Triflic Acid: A Superacid That Shaped Modern Chemistry
Historical Development and Significance
The world met triflic acid around the late 1950s thanks to efforts to push the limits of acidity in the lab. Chemists went searching for something stronger and more stable than the old standbys like sulfuric acid. Enter trifluoromethanesulfonic acid, better known as triflic acid. It started as a curiosity, made for experiments that needed something with a real punch. Over time, its reliability pulled scientists in. Word spread through journals and conference halls: here was a strong acid that refused to fuss with even the most stubborn reagents, and unlike some of its superacid cousins, triflic acid didn’t corrode all your glassware or leave a lab coated with toxic residues. In chemical culture, it isn’t just a reagent; it’s almost a rite of passage for researchers tackling tough reactions.
Product Overview
Triflic acid changed the game for organic chemists, bringing a superacid with remarkable stability and straightforward handling. Its structure—a trifluoromethyl group tethered to a sulfonic acid—holds up to water, resists oxidation, and stands firm in the presence of most solvents. Labs around the globe rely on it for tasks that would leave weaker acids struggling, whether you’re looking to knock off a protecting group or push through an electrophilic substitution. You won’t see it on the shelf at your local hardware store, but in research labs, its bottle stands out, instantly recognizable, protected behind glass doors and strict access protocols.
Physical & Chemical Properties
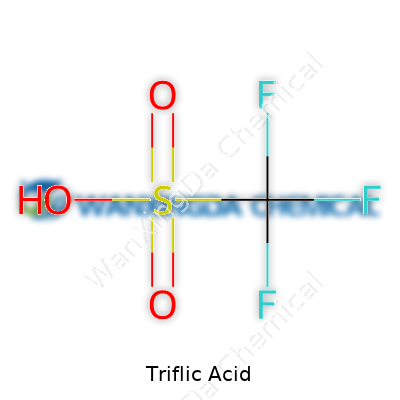
Out of the bottle, triflic acid pours as a colorless, almost oily liquid. If you came close for a whiff, you’d get a harsh, pungent smell—definitely not something you’d forget or want near your nose again. The hallmark of this acid lies in its acidity: the measured pKa value dips well into the negatives, making it one of the strongest proton donors in a chemist’s toolkit. Unlike other strong acids that can fizzle out in the presence of moisture or organize an outright attack on glass, triflic acid usually plays nicely with most containers and solvents. It mixes without protest in many organic reactions and rarely forms annoying byproducts, so the operator gets the selectivity and force that makes tricky chemistry possible.
Technical Specifications & Labeling
Bottles of triflic acid come stamped with clear hazard graphics because of its strength. You’ll read warnings about corrosivity and the need for protective gear—chemistry students quickly learn that gloves, goggles, and face shields aren’t optional here. On professional labels, manufacturers point out concentration, batch, and purity, but the real-world takeaway: treat every bottle with respect, and never rush the process of pouring or diluting it. In academic settings, seasoned lab staff give newcomers stern but necessary advice: spills can be dangerous, fumes are unforgiving, and don’t trust old glassware that might crack under the acid’s touch.
Preparation Method
Making triflic acid at industrial scale generally involves the chlorosulfonation of trifluoromethane, followed by hydrolysis. In the days before tight regulation, smaller-scale batches were made using hazardous reagents that demanded strict temperature control. These days, process chemists favor routes designed with minimal waste, closed systems, and recycling of byproducts such as hydrogen chloride and other volatile substances. I remember old lab stories about how one stray drip or splash during preparation could sideline a researcher with chemical burns—these are not tales meant to impress, but genuine reminders to respect the material.
