The Role and Realities of Triethylsilyl Trifluoromethanesulfonate in Modern Chemistry
A Glance Back: Historical Development
Working in the lab over the years, you start to realize that certain reagents come and go, but a handful tend to stick around for their reliability and flexibility. Triethylsilyl trifluoromethanesulfonate, often called TESOTf by people who have spent the better part of their days scrambling for that last ampoule in the fridge, traces its roots to the relentless pursuit for stronger, more reliable silylating agents. Chemists in the 1970s wanted to replace the bulkier, less reactive trialkylsilyl reagents when on the hunt for selective protection and activation. The explosion of organosilicon chemistry in that era set the stage for molecules like TESOTf, which managed to combine serious potency with manageable stability — not a given at that time, as anyone who's worked with wilder silicon-based reagents will admit. The improved accessibility of fluorinated sulfonate groups unlocked by the rapid growth in industrial fluorination marked a turning point that cemented TESOTf as a staple for synthetic organic chemistry.
Product Overview: Reliable Workhorse
Having reached for TESOTf in more than a handful of total syntheses and scale-up campaigns, it’s clear why this reagent finds its way onto so many benches. It brings a rare blend of high reactivity and selectivity when silylating alcohols, phenols, and amines. Unlike its cousins, such as trimethylsilyl chloride, TESOTf skips the typical game of repeated additions and laborious purification steps. Scribes of synthetic pathways love it for the clean transformations observed, particularly when protecting sensitive hydroxyl groups during multi-step procedures or enabling advanced glycosylation chemistry. You can see its fingerprints on countless MedChem journals each month, and its adoption continues to spread as labs value both time savings and reaction predictability.
Physical and Chemical Properties Matter
Good lab practice means getting up close and personal with each reagent’s quirks — believe me, one splash of TESOTf’s colorless, volatile liquid will leave a lasting impression. With a boiling point hovering around 50 to 55°C under reduced pressure and the unmistakable, acrid odor typical of triflate derivatives, you come to respect its volatility and aggressive reactivity. At room temperature, you face a highly flammable, moisture-sensitive liquid. Even minor environmental exposure can lead to decomposition, so the bottle never lingers uncapped. The molecule owes much of its punch to the electron-withdrawing triflate group bound to a silyl center, magnifying the electrophilic character and priming it for rapid attack on nucleophilic partners.
Technical Specifications & Labeling
Suppliers ship TESOTf sealed under inert atmosphere, usually in glass ampoules or PTFE-lined containers to lock out moisture. Genuine material arrives as a water-clear liquid, free of cloudiness or yellowing. Purity needs to meet the bar for advanced synthesis: levels upwards of 97-99% are standard. Sourcing from reputable vendors keeps unknown byproducts out of your reactions — an essential factor for scale-up or medicinal chemistry teams shooting for regulatory compliance. Safety labeling doesn’t just check the boxes – it speaks plain: handle with gloves, eye protection, work in a fume hood, and keep it away from every trace of water or exposed skin.
Personal Take on Preparation Methods
Learning to prepare organosilicon reagents, you gain an appreciation for the careful choreography that goes into TESOTf. Typical methods rely on the reaction between triethylsilyl chloride and trifluoromethanesulfonic acid or its silver salt. This route tends to minimize water in the reaction, which is crucial because any rogue H2O will burn you with hydrolysis or, worse, short-circuit the whole synthesis. Choice of solvent and low reaction temperatures help curtail side products. Some labs scale from small quantities using glassware, but industrial outfits shift to PTFE vessels and automation, aiming for both yield and reproducibility.
Chemical Reactions & Modifications
Cracking open the bottle of TESOTf, you know you’re in for brisk, predictable conversion of alcohols and secondary amines to their corresponding triethylsilyl derivatives. Its place on the list of go-to protection reagents comes down to sheer rate and compatibility. You toss it in, and reactions sing: fewer equivalents, often milder conditions, better site-selectivity, and less fuss downstream. In particular, protecting anomeric centers on sugars, selective silylation of polyhydroxylated steroids, and prepping building blocks in nucleoside chemistry all run smoother with TESOTf in the toolkit. Aside from classic protection, creative chemists engineer cross-couplings and SuFEx (sulfur–fluoride exchange) chemistry using the unique reactivity of the triflate group, nudging TESOTf into new synthetic territory every year.
