train employees and keep the workplace safe. Anybody supervising its use should ensure that workers get training on spill response and hazard recognition. I remember going through mandatory safety drills that felt tedious at first, but those drills proved vital when the unexpected happened. A minor spill might turn major if the cleanup team doesn’t know what they’re facing.
Looking for Safer Paths Forward
Regulation and common sense both demand strict oversight. Substituting less hazardous chemicals, where possible, makes sense. Organizations like the American Conference of Governmental Industrial Hygienists set exposure standards designed to protect workers. Compliance isn’t optional if long-term health matters to a company or community. Real progress comes from treating chemicals like sodium chlorodifluoroacetate with the respect and caution they require.
Everyone who deals with hazardous materials makes choices that ripple outward. Mistakes can lead to serious consequences, but deliberate care sets a standard for safety culture. It’s not a question of avoiding chemicals altogether, but about handling them smartly—with the training, infrastructure, and mindset to back it up. That’s how safety gets built, one conscious decision at a time.
There’s this chemical many folks in agriculture and pest management will run into—sodium chlorodifluoroacetate. It’s got a name that doesn’t roll off the tongue, but it does a job, especially as a herbicide. Every time I see substances like this move through a warehouse, my mind goes to the basics: how do you keep things safe for both people and the planet?
Real-World Storage—It’s About More Than Just a Container
People have a habit of thinking chemicals just need to stay out of reach. With sodium chlorodifluoroacetate, that’s only step one. This isn’t a sugar or table salt you can stash anywhere. Its molecular structure means that if moisture slips in, trouble follows fast. Even a tiny bit of water can kick off hydrolysis with toxic byproducts—gases like hydrogen fluoride have no place wafting around storage sheds.
From my time walking through farm co-ops and seed dealerships, I’ve learned you need walls that don’t sweat. Good storage uses closed barrels or containers lined with everything-proof inside, locked up tight. Some folks like steel drums lined with polyethylene, kept on pallets away from direct floor contact. I once saw heavy rains flood an old shed and (luckily) the barrels didn’t leak, but after that, nobody ever kept them on concrete without a buffer again.
Temperature and Light—Nature’s Role in Chemical Safety
Sun turns ordinary storage rooms into hotboxes. Sodium chlorodifluoroacetate starts breaking down fast above 30°C, and sunlight speeds it up. Most reliable setups sit in the shade, and climate matters. If you’re sweating just stepping inside, the chemical is too. Cooler, dry air matters—I’ve seen manufacturers recommend 15–25°C, though in the real world, you aim for stable over perfect. Heating and air conditioning are rare luxuries for most agricultural depots, but keeping windows covered or storing in the lowest, coolest floor helps. One winter, I saw a warehouse stacked too close to radiators, and folks had to toss several containers after the seals got brittle from repeated heating and cooling.
Segregation—Never Trust Chemistry to Luck
With storage, it’s about what sits nearby. I’m not a fan of stacking different chemicals together. Sodium chlorodifluoroacetate can react with acids and heavy metals, so warehouse rules ban any such neighbors. Once chemicals get stored together because it’s “just for a few days,” and then you get cross-contamination that takes hours to clean. Simple shelves, clear labels, and an eagle-eyed supervisor go a long way. I once caught a drum of sulfuric acid perched above herbicides—not once since has anyone doubted the value of dedicated spaces.
Concrete Steps for Safer Storage
Handling practices can always get better. Training matters. I’ve seen old pros catch problems a rookie would miss. Cataloging every shipment, labeling every drum, and coaching everyone walking the site on what to do if a spill happens—small details save big headaches. Clean-up kits should be easy to reach, and nobody should have to dig for a mask or gloves during an emergency. Local fire codes should set a high bar, and it’s worth aiming even higher. Storing sodium chlorodifluoroacetate safely means thinking ahead for storms, heat waves, and plain old human error.
Safe storage is not just a technical issue. It’s a daily practice and a sign that everyone in the supply chain respects both the work and the world outside.
Getting to the Chemistry: Why the Formula Matters
Most people probably won’t spend dinner conversation talking about sodium chlorodifluoroacetate, but the world of chemistry relies heavily on precise formulas. To pin it down simply, this compound carries the formula C2ClF2NaO2. For those of us using these names every day, that short string of letters and numbers provides a window into the way the compound behaves and how it works in various environments. Years at the lab bench taught me that understanding a compound’s formula doesn’t just help you pass an exam; it keeps you and your results safe, clean, and reliable.
Why Learn Chemical Formulas Anyway?
Clarity counts in science. You swap out just one atom and the whole story changes. Check sugar: glucose and fructose share the same atoms, but those atoms’ arrangement gives you entirely different results. Now, sodium chlorodifluoroacetate mixes sodium with a double shot of fluorine and a dash of chlorine, making it unique. Its makeup has drawn interest for applications in synthesis and sometimes even in specialty manufacturing. Having the right formula means researchers across the world speak the same language. The exact numbers—two carbons, one chlorine, two fluorines, one sodium, and two oxygens—give you confidence the discussion centers on the right substance.