Chemical Reactions & Modifications
Ask anyone who’s spent time in synthetic chemistry, and they’ll tell you triflic acid shows up when no ordinary acid will do. Its superacid nature cracks open stubborn carbon-oxygen, carbon-nitrogen, or carbon-silicon bonds. Chemists reach for it in Friedel–Crafts alkylations, aromatic substitutions, and protection/deprotection strategies for sensitive compounds. In my experience, triflic acid makes rearrangements and eliminations move forward without endless troubleshooting, boasting both speed and selectivity. On top of its direct use, you see chemists modifying it to form triflates—a versatile class of leaving groups in organic synthesis—which makes it the starting point for countless cross-coupling reactions. Whether pushing stubborn molecules into new shapes or activating them for coupling, triflic acid supplies power and predictability that enable ambitious projects.
Synonyms & Product Names
Triflic acid wears many hats in chemical literature. Officially, it goes by trifluoromethanesulfonic acid, but most chemists shorten this to triflic acid. You’ll sometimes see labels that say TFMSA, or just “triflate acid.” Fluorochemical suppliers use catalog numbers, but the core identity remains the same. No matter the name, seasoned chemists know exactly what’s in the bottle and how careful they need to be.
Safety & Operational Standards
Handling triflic acid safely means making preparation and caution your top priorities. You always suit up: double-layered gloves, chemical splash goggles, long sleeves, sometimes even specialized face shields when pouring. Fume hoods take on extra importance—the fumes sting nose and throat, and accidental contact with skin or eyes can cost you weeks of recovery. All spills call for fast response with absorption pads and neutralizing agents, and spent glassware must be rinsed thoroughly before it leaves the acid workspace. In my own experience, respect for triflic acid only gets stronger over time. You learn to double-check concentrations, never work alone, and communicate closely with your team during scaling. Incidents don’t happen often, but every chemist I know keeps memories of close calls as motivation for meticulous preparation and respect for protocols.
Application Area
Triflic acid has carved out a home in every area of synthetic organic chemistry. Academic groups lean on it to pull off cutting-edge rearrangements, cyclizations, and functionalization of complex natural products. In the pharmaceutical world, it’s brought in when making certain intermediates or drug scaffolds would hit a wall otherwise. Researchers tailoring polymers or exploring exotic materials appreciate its stability and speed. Beyond labs, the electronics industry benefits from its ability to help prepare high-purity semiconductor materials and fluorinated compounds. On a personal note, those years of running reactions that alternated between stubborn starting material and breakthrough moments usually found me pouring a measured dose of triflic acid and seeing the mixture change color or clarity in that telltale way. Nothing else matches it for reliability in a pinch.
Research & Development
Teams continue to push triflic acid into new territory. Enabling selective activation of otherwise inert bonds, facilitating rearrangements, or unlocking transformations for small-molecule drugs—these stories pop up in journals all the time. A special area of growth comes from using triflic acid in sustainable chemistry, trying to swap out reagents that produce more waste or hazardous byproducts. Organic electrochemistry occasionally borrows its strength for reactions triggered by current instead of heat, hoping to bring more efficiency and less environmental harm. In many academic groups, students learn the “old-school” methods, then brainstorm how a molecule like triflic acid could speed up reaction times or boost yields for complex targets. There’s an ongoing drive to find derivatives that might offer similar power with less corrosive demand, but so far, triflic acid sticks around as the benchmark.
Toxicity Research
Every powerful reagent brings its risks, and triflic acid ranks high on the list of substances that demand healthy caution. Reports from occupational safety research recount skin, eye, and respiratory damage caused by accidental exposure, along with case studies where inhaled vapors or delayed neutralization led to hospitalization. Long-term environmental impacts look manageable thanks to the compound’s reactivity and its tendency to break down or dilute when handled properly, but persistence in improperly treated waste streams raises questions for future regulation. Over the years, labs learned the hard way about the need for sturdy training programs and emergency drills. While chronic toxicity studies lag behind, anyone who’s spent time with superacids learns that gloves and fume hoods save more than just lab coats—they keep entire teams safer and research on track.