Synonyms & Product Names: Speaking the Language
Chemists trade in shorthand, and the terror of transcribing long chemical names fades with reliable abbreviations. TESOTf stands for triethylsilyl trifluoromethanesulfonate, though you occasionally see abbreviations such as TESOTf or simply “triethylsilyl triflate” in literature. Synonymous terms pop up mainly in patents or different catalogues, but the core identity rarely gets confused, thanks in large part to its distinctive triflate moiety. The acronym game in chemistry doesn’t always clear things up, so it pays to check CAS registry numbers or supplier descriptions to avoid a painful mix-up.
Safety & Operational Standards: Real-World Practice
Lab veterans stress the importance of prepping a workspace before ever unscrewing a vial of TESOTf. The stuff eats lab gloves right off your hands, even the thick nitrile ones. Spills fume up sharply, stinging your eyes and throat. Direct skin contact risks deep chemical burns. Comprehensive fume hood use, goggles, and suitable gloves are non-negotiable. Spills need immediate cleanup with inert absorbent, and contaminated glassware has to be quenched with damp, basic solutions out of harm’s way. Waste disposal follows the hazardous organofluorine protocols down to the letter. I’ve seen more than a handful of seasoned chemists left with nasty rashes for moving too quick or ignoring that last moist patch on a spatula.
Application Area: From MedChem to Advanced Materials
You flip through any organic chemistry or medicinal chemistry journal, and the legacy of TESOTf jumps off the pages. It is widely used for selective silylation in complex molecule synthesis, particularly in the clever protection of hydroxy groups during carbohydrate assembly, nucleoside analog development, and peptide modifications. Pharmaceutical chemists value its high reactivity and low tendency for overreaction, allowing for shorter, cleaner process routes. In material science, TESOTf enables surface modifications, offering routes to fluoro-organosilicon coatings with unique wettability and dielectric properties. Custom organosilicon compounds show up in everything from OLED displays to new-generation hydrophobic barriers, with TESOTf providing a straightforward silylation strategy that often saves time and raw materials.
Research and Development: Pushing Boundaries
Even after decades in circulation, TESOTf does not fade into laboratory background. Chemists continually probe for new uses beyond routine protection. Driverless optimization for late-stage functionalization and flow chemistry platforms draw on its high reactivity paired with manageable selectivity. The trend towards sustainable chemistry pushes researchers to explore greener preparation routes, seeking to reduce the use of chlorinated solvents, hazardous byproducts, and reliance on rare metals. TESOTf’s unique capabilites as both a leaving group and a silylation agent invite innovations in selective cross-couplings and bioorthogonal labeling, opening fresh directions for molecular editing in medicinal and supramolecular chemistry.
Toxicity Research: Respecting the Risks
Health hazards from TESOTf can seem abstract until you watch hydrolysis spike a local acid burn, or see a friend cough uncontrollably after a cap goes missing. Toxicity mainly arises from the release of triflic acid and the parent reagents’ capacity for deep tissue injury, particularly if inhaled or splashed. Chronic exposure data remain limited, though case reports suggest caution with repeated low-level inhalation. Research explores anti-inflammatory responses after accidental skin or eye exposure in animal models, comparing the outcomes against other silylating agents. Engineers and chemists alike advocate heavier-duty fume extraction and direct monitoring of airborne triflate-related species, particularly in crowded academic labs and scale-up environments.
Future Prospects: Charting the Next Decade
TESOTf’s footprint in synthetic chemistry shows no sign of shrinking. Emerging fields such as green chemistry and flow manufacturing continue to place this molecule at the top of the toolkit for rapid, localized silylation with minimal waste. There is growing demand for scalable silylating agents that avoid rare metals or environmentally taxing precursors, which will likely trigger more sustainable, energy-efficient processes for TESOTf production. Possible next moves involve engineered derivatives that blend triflate utility with tunable selectivity or enable entirely new reaction frameworks. Digital platforms for reaction planning and process safety drive safer, more predictable scaling across pharmaceutical and semiconductor production lines. Keeping one eye on new regulatory environments — particularly concerning perfluorinated waste control — will force chemists to balance the advantages of TESOTf with the obligation to keep both bench and planet safe for the next generation.
