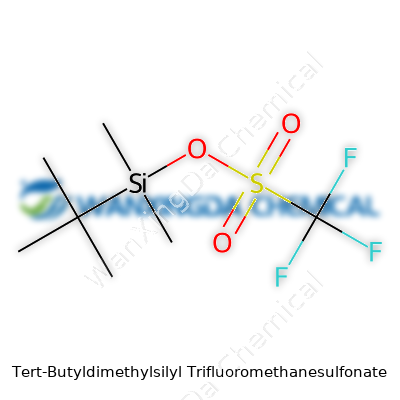
Tert-Butyldimethylsilyl Trifluoromethanesulfonate: A Closer Look at a Key Chemical Tool
Shaping Synthetic Chemistry: Historical Development
Years ago, chemists ran into the same problem again and again. Reactive groups like alcohols or amines would get in the way when building complex molecules, and blocking these groups reliably, then taking away the block when you want, became essential. This need shaped the search for protective groups. Through the late 20th century, research around silyl groups took off, thanks to their fine balance of stability and ease of removal. Out of this wave came Tert-Butyldimethylsilyl Trifluoromethanesulfonate—nicknamed TBSOTf—a reagent that made silyl protection quick, efficient, and less dependent on harsh conditions. Its ability to protect alcohols under mild situations, even with other reactive sites present, meant chemists could turn to it for solving messy synthesis challenges, whether in academic labs or industrial ventures.
What Sets TBSOTf Apart?
This compound stands out among silylating agents because it carries a bulky tert-butyl group next to the silicon. That sheer size gives it better selectivity—it attaches to exposed alcohols but leaves most other groups untouched. TBSOTf typically appears as a colorless or pale yellow liquid. The triflate group is what really powers up its reactivity, pushing the reaction forward at room temperature, and even working with less nucleophilic alcohols. It covers a gap left by the less reactive tert-butyldimethylsilyl chloride (TBSCl), doing the job in far less time and with fewer side-products. From practical experience, student and professional chemists consistently favor TBSOTf for reactions where time and yield matter, not just for ease of use but for the reliable results.
Chemical and Physical Traits that Matter
One lesson that every lab worker picks up quickly: a chemical’s quirks must be respected. TBSOTf boils at around 74–76°C under atmospheric pressure, but in air it’ll react with water, releasing toxic gases, and can even corrode glassware if left for too long. Its volatility means spilling a bottle can not only waste expensive reagent but may expose labmates to risks if ventilation fails. In solution, its triflate group draws electrons away, turning the silicon atom into a prime target for attack by alcohols and phenols. Good chemists take advantage of its high reactivity, fitting bulky silyl protection into delicate syntheses without disrupting other sensitive functions along the chain.
Getting Technical: Labeling and Standards
Labels serve as the first warning line. TBSOTf arrives clearly marked as highly corrosive and moisture sensitive. Packaging often uses PTFE-lined seals, since it chews through ordinary rubber and glass stoppers all too easily. Concentration standards vary among suppliers, yet bottles typically sit at high purity—no one wants their reaction sabotaged by trace acids or water. The chemical formula (C8H18F3O3SSi) and molecular weight usually get flagged for calculations. Recent years see increased insistence on GHS pictograms, with hazard codes stating risks of skin burns, severe eye damage, and environmental toxicity.
Reliable Preparation Methods and Real-life Trade-offs
Several routes have gained ground for making TBSOTf efficiently, but most still use the mix of tert-butyldimethylchlorosilane and silver triflate. This approach pulls out chloride as silver chloride, leaving TBSOTf behind in solution. The method works but can create waste that needs safe handling. Other ways involve swapping triflic acid for the silver salt, hoping to reduce cost and cut down on heavy metal waste. Both cases run into scale-up headaches. Anyone who’s worked behind the bench recognizes the smell and rushes to keep water out of the reaction. Even trace water can eat up yield in minutes. Research in greener syntheses continues, chasing less toxic by-products or recyclable solvents, yet current best-practices require tight control and strong environmental oversight.