Future Prospects
Work continues to expand the role of triflic acid, not just in bigger or faster reactions but in the push for greener, more efficient methods. Researchers look at ways to recover and reuse it to cut down on costs and environmental impact, and some try to pair it with catalytic approaches that squeeze more mileage out of every drop. There’s hope that more selective derivatives could offer the same performance with fewer hazards. In academic settings, the acid keeps its status as a teaching tool and problem-solver, while industry keeps investigating its application in making next-generation materials and drugs. Experience tells me that any leap forward in chemistry, from a new drug to a new polymer, starts with tools that offer both muscle and control. Triflic acid, with all its power and peril, will stay in the story for years to come.
What Triflic Acid Actually Does in Daily Lab Work
Triflic acid rarely shows up in conversations outside of advanced chemistry labs, but anyone who’s handled it won’t soon forget the experience. Chemically known as trifluoromethanesulfonic acid, this stuff earns the label “superacid”—which isn’t just a buzzword. Traditional strong acids like sulfuric acid feel tame next to triflic acid. In my own graduate work, even the veteran researchers treated the vial with more respect than just about anything else on the shelves.
So why do chemists keep coming back to something that can burn through gloves and glassware? Triflic acid brings an unmatched ability to create and break chemical bonds, especially those reluctant to react under mild conditions. In organic synthesis, it’s used to remove protective groups with surgical precision. It stands out in reactions like alkylation and acylation, letting chemists build complex molecules more efficiently than other acids can manage. Papers published on new medicines, high-performance materials, or cutting-edge electronic components often list triflic acid somewhere in the process.
The Double-Edged Sword of Superacidity
With great power comes real risk. Even short exposure to triflic acid’s fumes sends everyone back a few feet. For every successful synthesis, more than one lab worker suffers damaged gloves or skin. Its corrosive power spreads beyond the lab. Equipment needs special seals and barriers, racking up higher costs and making training a constant necessity.
There are environmental headaches too. Chemists push for cleaner, less hazardous reactions wherever possible, but triflic acid doesn’t break down easily and introduces new disposal challenges. For labs without well-funded waste handling, using this acid demands extra vigilance—or avoiding it altogether unless absolutely necessary. Improper disposal can lead to soil or water contamination that lingers.
Why Persistence Pays Off
Despite the drawbacks, the achievements unlocked by triflic acid deserve recognition. Drug development wouldn’t move as quickly—or sometimes at all—without the shortcuts it creates in making complicated molecules. Firms racing to develop new therapies for cancer, infectious diseases, or neurological disorders often turn to triflic acid for the time and cost savings it delivers. In materials science, designing ultra-pure polymers or novel battery materials relies on its unique acidity.
The story doesn’t stop at traditional chemistry. Some ambitious startup teams see triflic acid as a gateway to new manufacturing methods that slash energy usage or minimize toxic byproducts. Recent research explores “greener” deployment, seeking solvents and support materials that can contain the acid or allow for a closed-loop process. Policy makers and funding agencies could do more to support labs working on triflic acid alternatives or safer application methods.
Building a Smarter, Safer Future
For those of us who’ve seen triflic acid in action, caution and optimism go hand in hand. Its contributions to medicine, green chemistry, and high-tech innovations demonstrate the value of taking calculated risks—so long as everyone in the lab remains alert, honest about the hazards, and ready to adapt. Education needs to focus not just on the power of these reagents, but on what their use says about safety, ethics, and environmental responsibility. Through support for research and communication, science will continue unlocking the full potential of tools like triflic acid, minimizing harm along the way.
The Reality Behind Triflic Acid’s Reputation
Plenty of folks in labs get wide-eyed at the mention of triflic acid. It’s not some everyday household cleaner. Known in scientific circles as trifluoromethanesulfonic acid, this is one of the strongest acids you’ll find outside of science fiction stories. Its punch comes from how easily it hands off protons, and it doesn’t mess around if you give it the chance to react to living tissue or most common materials.
What Makes Triflic Acid Not Your Average Acid?