Practical Chemistry Behind the Scenes
Triethylsilyl trifluoromethanesulfonate doesn’t roll off the tongue, but it plays a helpful role in a chemist’s toolkit. In the lab, people often call it TESOTf. Folks outside synthetic chemistry may have never heard of it. Yet behind projects that make medicines, new materials, and some specialty plastics, this chemical quietly puts in the work.
The Real Use Case: Protecting Groups
Most of the time, chemists use TESOTf as a silylating agent. The point is to protect alcohol groups during chemical reactions—basically putting a shield over a fragile part of a molecule. This allows scientists to focus on reacting other pieces without messing up the sensitive bit. Anyone who’s spent time making new molecules can tell you: mistakes here mean wasted days or weeks. TESOTf helps avoid those headaches.
Supporting Drug Discovery and Research
Having reliable protecting groups matters most in drug discovery. People working on new medicines need ways to build complicated molecules in repeatable steps. TESOTf helps by adding a triethylsilyl group, which chemists can later remove under controlled conditions. A recent review published in the journal Chemical Reviews highlights how methods involving TESOTf have improved the efficiency of making antiviral and anticancer compounds. That means researchers can move a promising molecule from the lab bench to animal studies without getting tripped up by tricky side reactions.
Tough Reactions Demand Tough Tools
TESOTf has some muscle compared to simpler silylating agents like TMSCl (trimethylsilyl chloride). It often works well in challenging conditions, especially when moisture or other functional groups create problems. In my experience, if a student’s reaction wasn’t working and they switched over to TESOTf, results often improved. This kind of robustness doesn't just save time—it keeps projects moving forward.
Handling the Downsides
Like many things in chemistry, handling TESOTf comes with risks. It reacts strongly with water, so spills or sloppy technique can cause real problems. As someone who’s seen what happens when a careless rush turns a benchtop sticky and ruined, I can say the safety training isn’t just a formality. Fume hoods, gloves, and methodical habits matter. Inexperienced chemists sometimes underestimate this, but being careful isn’t optional.
Environmental and Cost Issues
On the environmental side, chemists need to think about the solvents and waste generated, too. Many labs rely on dichloromethane or other chlorinated solvents with TESOTf—not exactly eco-friendly choices. The industry is pushing toward greener options, but progress goes slow. Price can be another barrier. For small operations or universities with tight budgets, TESOTf’s higher cost compared to standard silylating agents means it's not the default. Projects often reserve it for cases where other methods fail.
Moving Forward
Chemists keep looking for safer, greener ways to protect alcohol groups, but TESOTf holds strong where milder agents fall short. Sharing data, refining protocols, and pushing suppliers to explore more sustainable options will help. Progress never comes overnight in chemistry, but every step counts—especially for those working toward better medicines or materials.
No Room for Carelessness in Handling Reactive Chemicals
Triethylsilyl trifluoromethanesulfonate stands out as a tool for chemists, making stubborn molecules easier to work with. It’s got a reputation for being an effective silylating agent in synthesis. The flip side? This compound can be dangerous. Many who’ve worked in organic chemistry labs know how fast things can go sideways with reagents like this one. After years in the lab, I’ve learned from mentors and from my own close calls—the rules for handling chemicals like these aren’t just recommendations written to please regulators.
Staring Down the Hazards
Triethylsilyl trifluoromethanesulfonate reacts aggressively with water, splitting apart and releasing toxic and corrosive fumes. The first lesson is simple: if you care about your health and your benchmates, don’t let moisture near it. A well-sealed bottle, never left open to air, and routine checks of the integrity of containers are part of daily life for those working with this substance. Skip those steps, and you’re asking for trouble, not just for yourself, but for everyone sharing the space.