Powerful Chemistry: Reactions and Next-Level Tweaks
TBSOTf’s main use lands squarely in silylation—putting on a tert-butyldimethylsilyl (TBS) group. It turns alcohol or phenol groups into silyl ethers, often in the presence of bases like imidazole or 2,6-lutidine, which soak up the acid generated. Carbohydrate chemists favor it for protecting diols differently, handing them selective handles during multi-step syntheses. In organometallic work, the reagent sometimes triggers rearrangements or installs basic silicon functionality that isn’t easily available by other routes. Sometimes, chemists even exploit its reactivity to introduce silicon into aromatic compounds, setting up for further transformations. In rare cases, the strong electrophilicity enables silylations in water, a remarkable feat for such a water-sensitive chemical.
Synonyms and Product Names in Real Practice
Names vary between catalogs and colleagues. Tert-butyldimethylsilyl triflate, TBS-triflate, or TBSOTf all designate the same bottle—and misunderstanding leads to mixups, especially across multinational collaborations. The synonyms pop up in purchasing lists, scientific papers, and lab manuals. Proper labeling and communication across research teams prevent costly errors, especially where different silylating agents might reside alongside each other. In practice, everyone just says “TBS triflate” because speed and clarity matter as much as precision during busy synthesis campaigns.
Staying Safe: Lab Realities and Strict Regulations
Standard lab safety training hits home with TBSOTf. Splashes lead to deep burns, both from the reagent’s strong acidity and its knack for breaking down moisture, generating corrosive triflic acid and siloxanes. Ventilated hoods, face shields, and gloves are non-negotiable—one careless move writes its own cautionary tale. Transport and disposal demand special procedures because the chemical classifies as hazardous both to people and the environment. Waste neutralization calls for controlled hydrolysis in ice-cold water under exhaust, and even then, disposal of siloxane-laden residues presses for updated guidelines. In my own experience, instructors hammer on the point: treat every vial as though it could turn deadly on contact, not just in theory but with scars to prove it.
Where TBSOTf Leaves Its Mark: Fields and Uses
The real beauty of TBSOTf comes from its role in targeted synthesis of drugs, natural products, and lab-scale building blocks. It graces complex assembly lines for antiviral or anticancer compounds and drives many routes toward oligosaccharides. Carbohydrate and nucleoside chemists rely on its selectivity for challenging protection strategies. Medicinal chemistry, where rapid analog synthesis is key to finding new medicines, leans on TBSOTf because fewer side reactions mean fewer purification steps. Process chemists in industry depend on its reliability to scale compounds for clinical trials, though the price and hazards keep them watching for alternatives. Organic electronics, increasingly, looks to silicon-containing motifs that start with silylation steps.
R&D: The Push for Improvement
Chemists around the world keep testing what this reagent can do, knowing well there’s room to stretch. Enzyme mimics and peptide chemistry explore if TBSOTf can introduce silyl groups to amino acids without damaging backbone structures. Green chemistry circles aim to cut waste, trying out continuous-flow setups that trap the hazardous waste as it forms. On the teaching side, the discussion often turns to using milder silylation options in undergraduate labs, not because TBSOTf lacks value but because its mishandling costs more than lost product—it can teach the wrong lesson about risk. Collaboration between industry and academia focuses on finding replacements with less toxic by-products or easier recycling, but so far, silicon’s flexibility keeps TBSOTf a frequent pick.
Measuring Risk: Toxicity and Research Findings
TBSOTf does harm through multiple routes—touch, inhalation, sometimes by delayed reaction after accidental exposure. Animal testing and industrial accidents both show potential for respiratory distress, skin corrosion, and organ damage under heavy exposure. Chronic toxicity data lags behind its more common cousin silyl chlorides, but the aggressive acid formation during hydrolysis means the reagent rarely makes it past short-term exposure studies. Many universities restrict its use to trained researchers for this reason, and newer platforms monitor airborne levels even in closed reactions. EHS (Environment, Health & Safety) staff pay special attention to the chemical’s presence on lab inventories.
