The Story of Trichlorotrifluoroethane: Past, Present, and What Comes Next
Looking Back: How Trichlorotrifluoroethane Rose to Fame
Trichlorotrifluoroethane, known to many as CFC-113 or by names like Freon 113 and Genklene, played a huge role in transforming how industries approached cleaning and refrigeration in the last century. Decades ago, factories and labs worldwide relied on CFC chemicals because they seemed to strike the right balance: strong at dissolving oils and greases, yet gentle enough to avoid corroding metal, plastics, and sensitive electronics. The smooth, non-flammable behavior meant less risk in the workplace, and industries loved how easily it evaporated without leaving residues. Researchers and engineers in the postwar boom embraced such chemicals out of necessity, rarely giving thought to downstream effects like ozone depletion simply because the science wasn’t there yet or seemed too abstract compared to immediate production needs.
Understanding the Product: Properties and Inside Knowledge
Trichlorotrifluoroethane sits in the family of chlorofluorocarbons. In practical terms, you’re looking at a clear, colorless liquid with a mild, slightly sweet odor. It’s heavier than air, doesn’t catch fire, and boils at a relatively low temperature just above room level. Chemical stability was its ticket to widespread use, holding up under storage and repeated cycling through cleaning or refrigeration systems. For a period, this characteristic made it a backbone compound in specialized sprays, vapor degreasing equipment, and as a precision cleaning fluid in electronics—especially when alternatives like trichloroethylene posed greater dangers to both workers and finished products. Regulatory labeling began highlighting the importance of proper ventilation, eye and skin protection, and storage away from heat long before environmental regulations came down harder in the 1980s and 1990s.
Preparation, Reactions, and the Broader Chemical Picture
Chemists typically built trichlorotrifluoroethane by halogenating ethane, carefully substituting hydrogen atoms with chlorine and fluorine under strict conditions. In the lab, small changes in temperature or pressure could force the process off track and yield contaminants, so there’s always been a premium on tight process control. On the reaction front, this compound resists breaking down except in the presence of ionizing radiation or at very high temperatures, a detail that, at first, seemed like an advantage but grew into one of its biggest public health weaknesses. It doesn’t react much in day-to-day use, which helped keep machines clean, but this very inertness is what let it drift upward, survive for years, and eventually wreak havoc on the planet’s ozone shield.
Names Through the Years: What We Still Call It
Most chemists, engineers, and safety professionals know this substance by its various names depending on context—CFC-113, Freon 113, 1,1,2-Trichloro-1,2,2-trifluoroethane, along with Ucon 113, and several less common trade labels. In my own experience, confusion over synonyms pops up all the time, especially in older equipment manuals or international standards. Sorting out whether a cleaning agent from the 1970s contains this specific CFC or a different blend matters for both compliance and safety audits. That need for clarity has only grown as rules governing use and disposal have tightened.
Using Trichlorotrifluoroethane: Then and Now
This compound once found a home in airplane factories, electronics assembly rooms, and research labs, stripping away oils and solder fluxes that older cleaning methods left behind. At one point, I worked alongside technicians using vapor degreasers that ran almost daily on CFC-113. They liked its efficiency, but even they noticed the headache and nausea after extended exposure in poorly ventilated shops. Those experiences mirrored findings in published toxicity research, where symptoms tracked closely with repeated exposure. As studies built momentum linking CFCs to ozone loss and health risks, industries moved, sometimes reluctantly, toward alternatives and much tighter occupational exposure standards.
Safety, Operations, and Hard Lessons Learned
Plenty of folks in maintenance or quality control can recall the shift in thinking following the pushback against CFCs. Safety standards moved beyond simple exposure limits to comprehensive controls, such as local exhaust systems, sealed solvent baths, and regular air monitoring. Training adapted to recognize early signs of overexposure—dizziness, irritation, even irregular cardiac response. Equipment designs improved to reduce leaks, recover solvent, and protect workers during filter replacements or drum changes. Some companies paid a high price for learning this the hard way, and those stories echo in every serious contemporary training session where legacy CFC equipment remains on site.