Practical Protection Never Gets Old
In my experience, people often treat protective gear like a formality—until that first splash, or the time the glove fails at a bad moment. Proper gloves—nitrile ones that actually resist powerful organics—goggles that fit snugly, and a clean lab coat end up mattering a lot. Steph, a colleague of mine, swore by thin latex gloves up until a reagent burned through in seconds. Ever since, she treats her gloves as a real line of defense. For this silylating agent, disposable gloves and eye protection aren’t negotiable. Think about your skin and eyes, not the cost or convenience.
Respecting the Fume Hood
No seasoned chemist preps this reagent out in the open. Fume hoods offer more than just insurance against bad smells. They stop noxious fumes from drifting into communal breathing spaces. Stomach-turning headaches and stinging eyes have taught most lab workers that a functioning hood makes a huge difference, not just to comfort, but to lung health over the long haul. Try to take shortcuts, and the whole lab pays the price.
Storage Woes and Smart Solutions
Pick the wrong shelf—or a cracked cap—and the lab becomes dangerous. Storage for these chemicals needs a plan. Keeping it cold, dry, and away from anything with water or alcohol stands as one of the few sure ways to avoid a real mess. My own lab’s policy puts desiccated storage above all. We make sure to label every container clearly, and we never reuse scoops or spatulas unless we know they’re bone-dry. Mixing up containers or skipping labeling spells disaster surprisingly fast.
Training and Emergency Prep Make the Difference
Experience tells me that training is good insurance. New faces in the lab sometimes shrug at the briefing, but watching a colleague scramble after a spill cures most nonchalance. We practice spill response and eyewash use for a reason. Knowing what to do—down to which chemical bucket gets which accident—turns would-be disasters into manageable hiccups. Regular reviews and refreshers keep safety from slipping into the background.
Investing in Long-Term Safety
Every risky chemical comes with a choice: rush through the prep and hope, or slow down and keep the work safe. Responsible labs set the right tone by making safety routines non-negotiable, not just for compliance, but out of respect for everyone present. Bigger picture, manufacturers and safety groups have a role here—by updating training material, urging clear labeling, and investing in better protective gear, they help reduce accidents at the source. Chemistry shouldn’t just be about discovery, but about sending everyone home healthy at the end of the day.
I remember the first day I handled something as reactive as Triethylsilyl Trifluoromethanesulfonate back in my early days at the university lab. The chemist in charge didn’t just toss me a datasheet — he walked me through the do’s and don'ts like my life depended on it. This isn’t some over-the-counter solvent you leave in a plastic bottle under the fluorescent lights. It’s a whole different animal, and safe storage is where most mistakes show up.
Moisture Turns It Ugly Fast
Triethylsilyl Trifluoromethanesulfonate, often called TESOTf, runs into trouble with water — not just splashes, even airborne humidity. You give this stuff access to moisture and pretty soon you've got decomposition, nasty byproducts, and possible pressure inside the bottle. I’ve watched an ignored cap develop sticky goo in just a few days. So, if you work with it, you start with firm habits: use airtight containers, check those seals, and favor glass over plastics which often let water vapor sneak in.
Temperature Matters More Than Labels Suggest
Chemists have heard enough about “store in a cool, dry place,” but with TESOTf, cutting corners means trouble. Cooler storage slows down unwanted reactions, so I keep it in a fridge rated for flammable chemicals, ideally at 2 to 8°C. You don't need a sub-zero freezer; regular refrigeration does the job. What’s not negotiable: keeping it away from heat sources. No one wants to clean up a spill caused by pressure build-up.
Ventilation: The Thing People Downplay
Storing reactive chemicals without a dedicated fume hood can fill a room with fumes if something goes south. Safe storage in a ventilated cabinet near a functional fume hood is key. Sure, some labs skip the extra step, but inhaling even minor leaks can cause headaches and irritation — I’ve had colleagues sent home over just a trace. A well-ventilated setup reduces those risks.