Next Steps: What’s on the Horizon?
Innovation doesn’t wait, even for well-established tools like TBSOTf. Academic groups test less toxic triflate surrogates and silicon-based protection strategies that deliver the same performance without using precious metals or producing persistent by-products. Startup companies chase greener chemistry, engineering flow reactors that minimize exposure and automate waste neutralization in real time. There’s also a push in medicinal chemistry to skip protection-deprotection cycles entirely, either by inventing robust catalysts or by crafting molecules that can survive more direct reactions. Yet until these advances become trusted at scale, TBSOTf stays not just in the textbooks but on the shelves, because its unique combination of performance, selectivity, and speed fits the real-life needs of modern synthesis.
Shielding the Right Pieces in a Reaction
Organic chemistry looks complicated on paper, but some tricks show up over and over in labs because they solve real problems. Tert-Butyldimethylsilyl trifluoromethanesulfonate, usually called TBSOTf, fits into this group. The name might be enough to scare off anyone outside the field, but it earns its stripes by making certain reactions smoother and more predictable.
TBSOTf steps in when chemists want to protect a part of a molecule from reacting. Sometimes groups like alcohols react too soon or too easily. If those sites get involved at the wrong stage, everything after goes wrong. By using TBSOTf, you can cover or “protect” those problem spots. What you end up with is a silyl ether, which stands tough through many harsh chemical steps. This temporary protection gives chemists the freedom to work on other sections of the molecule until it’s time to take the cover off again.
Saving Time, Preventing Waste
My earliest experiments with TBSOTf made me appreciate how much frustration it can save. Making a complicated molecule involves dozens of steps. A slip-up can ruin days of work. TBSOTf acts fast and works under conditions that don’t destroy delicate molecules. Some older silylating agents call for harsh bases or longer times. TBSOTf gets the job done in minutes, even with sensitive compounds. Less waste, fewer do-overs, more reliable chemistry.
Simplicity for Complex Synthesis
Modern drug development and natural product synthesis demand handling lots of functional groups. Each of those groups can react or fall apart under different conditions. Getting a new drug candidate ready for testing takes clever planning. TBSOTf gives chemists a tool that simplifies planning. Even in research outside pharmaceuticals, such as polymer chemistry or material science, TBSOTf finds a spot in the toolbox. It streamlines routes to complex targets where other protecting groups or milder silylation agents would fall short.
Environmental Hazards and Safer Alternatives
This chemical, like many lab staples, draws attention for potential hazards. TBSOTf reacts violently with water and releases toxic fumes if mishandled. Labs use fume hoods, gloves, and goggles, but things can go wrong. Waste handling needs care—nobody wants triflate and silyl waste in the water supply. I remember a near miss in our undergraduate lab, reinforcing that lab safety isn’t just for show.
Across the industry, there’s a push for greener chemistry. Some suppliers offer less toxic, less aggressive silylating agents, but nothing matches TBSOTf’s reliability and speed for tough jobs. Regulatory agencies and chemists both need to keep an eye on use patterns and waste treatment. Improvements have come through better solvent systems, smaller scale-ups, and more automated waste neutralization.
The Next Steps in Chemistry’s Toolkit
Real progress in chemical science often happens by solving practical problems. TBSOTf might not draw headlines outside specialist circles, but its role in chemical synthesis can’t be missed. It helps scientists dodge pitfalls, waste less material, and open new routes to complex molecules—tangible, daily wins that drive research forward. Responsible use and disposal practices keep the benefits flowing without piling up hazards for others. For anyone setting out to make tough molecules, TBSOTf stays a trusted tool, one that’s earned its spot through service, not promise.