Research, Toxicity, and the Road Forward
Detailed animal studies and case reports in humans outlined a risk profile not just for workplace exposure but for persistent contamination. Researchers discovered that low concentrations linger in the air long after visible vapors dissipated, posing risks down the line. Evidence around bioaccumulation remains slim, yet the global discovery that CFC-113 rises into the stratosphere and breaks down ozone molecules turned this from an occupational hazard into a planetary emergency. This hard-won knowledge transformed research priorities, launching quests for safe drop-in substitutes and new ways to recycle or destroy stockpiles. Development has yielded replacements that sidestep the specific environmental disasters of CFC-113, but none has offered the same blend of performance and safety at all levels, leading to ongoing work and debate in both industry circles and regulatory agencies.
Future Prospects: Lessons for the Next Chapter
Most developed nations now regulate trichlorotrifluoroethane tightly if not outright ban it, but its legacy lingers in countless older systems still in service, warehouses with barrels untouched for years, and environmental footprints that last beyond the careers of the people who made or used it. Industry and researchers still face the task of safely decommissioning these stocks, remediating contaminated sites, and ensuring training for anyone who might stumble across a forgotten stash. Innovators in chemical manufacturing and environmental science continue searching for effective, sustainable solvents that match the utility of CFC-113 without its destructive side effects. The lesson from CFC-113’s rise and fall isn’t just about one molecule. It’s about weighing immediate utility against long-term consequences and listening to new research with open minds. Every breakthrough or shortcut in modern manufacturing and research rides on the shoulders of what people once believed would be safe forever. As regulations evolve and old habits finally disappear, the era of trichlorotrifluoroethane serves as a reminder—a big technological leap doesn’t mean future generations won’t have to clean up after it.
What Are We Really Using Here?
Trichlorotrifluoroethane's name doesn’t exactly roll off the tongue, but you’ll probably know it better by its trade names or as a member of the chlorofluorocarbon (CFC) family. In my years covering science and public health, I’ve come to realize few people truly know what goes into industrial cleaners or older refrigeration systems. Yet this compound played a real part in shaping modern manufacturing and electronics.
Circuit Boards and Clean Rooms
Growing up around relatives who worked with electronics, I got an early look at the lengths folks would go to just to keep sensitive equipment clean. Trichlorotrifluoroethane, often called by its technical shorthand CFC-113, turns out to be a go-to solvent for degreasing delicate electronic parts. Its properties make it useful for rinsing off soldering residues on circuit boards, where a stray drop of oil can ruin an expensive piece of tech.
Industrial cleaning runs on effectiveness, but sometimes also on what’s least likely to damage the product. Trichlorotrifluoroethane doesn’t leave a residue, works quickly, and doesn’t produce a ton of heat when it evaporates. That means people cleaning circuit boards, precision gyroscopes, or even high-end optical lenses have counted on it for decades.
Chilling Out: Refrigeration and Beyond
Many older refrigerators and air conditioners once used blends that included trichlorotrifluoroethane for heat transfer. Its stability was seen as an asset. In practice, units with this chemical ran for years without breaking down, since it rarely reacted with metal parts or rubber seals inside the system. It also helped keep large military and aerospace systems cool, especially before safer or less damaging alternatives became available.
In industry, paints and coatings need cleaning tools that don’t create their own problems. I’ve seen mechanical shops use trichlorotrifluoroethane for surface prep and degreasing metal before painting or sealing, because it dries fast and doesn’t corrode the surface. Cleaners need something tough to cut through the gunk on heavy machinery. For a long time, this chemical filled that role.
Regulations and Rethinking Use
Over the last few decades, science uncovered that CFCs, including trichlorotrifluoroethane, damage our ozone layer. Multiple reports from NASA and the EPA have painted a clear picture here. Every time these chemicals escaped into the atmosphere, they contributed to thinning ozone and opening up more risk from ultraviolet light.
Countries moved quickly thanks to the Montreal Protocol, which pushed for a major phase-out of CFCs worldwide. Refrigeration, cleaning and solvent industries all pivoted away from trichlorotrifluoroethane and similar compounds, trying out substitutes with fewer environmental risks. Safer solvents like hydrofluorocarbons or newer, oxygen-safe mixtures now dominate where CFC-113 once ruled.
Looking for a Better Way
People in labs and manufacturing floors are still looking for replacements offering the same cleaning power without trade-offs for health and the environment. Some of the newer fluorinated solvents show promise, though they rarely match every feature of the old compounds. Regulations keep tightening, so the pressure stays on for scientists and engineers to innovate.
