Sodium Trifluoroacetate: Substance with a Growing Scientific Footprint
Historical Development
Looking back, sodium trifluoroacetate arrived on the scene as chemists searched for ways to pack more punch into their synthesis experiments. It didn’t take long for the compound to draw attention, especially in the world of organofluorine chemistry, where introducing trifluoromethyl groups can make or break a new molecule's design. Early literature reveals a steady rise in its use, particularly from the mid-20th century, as research labs worldwide started recognizing the benefits of this salt both for simple reactions and for more elaborate transformations. Leading scientists saw value in its stability and its knack for acting as a trifluoromethyl source long before companies started making larger batches for wider markets. The historical context really highlights the push-pull between a chemist’s desire for new tools and the realities of making those tools at scale with consistent quality, and sodium trifluoroacetate exemplifies that journey.
Product Overview
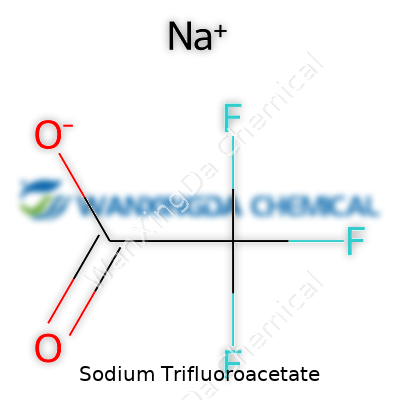
Sodium trifluoroacetate stands out as a white, crystalline powder that dissolves easily in water and many organic solvents. Its chemical formula, CF3COONa, might seem simple on paper, but anyone who's worked with it knows that simplicity brings its own kind of versatility. In my own work, as well as that of colleagues, the reliability of sodium trifluoroacetate in maintaining its integrity across different bench environments makes it a staple for researchers needing trifluoromethyl groups. You find it in research labs, quality control testing facilities, and even specialized manufacturing setups, often because its properties help chemists bypass steps that used to take a lot more effort, reagents, and time.
Physical & Chemical Properties
The salt presents as a solid with a fairly predictable melting point just north of 200°C. It remains stable in dry storage, but it pulls water from the air, which can trip up newcomers to the lab. That hygroscopic nature might seem like a nuisance, yet it’s a small trade-off when you consider how robust sodium trifluoroacetate stays under standard conditions. In solution, its ions split apart smoothly, delivering that sought-after CF3 group to reaction mixtures. This trifluoromethyl piece brings high electronegativity and low nucleophilicity, which means you can influence molecule behavior in subtle but powerful ways, whether you’re creating pharmaceuticals, agrochemicals, or specialty polymers.
Technical Specifications & Labeling
Bottles of sodium trifluoroacetate don’t hide any secrets if they’re labeled the right way. Researchers always check for purity above 98% before ordering, knowing contaminants can mess up critical syntheses. Product labels often list batch numbers, production dates, and specific storage instructions, which everyone learns to double-check if they value reproducibility in their results. Clear, up-to-date hazard statements about toxicity and recommended handling practices are not just legal boxes to tick—they keep those in the lab safe from unnecessary exposure or accidents. Based on personal experience, a missing label or ambiguous purity grade is enough to stall a project, especially where grant deadlines are on the line.
Preparation Method
Sodium trifluoroacetate usually comes from the neutralization of trifluoroacetic acid with sodium carbonate or sodium hydroxide. The reaction itself produces little fanfare—a simple acid-base neutralization—but the devil is in the details: temperature control, solvent selection, and careful work-up steps are what stop unwanted byproducts from sneaking into the final sample. Industrial synthesis methods tightened up these protocols to scale the process and keep impurities at bay. In laboratories like mine, preparing smaller batches can help if the store-bought material is out of stock, though purity control always becomes more challenging outside specialized plants.
Chemical Reactions & Modifications
As a precursor, sodium trifluoroacetate can deliver trifluoromethyl units in organic synthesis. That makes it popular for nucleophilic trifluoromethylation reactions and as a participant in metal-catalyzed transformations, such as copper- or silver-mediated couplings. It interacts neatly with a range of electrophiles and can support modifications that lead to pharmaceuticals with improved metabolic stability. Its high electronegativity means reactions are highly selective, which many medicinal chemists appreciate when tweaking the biological profile of drug candidates. At the same time, improper use may trigger unwanted side reactions, particularly with sensitive functional groups—something skilled chemists always keep in mind as they set up new experiments.