Unpacking the Challenge
In any research lab, someone has to think about what happens to all those odd bottles on the shelf. Tert-Butyldimethylsilyl trifluoromethanesulfonate, or TBSOTf for anyone who’s tried squeezing that full name into a notebook margin, belongs in a class of chemicals that can transform organic synthesis—when handled right. This isn't a household name, but a lot of molecule builders recognize it as a favorite for protecting groups during synthesis. Simple storage mistakes can lead to ruined reactions or real danger. That’s not just a nuisance; it wastes time and puts people at risk.
What’s Really At Stake
Anyone who has handled TBSOTf knows about its knack for reacting with water and releasing corrosive fumes. Cap not tight? There goes your reagent—and maybe more. Inexperience mixes poorly with this triflate, since even one careless shelf could turn a valuable investment into a sticky hazard. Back in my grad school days, I watched a colleague pop open a bottle that hadn’t seen a desiccator in months. Hiss, white fumes, and a mad scramble for the fume hood. The cleanup took far longer than a simple walk to the right cabinet would have.
Smart Storage Isn’t Fancy, It’s Just Fundamental
Lab safety posters can spill rules all day, but experience drives the point home. TBSOTf likes moisture about as much as your phone likes saltwater. Direct sunlight speeds up decay, so bottles left by a window don’t last long. Glass with a solid PTFE-lined cap beats plastic every time—leaks never end well in a chemical fridge. Most researchers I know trust a desiccator and a fridge or freezer for any silyl triflate that costs more than lunch. Saving a few steps today rarely pays off tomorrow if the reagent fails on the bench.
The People Side of Storage
Proper labeling, clear dating, and keeping that bottle upright and away from acids—these details keep both people and reactions healthy. Newcomers often underestimate how quickly a bottle can go south. I’ve seen journals report yields that no one can repeat, simply because a bottle suffered a few weeks tucked away by the wrong person. Respect for shelf life and sharp-eyed inventory controls aren’t bureaucracy; they’re what let everyone downstream trust the reaction happens as the paper describes. Those who’ve taught in busy labs know that mixing up the storage for silylating agents causes headaches that can ripple through a whole research group.
Moving Toward Smarter Labs
Better storage practices don’t require new technology so much as stubborn consistency. Introducing regular checks, avoiding shared refrigerators for moisture-sensitive compounds, and keeping emergency cleanup materials within arm’s reach can all prevent mishaps. Some labs invest in digital inventory systems to track reagent age, but even a shared spreadsheet shared with regular inspections pays off. Manufacturers recommend refrigeration, desiccation, and tamper-proof containers for a reason, and following those cues aligns with the highest safety and research standards.
Beyond the Bottle
This isn’t just about one chemical; it’s about raising the baseline in every lab. Routine, not roulette, helps research reach reproducibility. Long-term success, both for safety and for synthesis, rests on building habits that respect chemistry’s quirks. TBSOTf proves that a little storage wisdom goes a long way—toward safer chemists and more reliable results.
Every chemist who has spent time in a lab learns to respect certain reagents. Tert-Butyldimethylsilyl trifluoromethanesulfonate (TBSOTf) belongs in that club. A bottle of this colorless, almost innocuous-looking liquid hides a level of reactivity that can catch even seasoned researchers off-guard. Handling it safely means thinking through each step—long before opening the cap.
The Risks in the Bottle
TBSOTf is a powerful silylating agent often used for protecting groups in organic synthesis. It reacts quickly with water, acids, and bases, producing trifluoromethanesulfonic acid as a byproduct—something known for its corrosive power. On skin, it leaves burns. In the eyes, it does lasting damage. The fumes are no joke either; they can burn deep in the lungs and irritate mucous membranes. I remember a graduate student who had to pause a project for weeks after an accidental spill left him with a blistered hand—so gloves and good habits matter.