We’ve all benefited from precision cleaning in modern life, whether buying a smartphone or flying on a plane. The lesson here comes down to balancing technical advancement with environmental humility. Today’s replacements demand constant scrutiny — what’s clean today shouldn’t leave a mess for tomorrow.
Looking at Safety Beyond the Label
Trichlorotrifluoroethane sounds more at home in a laboratory notebook than in regular conversation. Most folks remember it as a solvent, or sometimes it comes up in discussions about old air conditioners and cleaning solutions. Back in the day, industries turned to this chemical for its reliability. It could clean up electronic components, degrease machinery, and run in refrigeration units. On the surface, handling it looked simple—gloves, a mask, and good ventilation seemed enough. But there’s more to this story than what you’d read on a label.
Unpacking the Health Hazards
Breathing in the vapors of trichlorotrifluoroethane doesn’t cause a strong irritation the way some solvents do. That makes it easy to underestimate. Through the years, I’ve met technicians who worked in workshops or electronics plants who didn’t think twice about an open drum. Some would develop headaches, light-headedness, or coughs by the end of their shifts. The risk doesn’t always appear plainly, but chronic exposure stacks up. The US Centers for Disease Control and Prevention flag this chemical as a potential hazard for the heart, liver, and nervous system. Exposure sometimes leads to deadly arrhythmias, especially when someone is already sensitive or working in tight quarters. Skin contact dries you out and sometimes leaves a painful rash. These aren’t far-fetched scenarios—they show up in occupational health records.
The Environmental Toll
This chemical doesn’t only affect those handling it. Trichlorotrifluoroethane belongs to the family of chlorofluorocarbons, or CFCs. For a while, people focused mainly on the environmental blowback. CFCs punch holes in the ozone layer, which increases the risk of skin cancer and harms crops and fisheries by letting more ultraviolet radiation reach the ground. Recognizing the problem, international agreements like the Montreal Protocol drove most countries to phase it out from everyday use. Anyone who still keeps a stash of this solvent—maybe in an old cleaning kit or refrigeration system—now faces regulatory pushback about disposal and storage. Leaks don’t just break the law. They damage a fragile layer that protects us all.
Accepting What Responsibility Looks Like
There are some chemicals people can handle without worry, but trichlorotrifluoroethane doesn’t belong with them. Instead of downplaying its risks, workplaces started training employees about the dangers and how to use personal protective gear properly. In the electronics field, swapping to safer solvents takes effort, but the evidence speaks for itself. By moving away from this old standby, companies have cut health complaints and helped protect the environment. The transition isn’t always smooth, especially for smaller shops worried about cost. Groups like OSHA and NIOSH provide guidance and practical manuals for workplaces making the switch, and some states offer grants to help small businesses upgrade to safer cleaners.
Moving Forward with Lessons Learned
Handling trichlorotrifluoroethane doesn’t just demand a set of gloves and a fume hood. It asks for long-term thinking and a willingness to look for better alternatives. From seeing friends struggle with solvent headaches to working on community campaigns that explained ozone dangers, I came to respect the weight of these choices. We protect each other by respecting these chemicals, by investing in training, and by switching to safer options where possible. What’s at stake isn’t written in warning labels alone—it shows up in long-term health, in public trust, and in the air all of us breathe. The push to retire trichlorotrifluoroethane marks a step toward a kind of stewardship that reaches beyond the lab and into everyday life.
Understanding the Real Reach of Trichlorotrifluoroethane
Trichlorotrifluoroethane, often known by its industrial shorthand as CFC-113, has played a backstage role in electronics cleaning and refrigeration for decades. The technical language thrown around can make it seem distant from daily life, but its impact lands a lot closer to home than most realize. Every time someone talks about ozone holes or climate change, chemicals like this sit right in the conversation. From the 1970s into the early 1990s, this compound could be found in everything from cleaning solvents to refrigerant canisters. Nobody really thought much of it back then. People trusted in the science and the companies that provided these products.