Synonyms & Product Names
Chemists often encounter sodium trifluoroacetate under alternative names like trifluoroacetic acid sodium salt or TFA sodium salt. Databases and suppliers stick to these designations, so confusion doesn’t take hold on the shelf or in the documentation. Expert users get used to seeing several names thrown around depending on whether they’re reading a research paper, browsing catalogues, or reviewing patents—staying vigilant about synonyms remains vital to avoid accidents or misinterpretation, especially when ordering chemicals for time-sensitive projects.
Safety & Operational Standards
Direct exposure to sodium trifluoroacetate can irritate skin, eyes, and mucous membranes, prepping any newcomer for the necessity of gloves, goggles, and good ventilation. Standard safety protocols—like using containment trays, ensuring waste ends up in the right disposal buckets, and having proper training on spill response—aren’t just “best practices” on a wall chart but habits that keep people productive and healthy. Over the years, the most responsible labs I visited ingrained safety briefings and hands-on demonstrations into their onboarding, so no one faced uncertainty about the risks. Common sense beats compliance lip service any day.
Application Area
Pharmaceutical research leans on sodium trifluoroacetate to build drugs with better metabolic performance, longer lifespans in the bloodstream, and tailored bioavailability. It features in the synthesis of agrochemicals that help farmers fight pests with fewer applications, reducing environmental load. Polymer scientists, too, rely on sodium trifluoroacetate to craft high-performance materials for coatings, electronics, or special membranes. Diving deeper, the compound even finds application in nuclear magnetic resonance (NMR) as an internal standard, boosting the accuracy of routine analyses. Whenever a trifluoromethyl group needs to stick firmly to a molecule, this salt steps up.
Research & Development
Academic and industrial labs treat sodium trifluoroacetate as a foundation for developing next-gen molecules. Researchers keep pushing to find greener ways to use or replace the salt, especially given growing interest in environmental sustainability. In my own group, we’ve seen rising focus on catalytic applications, where a small amount stretches farther and produces less waste. Scholarship from leading chemical journals chronicles new approaches for harnessing its reactivity while minimizing environmental footprint—showing how scientific curiosity and regulatory shifts motivate real technical leaps. Those who work close to application often push boundaries, using sodium trifluoroacetate in drug discovery or polymer innovation that couldn’t be reached with a blunter toolkit.
Toxicity Research
Sodium trifluoroacetate doesn't earn a reputation for acute toxicity at low doses, but carelessness piles up long-term risks. Studies, including ones I’ve reviewed closely, warn about possible environmental buildup due to persistence in water systems, a real concern as chemical runoff becomes a hot-button topic. Chronic exposure to high concentrations may bring risks of organ damage or disruption of normal metabolic processes, though reported cases among trained professionals following safety protocols remain rare. With watchdog groups and regulators shining a brighter light on possible ecological impacts, ongoing research probes how much accumulates in aquatic environments and whether standard disposal methods keep levels in check.
Future Prospects
Trifluoromethyl chemistry isn’t going out of style. Demand for sodium trifluoroacetate, especially in pharmaceutical and materials science, will likely grow as more applications surface. The drive toward sustainable chemistry urges researchers to scout for alternatives with lower environmental impact or to upgrade waste management around production and disposal. Efforts to improve biocatalytic or electrosynthetic routes could cut hazards and production costs simultaneously. In my experience, staying ahead on safety data and regulatory developments pays off. Scientists and engineers leading the field will shape best practices, making sodium trifluoroacetate a safer, greener cornerstone of modern synthesis long into the future.
Chemistry’s Hidden Hand
In most people’s minds, chemistry doesn’t get much fancier than vinegar and baking soda. Those working behind pharmaceutical innovation or crop protection have a different perspective. Sodium trifluoroacetate may not ring bells for most, but it plays a quiet supporting role as a building block in labs worldwide. Its real value shows up in the way it speeds up or guides chemical reactions that produce life-saving drugs or materials with unique properties.
Beyond the Obvious: Why the "Trifluoro" Part Matters
I remember chatting with a chemist who explained how the fluorine atoms in sodium trifluoroacetate make things possible that just can’t happen otherwise. Three fluorines change a molecule’s habits—boosting strength, stability, and even shifting how the body handles a medicine. That kind of fine-tuning would be tough, if not impossible, without a powerful ingredient like this. The compound carves out space in the crowded chemistry landscape for creative problem solving, from shifting reaction pathways to tweaking electronic properties.