Personal Precautions: Beyond the Basics
PPE stands as the first defense. Gloves like nitrile or butyl rubber do the job, since latex tends to degrade with strong reagents. Eye protection isn't just a box to tick —the tiniest splash can threaten your vision, so goggles with side shields matter just as much as gloves. A fitted lab coat closes the circuit. Some labs add a face shield for extra safety, particularly when there’s risk of splashing; it might look clumsy, but after some close calls, it no longer feels excessive.
The Role of the Fume Hood
Pouring or measuring TBSOTf should only happen in a chemical fume hood. This isn’t just about comfort; it’s about survival. Inhalation of fumes might not feel dangerous at first, but chronic exposure brings risks that keep showing up in occupational health studies. Keeping work inside the hood also lowers the odds of a stray splash causing harm—not only to you, but to anyone nearby.
Managing Spills and Disposals the Smart Way
TBSOTf does not play nice with water or alcohol, which means buckets of water or damp towels are dangerous choices for spill cleanups. On the rare occasion I’ve seen a small spill, absorbent pads rated for strong acids and bases paired with a generous application of sodium bicarbonate or calcium carbonate helped neutralize the mess before disposal in a labeled hazardous waste container. Regular staff training, including practice sessions with mock reagents, makes a world of difference when speed and accuracy matter most.
Why Vigilance Never Gets Old
It’s easy to let routine beat caution over the course of a long research project. Complacency grows quietly as deadlines pile up. That’s when accidents sneak in—a misplaced pipette tip, a shortcut with safety gear, or a rushed transfer. Institutional support plays a role, too. Frequent, accessible safety refreshers give teams the tools to protect each other and themselves. Notices on hoods, live review sessions, and good, clear incident reporting help embed a culture that treats every hazardous reagent with respect, regardless of how many times it’s handled.
TBSOTf isn’t unique in demanding caution, but its dangers sit close to the surface. Science needs those stories from the lab, not just to list dos and don’ts, but to remind us why practice and preparation turn a risky reagent into a safe tool. The right approach keeps the work moving, hands unburned, and eyes on the next discovery.
Getting to Know TBSOTf and Its Impact in Labs
Ask folks who work daily with synthetic chemistry what chemicals pop up most when protecting alcohols, and Tert-Butyldimethylsilyl Trifluoromethanesulfonate stands out right away. Its chemical formula, C8H18F3O3SSi, may look intimidating at first glance, but for chemists, the significance is pretty clear. This reagent sits right at the intersection where organic electronics, pharmaceutical labs, and university research often meet.
A big reason for its popularity comes not from hype, but straightforward practicality: TBSOTf, as folks call it in shorthand, reacts swiftly with alcohols and phenols to introduce a TBS group—a silyl ether—making sensitive molecules tougher against moisture or unwanted reactions during later steps. That stability can make or break a multi-step synthesis. This isn’t about trends; it’s about getting reliable results and avoiding failed experiments that waste both time and costly resources.
Why the Exact Formula Matters
Look beyond the letters and numbers in C8H18F3O3SSi; each part of this molecule plays out on the lab bench. The tert-butyl and dimethylsilyl pieces confer steric bulk, making the resulting silyl ethers more resistant to acid and base than their trimethyl counterparts. The trifluoromethanesulfonate group delivers high reactivity. It’s not just a mouthful from the periodic table; this combination helps push reactions forward even where other silylating agents would fail. This matters when tweaking yields or running reactions on delicate fragments where every milligram counts.
Labs around the world handle hundreds of synthetic challenges that can’t be solved by off-the-shelf chemical solutions. Having trustworthy access to reagents like this one is foundational for chemists pushing the boundaries in fields from new drug leads to energetic materials. Cutting corners with the formula—trading purity, say, or using an analog without understanding the differences—sets a project back in a hurry.
Beyond the Formula: Navigating Real-World Challenges
Working with TBSOTf isn’t just signing off an order form. This reagent often comes as a clear, moisture-sensitive liquid that really needs careful storage and handling. A single careless moment—leaving a bottle exposed to humid air—can ruin an entire batch, leaving that precious formula as little more than contaminated waste. Chemists learn this lesson early: don’t take shortcuts with handling or you’ll face setbacks more often than not. The push for good practices, clear labeling, and robust training arises not from bureaucracy, but from lived experience.