Walking through a chemical storeroom, you see a range of acids—some fume, some corrode, some stain. Then there’s the bottle of triflic acid tucked away with all sorts of hazard labels. This stuff will tear through glassware, metal, even skin, given half an opportunity. If a splash lands even on glass, damage isn’t just likely—it's nearly certain. It doesn’t just sting; it keeps burning, turning minor slips into major accidents. I’ve seen chemists with years of experience double-check their gloves, goggles, and well, their courage, before cracking open a container.
A lot of people have worked with sulfuric acid during college chemistry labs. Triflic acid brings that memory back and multiplies the danger. It has no time for hesitation—one mistake and you’re rushing to the emergency shower, hoping you got to it fast enough.
Who Handles Triflic Acid?
Only folks with advanced training even think about using this acid. You won’t find it in classrooms or general industry use, and for good reason. Most often, researchers running advanced organic syntheses use it—especially those after reactions you can’t pull off with anything less aggressive. In basic science, pushing molecules around needs serious tools, and triflic acid does the job for a select handful of cutting-edge experiments.
Why the Hype? Is the Danger Overblown?
No doubt, some stories stretch the truth for drama. Still, the threat here is absolutely real. The Centers for Disease Control and NIOSH flag it as a severe corrosive. Open bottles or careless transfers can fill a fume hood with acid vapor. Even the vapor attacks skin and eyes. Long sleeves and face shields matter as much as gloves, if not more.
Some labs that learned the hard way now keep calcium gluconate gel and special spill kits on hand, just in case. Even a few college incidents—some resulting in long-term injury—led to stricter institutional training. Chemical manufacturers openly detail the risks online, and even casual lab safety conversations often concede, “If you can avoid it, do.”
Making Research Safer—Real Solutions
Fires, spills, and splashes become less frequent with strict protocols. Double-gloving, slow additions, working inside a robust fume hood—these aren’t optional steps. Many research groups now train rookies with milder acids and only move on to triflic acid after repeated safety sign-offs. Automatic pipettes and sealed transfer systems further limit exposure. Some facilities even require written logbooks for each gram handled.
There’s more to be done. Pushing suppliers to provide better packaging that limits the risk of splashes would help. Supervisors keeping hands-on for all steps, not just sign-off forms, can bring real change. And it’s smart for research institutions to support better spill response and medical care, rather than hope nobody ever needs it.
Working with triflic acid isn’t about bravado. It’s about respect—for chemistry, for the colleagues down the bench, and for everyone who steps into a lab tomorrow.
The Harsh Reality Behind Triflic Acid Storage
Triflic acid doesn’t mess around. After years watching chemists wrangle with dangerous substances, this one stands out for its sheer might. Just a drop splashed from careless handling will eat through skin, mess up metal, and throw anyone’s confidence in their safety training out the window. Some folks call it the king of acids, and not as a joke. Chemists use it to create groundbreaking molecules for medicines, electronics, and specialized plastics—stuff that makes modern life possible, but not without a price.
Few things remind us how thin the shield between discovery and disaster really is. Open a bottle of triflic acid in the wrong room, or store it like you do regular lab supplies, and you’re gambling with people’s lives and possibly the lab’s whole existence. It boils at over 150°C, but that’s no comfort with its fumes gnawing their way through containers and nasal passageways. I've seen new researchers try to treat it like sulfuric acid, with heavy gloves and a face shield. Those help, but triflic acid’s way nastier. Simple latex gloves dissolve fast. Even glassware will eventually pit or etch if it isn't checked regularly.
What Safety Looks Like in Practice
If you’re storing a monster like this, use containers built for true endurance. Many labs trust heavy-walled PTFE (sometimes called Teflon) bottles. Glass with PTFE liners works, but you never want to see drops collecting around cracked seals. Always double-check compatibility charts before pouring anything. Never use containers with metal fittings or standard plastic caps.