Pharmaceuticals Count on Reliability
Sodium trifluoroacetate helps chemists stitch together tricky molecules that form cancer drugs, antiviral agents, and treatments for neurological diseases. Fluorinated compounds often resist breakdown inside the body longer than their non-fluorinated cousins, letting a drug stay active for just the right amount of time. Adding trifluoroacetate into a synthesis offers a shortcut, helping scientists hit the target faster and with fewer impurities. Precision saves both time and money, qualities any manufacturer and patient can appreciate.
Farming’s Silent Assistant
Agriculture deals with tough problems—pests adapt, weeds evolve, and the global population keeps growing. Sodium trifluoroacetate steps up as an ingredient in research for advanced herbicides and pesticides. Its unique chemistry helps create active ingredients that can survive sunlight or rain, or that break down just when the crop needs them to. For farmers fighting resistant weeds, these products offer another tool in protecting yields without hammering the land with higher chemical doses. The stakes are real: better chemistry means better food security.
A Growing List of Concerns
As much as innovation matters, questions about environment and safety can’t be ignored. Fluorinated molecules hold up astonishingly well in nature. They don’t break down easily, which sparks real worries about long-term buildup in water or soil and what that could mean for future generations. There’s more heat now on manufacturers to rethink how these compounds are made and used, balancing progress with respect for the environment. Scientists are already working on new steps to break down or recycle these chemicals and researching alternatives that offer similar benefits but with less baggage for communities and ecosystems.
Toward Smarter Use
In the end, sodium trifluoroacetate represents both a chemistry triumph and a responsibility. Relying so heavily on any one class of chemicals means the spotlight stays on safety, transparency, and ongoing research. Supporting more open data sharing allows different industries to spot problems early. Governments and industry groups pushing for stewardship can help speed up safer disposal methods or greener replacements. Smart innovation means paying attention not just to what works today, but to what will last for generations without causing regret.
The Realities of Handling Chemicals Like Sodium Trifluoroacetate
Sodium trifluoroacetate pops up in conversations among chemists, researchers, and those keeping an eye on what’s used in labs and the larger environment. Anyone who’s spent hours in research labs knows there’s always something new to learn about what we’re working with. The question about its safety isn’t just a matter for scientists; it affects communities, the environment, and workers who handle these compounds every day.
Digging Into Toxicity
I learned early not to fall for the idea that any unfamiliar chemical is automatically “more dangerous.” Still, evidence about sodium trifluoroacetate gives reason for concern. It contains three fluorine atoms, and that feature signals persistent, hard-to-break bonds. Compounds with strong carbon-fluorine links often linger in the environment. This stability might sound convenient for industrial use, but it works against us when these substances accumulate over time.
Research shows sodium trifluoroacetate breaks down into trifluoroacetic acid, which doesn’t just vanish into thin air. Instead, it resists natural degradation, contributing to its persistence in water and soil. Environmental scientists have flagged trifluoroacetic acid as a compound that doesn’t flush out easily. Over the years, this can result in accumulation in aquatic environments, which raises a real risk for plants and wildlife, not just people in lab coats.
Human and Environmental Impact
I’ve seen specialists handle a lot of mystery powders, but few treat sodium trifluoroacetate as harmless. Direct exposure may irritate the eyes, skin, or lungs, based on the material safety data from various chemical suppliers. There’s also the reality of what happens after disposal. Anything with strong fluorinated bonds can persist far beyond the expected laboratory shelf life. It filters into wastewater, and municipal plants often lack the tools to break it down completely.
Some scientists worry about bioaccumulation. Though not enough long-term studies have nailed down every risk, related fluorinated chemicals have shown up in animals and drinking water sources. The European Chemicals Agency and similar organizations worldwide have flagged the whole group of per- and polyfluorinated compounds for paying special attention to how persistent and tricky they can be in natural settings.
Potential Solutions: Managing the Risks
Safe practice starts with knowing what you’re dealing with. Relying on personal protective equipment in the lab isn’t enough. Good chemical hygiene means tracking waste, using fume hoods, and avoiding releases into the open environment. Communities can press for tighter controls and disposal guidelines at both local and industrial levels, especially since breakdown products like trifluoroacetic acid rarely just stay put once released.
Clear communication matters. I remember times when information about chemical hazards lagged behind actual risks. Regulators must step up and update safety recommendations as research comes in. Chemical manufacturers can support this by being transparent about toxicity data and environmental fate. People who work with these substances deserve the straight facts, not just a perfunctory hazard symbol on a label.