What Really Happens: Ozone and Beyond
The story changed once atmospheric scientists connected the dots. Trichlorotrifluoroethane, part of the larger chlorofluorocarbon family, doesn't break down easily. It floats up and lingers in the upper atmosphere, releasing chlorine atoms under ultraviolet light. Just a few of those chlorine atoms can destroy thousands of ozone molecules. For decades, this chemical ate away at a shield that keeps harmful ultraviolet rays from reaching Earth's surface. The science became undeniable. Higher UV levels lead to increased skin cancer risks, eye problems, and crop damage. The impact doesn't just stay up in the atmosphere either because those rays touch everything, from natural ecosystems to city parks and front yards. Studies from NASA and international agencies paint a clear picture: regions under thinner ozone see stronger UV and higher associated health risks. I remember seeing faded signs and sun-bleached plants in places where the ozone took a real hit—it was a clear message: some changes can’t be ignored for long.
Climate Change: The Hidden Danger
People often zero in on greenhouse gases like carbon dioxide and methane, but Trichlorotrifluoroethane also packs a punch as a heat-trapper. These molecules, once released, stay in the atmosphere for years. According to the Intergovernmental Panel on Climate Change, the global warming potential of CFC-113 is thousands of times higher than that of carbon dioxide. That means every stray ounce adds up, contributing to the rising temperatures I see in my own city—hotter summers, more unpredictable weather, and a ticking clock on familiar ways of living. It's not some abstract chemistry lesson; it's about real people contending with real changes right outside their windows.
Cleanup and Solutions
Regulations like the Montreal Protocol mark a turning point. Countries came together to limit production, driving a gradual drop in atmospheric concentrations. Scientists started tracking these molecules with precision. Manufacturers phased out most CFCs, and replacements came in that break down faster and don't harm ozone in the same way. Progress hasn't been perfect, though. Stockpiles and old equipment still leak small amounts of Trichlorotrifluoroethane into the air. The lesson here runs deep: action matters, but so does vigilance. Governments need to keep old stock under wraps, and industries should make sure replacements are truly safer.
Personal responsibility plays a role too. For anyone working with old machines or electronics, safe disposal cuts off another source of leaks. Every step, from international agreements to local recycling, matters in repairing the scars left by CFCs. The story of Trichlorotrifluoroethane reminds me that small changes—both good and bad—echo much louder than anyone expects.
Keeping Communities and Workers Out of Harm's Way
Trichlorotrifluoroethane sounds like something you'd never run into outside a chemistry lab, but for decades, it showed up in places as unassuming as an office’s air conditioner or packing plant’s degreasing pit. Because of its convenience, many businesses relied on it, not fully considering its impact on the planet or on human health. My early days working with vintage electronics taught me the importance of handling these substances with a kind of respect that goes beyond ticking off boxy regulations.
Why Storage Isn’t Just a Box-Ticking Exercise
Trichlorotrifluoroethane can leak invisible gases with a strong environmental punch. These vapors sneak out more easily than most people realize. Back in the day, I’d notice old canisters stashed under a workbench, sweating in the summer sun. It struck me that storing chemicals like this isn’t just about clearing legal hurdles — it’s about preventing leaks that slip into the air or groundwater.
For businesses or schools still holding these old chemicals, I always recommend keeping them in sealed, original containers, away from sunlight and any possible heat source. Ventilated, locked cabinets shield workers from unexpected exposure, and they keep children or unsuspecting coworkers from stumbling onto trouble. The right storage spot makes all the difference. Just labeling a drum isn’t enough. It’s worth going the extra mile to check containers regularly for bulges or rust. If a single canister bursts, cleanup quickly evolves from a small hassle to a massive headache.
Disposal: No Place for Good Intentions Alone
Disposing of trichlorotrifluoroethane brings real ethical responsibility. Some folks figure they can just dump “old chemicals” down the drain, but this shortcut can poison water supplies and add to ozone layer damage. I once saw an entire community up in arms after contaminated runoff from a scrapyard reached a municipal pond. It reminded everyone — including myself — that ignorance doesn’t excuse inaction.
Professional hazardous waste facilities offer the only course worth trusting. Licensed handlers use high-heat incineration with tightly controlled emissions to break trichlorotrifluoroethane down without pumping toxins into the sky. Calling the local waste management board or environmental agency will connect most people with the right contractors. While hauling away outdated cans might cost a little, it doesn’t compare to the cost of cleanup once the stuff gets loose.