Don’t Trust a Label from Last Semester
Over time, labeling and record-keeping start to feel like homework — tedious and easy to forget. But storing a bottle without the right date or initials leads to mix-ups and outdated materials. Routine checks stop small slip-ups from snowballing. I always mark the opening date, so I don’t stumble across a bottle past its prime. Disposal schedules and regular visual checks keep surprises in check.
Education and Training Save Property (and People)
Every year, chemical accidents trace back to one person who thought they already knew what to do. There’s a reason experienced chemists drill safety habits into new hands. Annual refreshers, summer safety quizzes, and walkthroughs in labs aren’t a waste of time. If everyone learns how TESOTf can bite back, accidents drop. Having spill kits and emergency eyewash nearby does more than meet a requirement — it’s backup for when someone’s guard drops.
Storage Safety Means Respecting the Risks
Working with chemicals like Triethylsilyl Trifluoromethanesulfonate means facing up to the very real hazards. Storing it wrong creates risks that pile up quietly until something gives. Safe practices, a good fridge, airtight bottles, dry spaces, clear labeling, and ongoing safety education make a world of difference. Years of working with reactive materials have convinced me — you can’t afford shortcuts, not with stuff this reactive. The extra care isn’t about following a rule, it’s about trusting you’ll go home with nothing more than the memory of another day in a safe lab.
Structure Matters in Chemical Reactions
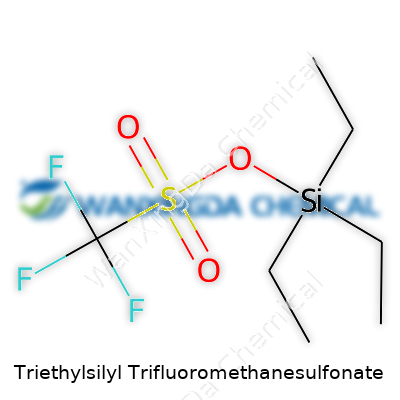
Triethylsilyl trifluoromethanesulfonate, better known as TESOTf to the organic chemist crowd, carries a fairly complex chemical structure. Compared to household names like table salt or vinegar, TESOTf comes with a much more specialized molecular makeup. The molecule sports a silicon core that's attached to three ethyl groups, forming the “triethylsilyl” part. At the business end, you find the triflate group—trifluoromethanesulfonate. On paper, the structure looks like this: Et3SiOSO2CF3, where "Et" stands for an ethyl group.
Now, it might sound like chemistry trivia, but the combination of these groups punches above its weight in organic synthesis. The silicon-oxygen connection with the heavily electronegative trifluoromethanesulfonate group turns TESOTf into a strong silylating agent. Scientists use it when they want to protect alcohols or phenols during tricky transformations. I remember messing with some of these reagents in grad school, always double-checking my glassware since triflate reagents tend to wreak havoc on wet or unwashed setups.
The Science Behind the Reagent’s Bite
What makes this structure so effective comes down to electronics. The triflate group steals electron density, making the silicon center even more eager to react with available nucleophiles like alcohols. This property means chemists can swap out hydrogen atoms in alcohols for a triethylsilyl group, blocking the site and preventing unwanted reactions down the line. The “protecting group” strategy isn’t just academic theory; it’s routine across medicinal chemistry and materials research, where fine-tuning molecules becomes critical.
I’ve seen labs grind to a halt because lesser reagents couldn’t keep up with TESOTf’s selectivity and strength. In drug development, one wrong move with an unprotected alcohol can tank an entire synthesis. That makes a reliable reagent like TESOTf an everyday hero behind the scenes. I used to joke that working with triflate reagents keeps you honest—if you cut corners, your product disappears, your column chromatography gets messy, and your time goes down the drain.
Handling Perils and Potential Solutions
With great reactivity comes great responsibility. TESOTf doesn’t play nice with water. The molecule’s triflate group reacts quickly and degrades, creating a headache for anyone who isn’t methodical with their drying protocols. Every organic chemist learns quickly that shortcuts in drying solvents or cleaning flasks rarely pay off. One good way to sidestep moisture headaches is to invest in properly maintained gloveboxes or Schlenk lines and to stash reagents in airtight containers. Small investments in glassware and desiccants pay dividends in yield and sanity.