Final Thoughts
Meanings behind chemical names don’t stop at the laboratory door. Once chemicals leave the lab, they step into the wider world with all the consequences that brings. Sodium trifluoroacetate proves how a compound built for industrial strength can turn into a long-lasting problem. It pays to stay on top of the data, push for stronger safety measures, and treat such substances with the respect our shared environment demands.
What’s in a Chemical Formula?
Chemistry doesn’t work like cooking. There’s no pinch of this and a dash of that—every atom needs the right place on the dance floor. Take sodium trifluoroacetate, a name that rolls off the tongue easier than it rolls off the lab bench. Its formula, C2F3NaO2, might look like a secret code, but it carries a lot of meaning. It tells you the recipe: two carbons, three fluorines, one sodium, and two oxygens. These aren’t just numbers—they tell chemists exactly how to build the molecule and predict what it might do once it’s out in the wild.
Real-World Impact of Getting the Formula Right
My first memory of a screw-up with a formula came in university. Our lab group used sodium acetate instead of sodium trifluoroacetate for a project. At first glance, it felt like a minor swap. Chemistry showed us otherwise. Bubbles replaced crystals, and the reaction we needed turned into an odd mess. It wasn’t a total loss, though—every mistake in a lab is a learning moment. The formula wasn’t just a label on a bottle. It was a roadmap, and we’d taken the wrong turn.
The formula for sodium trifluoroacetate matters most in the hands of folks using it. Organic chemists count on that trifluoromethyl group to bring special properties—a real gamechanger in making pharmaceuticals. The fluorines tweak the molecule’s personality. Drugs built with the help of sodium trifluoroacetate tend to resist breakdown inside the body. That helps the medicine hang around long enough to do its work.
The Push for Responsible Practice
There’s another side to this. Specialty chemicals, especially those with fluorine, keep regulators awake at night. Fluorinated compounds don’t just disappear. If the chemistry community ignores the legacy of persistent pollutants, society foots the bill. Waste from a careless reaction travels downstream, literally and figuratively. The plain fact is that sodium trifluoroacetate isn’t a kitchen-chemistry ingredient. Responsible labs consider the full lifecycle—from raw material sourcing to what leaves the drain.
Learning and Solutions
Some answers start with education. A strong grasp of formulas acts as a filter against mistakes. The old shortcut of just grabbing chemicals based on similar names or appearances needs to end. Teaching students to check and double-check formulas creates better scientists and safer research spaces. Modern software does help by flagging mismatches between the intended reaction and the actual chemicals on hand. Still, no tool replaces proper training.
On a bigger scale, companies can invest in greener chemistry—developing processes that use less hazardous reagents or swap out persistent fluorinated compounds. There’s plenty of work ahead, but none gets done without knowing, to a tee, what sits behind each formula. Sodium trifluoroacetate’s formula serves as a reminder that every element matters, both in discovery and in responsibility.
Straight Talk About Safety
Chemicals like sodium trifluoroacetate don’t play around. I’ve spent enough time in labs to know that respect for storage rules isn’t about following paperwork—it’s about people’s lives. Science books can list features and give instructions, but anyone who’s ever smelled a surprise waft from a cracked vial knows things get real fast when rules get bent. Sodium trifluoroacetate, used for specialized synthesis and lab work, packs a chemical punch due to its fluorine content and reactivity, so dropping the ball on safe storage sets up unnecessary risk.
Seeing Hazards Before They Happen
Sodium trifluoroacetate offers stability as a powder or crystalline solid, but stable on paper doesn’t mean harmless in practice. Improper storage turns a routine stockroom into a hazard zone. Exposure to moisture, high heat, or acids can trigger release of toxic gases, including hydrogen fluoride—anyone who’s had a whiff of HF knows it burns more than just your nose; it can chew through flesh and bone.
I’ve seen too many colleagues get casual about storage and toss incompatible chemicals onto cluttered shelves, trusting labels and lids to do all the work. This is where danger creeps in. The stuff attracts water from the air, so leaving a cap loose or storing it where humidity fluctuates shortens its safe shelf life. Over time, caked lumps and degraded contents tell you the chemical’s changed, but if nobody checks, that warning gets missed.
Simple Storage Rules Everyone Can Remember
Some mistakes stick with you. My first lab job drilled in the basics: stash sodium trifluoroacetate in a cool, dry space, far from acids and anything holding moisture. I can still picture the heavy-duty plastic containers, desiccators packed with fresh drying beads, and airtight jar lids. Every scientist knows the panic of searching for a chemical and finding it in the wrong spot, or worse, with a container sweat-soaked or crusty from exposure.