Habits and Policies That Actually Work
A lot rests on simple habits. Anyone storing these chemicals should keep detailed records and make regular checks part of their week. Bringing in outside experts to inspect old stockpiles removes guesswork. Schools and businesses need ongoing training for new staff, not just a one-time lecture. This ensures everyone understands the importance of careful storage and safe disposal.
Vigilance matters just as much as science, especially in small operations where people wear many hats and safety routines can slip. More government incentives or rebates to offset disposal costs would help, giving folks fewer reasons to stash these chemicals “for later.” Responsible handling always pays back by preventing accidents that can damage much more than one’s bottom line.
Every container, handled right, is one less danger for our homes, workplaces, and the air we all share.Perspective on a Persistent Compound
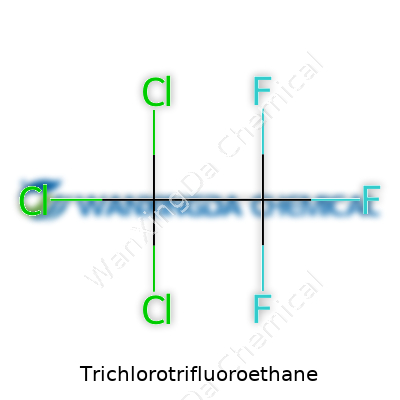
Trichlorotrifluoroethane brings back memories of old science classrooms and stories about ozone warnings from the late 1980s. Its chemical formula—C2Cl3F3, known as CFC-113—marks it out as a member of the chlorofluorocarbon family, notorious for their impact on the upper atmosphere. This compound caught attention not just for what it could clean or dissolve, but for its stubbornness in the environment.
People used it in electronics and dry cleaning because trichlorotrifluoroethane didn’t burn easily and didn’t react with much. You could pour it on a greasy circuit board, and the oil would vanish, leaving the silvery metal behind. This non-flammability stemmed from the powerful bonds between the carbon, chlorine, and fluorine atoms. The molecule itself holds together tightly; few things break through those halogen-guarded walls. This meant safety for workers, but it also spelled trouble once the chemical left the ground and drifted upward.
Stability: Double-Edged Sword
The real story with trichlorotrifluoroethane lies in its chemical stability. The bond strength between the carbon atoms and their attached chlorine and fluorine allows the compound to float through the environment almost unchanged. Sunlight in the lower atmosphere does next to nothing. Trichlorotrifluoroethane lingers in the air for decades, and this gave industries a tool that didn't corrode equipment or cause sudden accidents. It cleaned, it transported heat in refrigeration, and it kept havoc at bay inside factories.
The same stability that made these applications shine soon became the downfall. Once these molecules rose into the stratosphere, ultraviolet light had just enough energy to split the chlorine away. That single atom wandered off, breaking apart ozone and setting off a chain reaction. Researchers noticed the ozone layer thinning over Antarctica, and years of evidence pointed back at CFCs like trichlorotrifluoroethane.
A Legacy of Responsibility
Handling trichlorotrifluoroethane now comes with an environmental burden. Regulations like the Montreal Protocol phased out its use, sending engineers back to the drawing board. Factories and labs pivoted to less persistent chemicals—hydrofluorocarbons, for example—because these alternatives don’t last as long in the atmosphere or harm ozone to the same degree.
Facts remain: trichlorotrifluoroethane is a dense liquid at room temperature, it evaporates easily, and it resists breaking apart below the ozone layer. It won’t ignite, so fires are rare, and it won’t turn yellow or fizz when mixed into standard lab solvents. People trusted it for years because it seemed harmless enough. Only after decades did the chemical’s endurance prove to be its biggest threat.
Charting a Smarter Path Forward
If there's something to learn from the history of trichlorotrifluoroethane, it’s a reminder to track where chemicals travel outside the lab and what they do beyond their first use. The responsibility doesn't stop after a product leaves the factory floor. Modern chemists look for substances that do the job, break down when their work’s done, and don’t cause long-haul problems for communities or the world at large.
Smart regulation, careful study, and open discussion between companies, researchers, and the public all helped move the world past trichlorotrifluoroethane. Progress depends on putting chemical insights to work for a safer, more balanced future. That means being honest about the trade-offs between performance and persistence—and willing to change course in light of new evidence.