Tuning the structure of compounds like TESOTf holds big promise for chemistry. Swapping out the triethylsilyl group or adjusting the triflate moiety can unlock new reactivity for different challenges. Modern labs are trying greener alternatives, looking at less hazardous solvents and milder reaction conditions. There’s room for innovation here, especially as chemists aim to reduce chemical waste and move away from harsh, resource-intensive procedures.
Final Thoughts: Not Just for Specialists
TESOTf may sound like just another reagent, but its structure shows how much power hides in molecular design. From undergraduate labs to commercial drug pipelines, the efficiency and reliability of silylating agents influence everything from timelines to costs. Talking about the chemical structure isn’t just academic navel-gazing—it’s the foundation of smarter, safer, and more efficient chemistry. That’s the lesson that sticks long after the last flask has been cleaned and put away.
Unpacking Its Uses in Synthesis
Some reagents zip through the pages of an organic chemistry textbook as everyday tools. Triethylsilyl trifluoromethanesulfonate—many just call it triethylsilyl triflate—plays a sturdy supporting role in the hands of both lab chemists and process researchers. I’ve seen its glass vials sitting beside flasks where careful hands shape molecules for new medicines and materials. Its popularity owes much to how reliably it guards sensitive alcohol groups as triethylsilyl ethers.
Selective Protection: Shielding Alcohols with Confidence
Most bench chemists have hit a wall, struggling to keep alcohol groups from interfering in a multi-step sequence. The challenge: you want that alcohol around at the end, but you can't let it react right now. Triethylsilyl triflate steps up to lock them down quickly and under mild conditions. Its popularity comes from the gentle way it “protects” these groups compared to bulkier silyl agents or those that crave water-free glassware and hours of stirring.
Instead of fighting your solvent or needing lab-grade patience, triethylsilyl triflate does the job even with hindered or less reactive alcohols. Chemists have leaned on its speed; set up the reaction, stir briefly, and finish the job. With water and oxygen kept out of the mix, you get high yields and clean products that make it easy to keep the synthesis pipeline moving.
Leaving Cleanly: Gentle Deprotection
A protecting group that overstays its welcome complicates everything. Triethylsilyl groups pop off smoothly when exposed to light acids. This feature matters a lot when the target molecule itself prefers mild treatment. I always prefer a path that lets me remove a protecting group without risking the carefully assembled pieces I’ve spent days or weeks building. Triethylsilyl triflate fits this need, offering straight-forward removal with dilute acid or even certain fluoride salts.
Pharmaceutical and agrochemical labs value this predictability. Nobody wants to rush a synthesis only to see a key intermediate break down at the end. Clean deprotection avoids waste and late-stage headaches, either in early discovery or scale-up campaigns.
Beyond Alcohol Protection
Triethylsilyl triflate's fame rests on its value as a protecting agent, but it doesn't stop there. In experienced hands, it activates certain carbonyl compounds. This increases the reactivity of electrophiles in glycosylation or acylation steps—important where you need glycoside or ester linkages, but can’t accept sluggish reactions.
I’ve seen chemists use it for the silylation of enolates as well, setting up “latent” functionalities for downstream steps. Its utility scales with creativity: in custom syntheses, its reputation as a dependable silylating agent lets researchers tackle tough transformations on sugar scaffolds, nucleosides, and even peptide projects.
Room for Improvement and Responsible Handling
With its potency comes responsibility. This reagent stays on the shelf of experts. I learned early on that it demands dry conditions and thoughtful disposal. One drop of water in the flask, and you get the unwanted triflate acid, which can chew up products and glassware alike. Mistakes here waste valuable starting material and create safety risks. Safer analogs do exist, but they usually require harsher conditions or deliver lower yields. Some labs push for greener options, emphasizing waste reduction and recycling strategies to reduce the environmental footprint that follows large-scale use.
The basics of chemistry haven’t changed, but smarter handling, fresh protocols, and better waste collection all help keep the focus where it belongs: making new molecules, not cleaning up messes.