Good lab practice isn’t about making things safe for their own sake; it means respecting your coworkers and keeping research on track instead of calling emergency services. Anyone stocking shelves should double-check for leaks, confirm the container’s integrity, and make certain nothing potentially reactive sits nearby. Temperature swings spell trouble. Basements prone to flooding, rooms near windows, or makeshift cabinets next to the autoclave are all bad news.
Building Safer Habits, Not Just Protocols
Some labs toss up generic signs and call that training. Real preparation starts with hands-on review—open a fresh container, feel that crisp click when the lid seals, and check the humidity indicators inside desiccators. Lab managers should schedule regular checks for broken seals, old samples, and expired stock. Encourage everyone to speak up if they spot something out of place; silence often leads to trouble.
It’s tempting to ignore “just one more bottle” left in a corner. In reality, one overlooked mishap can cause injury, research losses, or worse. Digital logs, clear labeling, and accountability make a world of difference in day-to-day organization. I’d rather be the person double-checking the shelf than the one explaining why the fire alarm went off.
The Bottom Line for Everyday Chemists
Sodium trifluoroacetate doesn’t need fancy solutions—just steady habits and respect for its potential. In my experience, staying attentive trumps fancy equipment. Dry, cool, separated storage keeps accidents at bay and builds a culture where safety is just part of how business gets done. Nobody should learn the hard way how messy shortcuts can get.
Why Careful Handling Matters
Sodium trifluoroacetate isn’t your average lab salt. Its unique mix of properties makes it useful in organic synthesis, but its chemical punch brings real hazards. I’ve spent plenty of hours in labs that handled nastier chemicals, and if there’s one thing you learn: safety is built into every small decision. The risks with sodium trifluoroacetate might not grab headlines, but nobody wants health headaches or an incident scene because of shortcuts.
Where Trouble Starts
The story starts with the powder itself. Sodium trifluoroacetate looks innocuous, but dust from the stuff can irritate eyes, airways, and skin. It dissolves in water, which makes spills a hassle if they go unchecked. Even though it doesn’t react explosively, combining it with acids in the wrong way can unleash toxic fumes — a rotten deal for anyone breathing them in.
Real-World Precautions
Every lab I’ve worked in has leaned hard on the basics: gloves, goggles, and lab coats. That’s not just busywork. Gloves keep your skin from contacting a compound that’s harsh and persistent. Fume hoods factor heavily into the workflow, since inhaling vapor or dust isn’t just uncomfortable — it can leave your lungs burning and your head spinning.
I’ve messed up a glove or two and felt the burn after picking up a bottle with residue. Even tiny skin contact left a lasting sting. That’s why wearing the right gloves, making sure they fit, and changing them at the first sign of damage became a habit fast. Good habits are the lifeline here.
Beating Complacency
A big risk comes from getting too comfortable. Rushing cleanup or treating this compound like table salt invites small accidents that roll into bigger health problems. Most people in research or industrial settings hear safety briefings often, but enforcement matters. A clear protocol for cleaning spills — like having neutralizing agents and disposable towels ready — can make stressful moments manageable and cut down the chance for mistakes. Training shouldn’t wait for someone to ask; it should feel as common as turning on a light before walking into a room.
No Place for Shortcuts
Waste tends to slip under the radar. Improper disposal can spark environmental headaches long after a job’s done. Putting sodium trifluoroacetate waste in the right container, with proper labeling, keeps hazards away from unsuspecting hands. I’ve known labs that ran into trouble because someone tossed the wrong powder into a communal bin. The cleanup wasn’t fun, and the fines hurt. Containerizing and tagging every time, every batch, is the smartest bet.
Solutions: Put Safety in Reach
Putting safety data where people actually use the chemical helps more than folders gathering dust. Safety training, glove stations, eye wash, and emergency showers within reach make a difference. Communication works wonders — stickers, bright signs, or a two-minute safety chat before work starts remind people not to treat powerful chemicals like afterthoughts.
No shortcut or overlooked step is worth the aftereffects: exposed staff, invisible injuries, or damage that’s tough to undo. Respect for chemistry is real-world wisdom that keeps everyone healthy, keeps labs working, and keeps the stories about sodium trifluoroacetate in the textbooks, not in the headlines.