Lock it in a ventilated acid-resistant cabinet, far from regular pathways. Don’t stash it under a basic fume hood and walk off thinking it’ll take care of itself. The vapor can corrode everything—ventilation in these storage cabinets should vent outside, with materials designed to resist strong acids. Keep it below eye level. If someone fumbles, the bottle falls, not the acid. More than once, protocols have saved fingernails and eyesight thanks to storing these bottles low.
Moisture’s another enemy here. Water transforms triflic acid from angry to explosive, spitting out toxic gases and energy. Keep desiccants handy when retrieving or returning containers. Wipe down surfaces and double-bag bottles before closing the cabinet.
Factual Support: Why All This Trouble?
Triflic acid can dehydrate organic matter on contact. It eats through common organic polymers, corrodes many metals, and emits gases that attack electronics. The American Chemical Society ranks it among the most potent non-oxidizing acids, used in specialized industrial and research settings. One splash on exposed skin can cause severe burns in moments. According to the National Institute for Occupational Safety and Health, immediate and thorough rinsing is necessary after any exposure.
Incidents from neglecting storage rules don’t just lead to injuries—they prompt evacuations, costly facility overhauls, or lab shutdowns. Regulatory agencies investigate every triflic acid accident. That’s not bureaucracy, but hard-earned experience from tragic outcomes.
Improving Triflic Acid Storage Moving Forward
Labs working with this acid ought to revisit training every year and quiz even the senior staff. Make safety drills about triflic acid a routine. It helps to keep updated signage around storage and designate one or two trusted people for all handling and inventory tasks. The goal isn’t to make anyone paranoid—it’s to act like triflic acid is always waiting for a slip.
Labs do their best work when everyone understands that some advances demand respect, not shortcuts. Storing triflic acid safely keeps research moving and people safe, and there’s no reason to settle for less.
A Powerhouse in a Bottle
Triflic acid carries a reputation not just for its catchy name but for the punch it packs in science labs. Chemists know it as one of the world’s strongest acids, which matters more than it might sound. This stuff does more than bubble and fizz. It brings relentless chemical muscle wherever it turns up.
An Acid Above the Rest
I remember my first introduction to triflic acid. White fumes rose off the bottle–that’s the warning you never ignore. Pour water into it, you don’t see gentle mixing—you get steam and violent heat. Power likes company, so triflic acid hangs with a crowd of chemicals known for moving mountains in organic synthesis. The acid strips away tough groups in molecules that would shrug off most other attempts. Need to install or yank off a functional group in a stubborn compound? Triflic acid steps into the ring without flinching.
Not Just Strong—Consistently Reliable
Working with it pounds in a simple lesson: not all acids act the same. A strong acid can donate protons easily. Triflic acid does it in spades, but that’s not the end of the story. Unlike sulfuric or nitric acid, it doesn’t oxidize the daylights out of everything it touches. The molecule’s trifluoromethanesulfonic backbone (CF3SO3H) stands out for staying stable and not throwing in unwanted side reactions. In messy chemistry, that’s a lifesaver. Scientists trust it as a catalyst for making medicines, polymers, and new materials that keep tech moving forward.
Handling and Hazards
There’s a darker edge. The power of triflic acid doesn’t offer any mercy. I’ve seen gloves dissolve and glassware etched, just from sloppy handling. Its fumes drift in the air, itching the throat long after leaving the fume hood. No training can erase the stress of triple-checking every move when a drop on the skin spells a trip to the hospital.
From a safety perspective, triflic acid’s hazards get compared to hydrochloric acid or even hydrofluoric acid, but with its own mean streak. One tiny bottle calls for layers of safeguards—face shields, special gloves, and equipment designed to resist corrosion. Spills require immediate action, and nobody works alone with it.
Why Use It at All?