Real-World Uses and Safety Concerns
No one in the field takes new compounds lightly. Fluorinated compounds can have surprising health effects. I remember a colleague who always wore heavy gloves and eye protection when moving substances with fluorine content around. Lab safety protocols highlight that even minor exposure sometimes triggers big issues if you miss a detail. The right formula provides a foundation for mapping out reaction paths and anticipating what might go right—or wrong—in the glassware. In research, these details matter, not for trivia but to avoid costly or dangerous mistakes.
Trust and Accuracy: Fighting Misinformation
Searching online can easily lead to confusion if claims aren’t backed by clear, correct facts. There's a lot of chemistry noise out there. Reliable information builds trust. Out-of-date or mistaken formulas wasted more of my time during early research than I care to admit. In the academic world, accuracy doesn’t just win prizes—it keeps reputations intact. By agreeing on formulas like C2ClF2NaO2, teams share work, compare results, and push their fields forward without redundancy or error. That’s something no one should take for granted, especially as information spreads faster than ever.
Tackling Education and Public Understanding
Chemistry needs more plain talk. Many kids leave school thinking the periodic table belongs to someone else. I’ve mentored students who only found excitement in science when the puzzles connected to real stories. Getting hands-on with formulas turns murky concepts into clear visions. By focusing on cases like sodium chlorodifluoroacetate and its distinctive combination of atoms, learners of any age see chemistry as practical, almost personal. This build-up of curiosity and skill can spur the next wave of innovation or maybe just help someone ace their test.
Charting the Way Forward
Teaching, learning, and working with chemical formulas will always matter in research, manufacturing, and medicine. Ensuring that the right information moves from scientist to scientist—without shortcuts or errors—protects both progress and safety. We owe it to ourselves and to the next generation of problem-solvers to provide clear answers, ask sharp questions, and keep building knowledge one formula at a time.
Facing Toxic Chemicals Head-On
Sodium chlorodifluoroacetate isn’t a name that rolls off the tongue. It’s one of those chemical compounds most people would skip right past if they spotted it on a safety sheet. Despite that, it’s got a reputation you can’t ignore—used in pesticides, with properties that can pose real threats to people, wildlife, and the environment.
Why Care About Disposal?
Disposal can turn dangerous in a hurry with chemicals like this. In high school, our chemistry teacher insisted: treat all unknowns like they’re hazardous. Years later, the wisdom stuck. Dumping hazardous waste in the regular trash or down the drain invites contamination of drinking water and soil. When stuff like sodium chlorodifluoroacetate enters rivers or municipal water systems, purification may not filter it out. Wildlife and people pay the price, often without knowing what went wrong until it’s too late.
Incineration: Not Just Burning Trash
Most hazardous chemicals can’t go into standard landfills or trash bins. Specialized incineration breaks down compounds at extremely high temperatures so harmful byproducts don’t survive. This process isn’t as simple as firing up a backyard burn barrel—it calls for a regulated, contained environment with scrubbing systems so that anything released from the stack is as clean as technology allows. Reliable incinerators follow laws set by environmental agencies to keep air, soil, and water safe from fluoride emissions and persistent toxins.
Rules Give Clarity—And For Good Reason
Laws and public safety standards around chemical disposal exist because, in the past, companies dumped dangerous substances with little oversight, leaving decades-long legacies of poisoned groundwater. Modern regulations zero in on risk points in the disposal chain, requiring careful labeling, restricted transport, and documented handoff from creator to final treatment facility. It feels like a burden for smaller labs or companies, but skipping those steps can come with steep fines and, worse, real public harm.
Responsibility Starts Before the Wastebin
An ounce of prevention saves trouble later. Many labs and agricultural operations switched to less toxic options as soon as alternatives showed up. Storing only as much sodium chlorodifluoroacetate as absolutely needed cuts back on disposal costs and risk. Training staff to spot expired or unused chemicals keeps the supply closet from turning into an accident-in-waiting.
Transport: Not for Amateurs
One of the most overlooked points is what happens when waste leaves the premises. Hazmat drivers licensed for dangerous goods operate under strict rules for packaging and transport. Tampering with those standards can put communities along the route at risk. Accidents on highways involving chemical spills show how fragile safety can be if protocols get ignored.
Pushing for Stronger Systems
Better disposal solutions come from a mix of updated tech, prevention, and enforcement. Government can help fund safer incinerators and tighter waste tracking. Companies that make and use these chemicals should invest in alternative formulas and regular staff training. Even people in the loop—warehouse workers, janitors, truck drivers—benefit from honest information and practice drills so no one is caught off guard.
The Road Ahead
The story of chemical disposal isn’t just about one compound, but about setting higher standards for safety and environmental respect. Each time a lab or farm disposes of sodium chlorodifluoroacetate correctly, it’s a win for everyone downstream—literally and figuratively. The real progress comes from never assuming the small stuff doesn’t matter.