Another piece of the puzzle involves cost and sustainability. The reagents in this class don’t come cheap, and the triflate group raises questions about environmental impact. Chemistry teams can address this by scaling reactions precisely, recycling solvents wherever possible, and considering less hazardous alternatives for large-scale work. Advances in green chemistry have started offering more tools for this, but for many specific transformations, TBSOTf still carries the day.
Solutions: Integration and Responsibility
Keeping safe and efficient use front and center involves not just knowing the chemical formula, but investing in ongoing education and community sharing of best practices. I’ve worked in labs where one scientist’s small procedural tweak saved days of work down the hall. That sort of community knowledge, paired with clear-eyed attention to reagent quality, storage, and disposal, keeps this powerful tool both accessible and aligned with health and environment.
Tert-Butyldimethylsilyl Trifluoromethanesulfonate, by its formula, reminds us of chemistry’s complexity and its impact on fields that shape medicine, materials, and technology. By respecting both the details and the broader context, researchers keep the focus where it belongs: reliable science and responsible innovation.
How a Tricky Reagent Improves Real Chemistry
Every organic chemist hits that point where sensitive functional groups throw a wrench into otherwise clever reactions. Alcohols and amines, with their eager hydrogen atoms, step in and ruin the best-laid synthetic plans. I remember countless late nights trying to keep phenolic groups from reacting with every electrophile in sight. In nearly every research building, chemists know protecting these groups makes a world of difference.
That’s where tert-butyldimethylsilyl trifluoromethanesulfonate—TBSOTf for short—steps in. The name might twist tongues, but its job is simple: put a jacket on those unruly alcohols and amines, letting a reaction focus on the parts you actually want to change. TBSOTf builds a protective silyl ether around a hydroxyl group, making it less reactive for a while, until you’re ready to take it off.
Why Use TBSOTf Over Old School Silylation Reagents?
Old-school chemists used reagents like TBSCl, but these can be sluggish, need strong bases, and end up giving more byproducts. TBSOTf turns this step from an ordeal into a five-minute pipetting job. Add it to your alcohol in the presence of a base, and out comes the silyl ether, even when your substrate drags its heels or has stubborn neighbors. I’ve watched reactions go from hours to minutes, with fewer side products and better yields.
This isn’t just about saving time. With TBSOTf, you get protection for acid-sensitive sugars, enols, and other finicky groups that might not survive a more basic environment. The only caveat: you need to treat it with respect. Water ruins TBSOTf; it practically hisses if a drop hits your reaction flask. Gloves, dry tubes, and the patience not to rush—I’ve learned these aren’t negotiable.
Lessons from Lab Benches: Real Use Makes for Better Science
Over the years, TBSOTf has become the unsung hero in my toolkit for multi-step syntheses. Take peptide chemistry. You want to protect a serine hydroxyl without knocking your amide bonds off balance. TBSOTf wraps it up, lets you build the rest of the molecule, and then a touch of acid or fluoride brings the original group right back. The same holds for natural product synthesis, where one misstep can waste months of work.
Risks, Missteps, and Smarter Solutions
Like with any strong reagent, careless handling leads to waste, failed experiments, or worse, injuries. More than once, I’ve seen a distracted grad student watch a whole batch fizzle because humidity crept into the glovebox. Manufacturers could help by improving packaging—smaller, more manageable ampoules limit air contact and keep more people safe. Better training, smarter lab sharing, and clear protocols reduce wasted chemicals and protect junior researchers.
As science pushes deeper into complex, lifesaving molecules, we need tools that pull their weight. TBSOTf offers tried-and-true reliability for chemists who respect its quirks. Through experience, TBSOTf taught me that the right protection can make all the difference between a failed plan and a finished compound on the desk.