So, why not find something gentler? Nothing else will do for certain chemical transformations. Pharmaceutical scientists lean on its strength and reliability. It helps make advanced molecules needed for life-saving drugs. Battery materials, superconductors, and petroleum research all find jobs for triflic acid that would stump weaker contenders. I’ve met chemists who hate the stuff, but none deny its results.
Looking for Safer Solutions
Everyone in the lab wonders if there’s a safer champion waiting in the wings. Research keeps hunting for alternatives. Some companies try enzymes or less aggressive acids, but the world of advanced chemistry doesn’t let go of triflic acid easily. Regulations get tighter each year, pushing labs to cut unnecessary risks, but as of today, nothing replaces triflic acid when strength and stability need to show up together.
The takeaway feels simple. If you spend your days around molecules that don’t play nice, you learn respect for triflic acid. Its properties push science forward, but with a running reminder: every breakthrough comes with risk, and safety stands side-by-side with progress.
The Real Dangers of Triflic Acid
Anybody who’s worked in a lab for more than a semester knows triflic acid as the kind of chemical that makes even the most seasoned chemist pause before popping open the bottle. There’s no sugar-coating what this stuff can do. Triflic acid can eat straight through common gloves and even some plastics. Skin contact isn’t just a matter of irritation; it’s a one-way ticket to serious burns. Inhaling the fumes gets the lungs and respiratory system into the danger zone. Stories circulate among students and researchers. No one forgets the warning: this acid doesn’t forgive mistakes.
Practical Precautions from Real-Life Experience
My first encounter with triflic acid happened during an undergrad summer internship. The principal investigator drilled three points home before anyone did anything: don’t trust your instincts, trust your PPE, and always move slow. Here’s what my mentor emphasized, lessons I still carry.
Gloves matter; but nitrile or latex alone won’t cut it. Heavy-duty neoprene gloves work better, and checking for holes before starting means everything. Next, not all lab coats are equal either. A splash of triflic acid goes through cotton like water. I switched to an acid-resistant apron over my coat, plus face and eye shields. Once, I watched a tiny droplet of acid sizzle a hole in an old lab bench. It left behind a black scar, a permanent reminder to double up on protection.
Ventilation isn’t optional. Working in a regular fume hood keeps those toxic fumes out of your breathing zone. Breathing them in feels uncomfortable at best; in reality, chronic exposure can lead to real lung damage.
Preparation and Handling—No Shortcuts
It pays to set up everything before breaking the seal on the bottle. Keep neutralizing agents like sodium bicarbonate nearby—spill kits and emergency eyewash showers should never be a distant afterthought. I’ve seen researchers fumble for the nearest sink, which often leads to panic. Long ago, during my stint at a university chemistry department, we lost a cartful of samples because somebody left sealing caps loose. The acid made quick work of the plastic causing an overnight mess.
Always add acid to water, never the other way around. Dumping water into a concentrated acid means dangerous splashing and heat. There’s a reason this rule gets repeated in every lab safety session. Starting with cold, slow mixing in a sturdy container keeps accidents out of the news.
Learning from Accidents and Setting the Bar Higher
Mistakes still happen, even to the cautious. That’s why honest incident reporting matters more than saving face. Skipping a step or assuming old gloves work just as well as new ones sets up colleagues for harm. Everyone benefits when labs share near-misses and injuries openly, so new procedures come out of real-world experience.
Better training programs, more realistic simulations, and refresher courses each semester can raise both awareness and basic instincts. Some research groups bring in experienced chemists to share what they've learned the hard way. That’s the kind of peer-based mentoring which builds a culture of respect, not complacency.
Building a Culture That Values Health
It’s easy to treat triflic acid like just another bottle on the shelf, but the stakes run higher than clean beakers and impressive reaction yields. Nobody benefits if the pursuit of cutting-edge science comes at the cost of scars or lost work time. Taking safety personally, respecting the hazards, and making better habits routine serve everyone in the lab and beyond. If safety kits get replenished, procedures updated, and people keep swapping near-miss stories, that’s progress worth fighting for.