Triflic Anhydride: A Closer Look at A Chemical Powerhouse
Historical Development
Chemistry doesn’t stand still. Tools that chemists use today started as ideas and experiments in crowded labs of the past century. Triflic anhydride, or trifluoromethanesulfonic anhydride, emerged from this vibrant search for better reagents. When researchers in the mid-1900s began exploring sulfonic compounds for their unique leaving group properties, they weren’t out for another sulfur-rich experiment—they chased real improvements in synthesis. In 1964, scientists rolled out triflic anhydride as a way to make sulfonation faster and cleaner, changing how organic transformations got done. The arrival of this reagent didn’t come out of a vacuum; it grew from decades of incremental lessons from sulfur, oxygen, and fluorine chemistry, each round making reactions sharper, products purer, and yields higher.
Product Overview
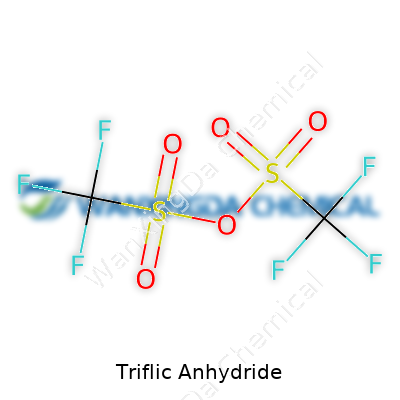
Stare down a glass ampule of triflic anhydride, and you see a clear, sometimes slightly yellow liquid. The real power lurks in its structure: two trifluoromethanesulfonyl groups lashed together by an oxygen atom. It stands apart for its fierce reactivity. Labs across the world reach for it to activate carboxylic acids, to create robust sulfonate esters, and to push along reactions that would sputter with weaker choices. Chemists value efficiency. With triflic anhydride, what once took hours can settle out in minutes, often with better yields and fewer side-reactions chewing up precious building blocks. Over years in a research environment, I’ve noticed it’s the first line of attack in stubborn functionalization steps.
Physical & Chemical Properties
Triflic anhydride is a heavy liquid, with a boiling point around 81-84 °C under reduced pressure and a density over 1.6 g/mL. It smells sharply acidic, not a surprise given the lurking power of the triflate group. Unlike other acid anhydrides, it packs a punch even at room temperature. Water doesn’t stand a chance—upon contact, triflic anhydride reacts violently, with fumes and sizzling heat that remind you this isn’t a chemical for the unwary or the unprepared.
Technical Specifications & Labeling
Look on a lab shelf and a bottle of triflic anhydride demands a cautious approach. Labels warn of severe burns, high reactivity, and the need for chemical-resistant gloves and fume hoods. Experts don’t cut corners with this stuff. Exact technical details matter in the lab; high purity and dryness mean fewer unwanted surprises. Over the years, I’ve seen that even tiny impurities can turn a straightforward activation into a wasted afternoon cleaning glassware and chasing down byproducts.
Preparation Method
Older textbooks talk about the classic route: reacting triflic acid with phosphorus pentoxide. The process takes careful temperature control and water exclusion. Scales up only as far as someone feels comfortable with the risks. Modern suppliers still stick close to these roots, with tweaks for efficiency and containment. Nobody enjoys dealing with hazardous off-gassing, so tightly controlled reactors and air-free techniques rule the day. Lab-scale methods remain mostly unchanged, favoring simplicity over fanciness.
Chemical Reactions & Modifications
Few reagents match triflic anhydride for sheer impact in breadth of chemistry. In synthesis, it turns mundane alcohols into triflates—leaving groups that open the door to almost any cross-coupling you can name, from Suzuki to Stille and beyond. It makes acids into mixed anhydrides ready for peptide couplings. It cleans up sluggish Friedel-Crafts acylations and flips on dormant rings in aromatic substitution. I’ve seen students try to “get by” with lesser agents, only to run back to triflic anhydride after a string of failures. Its modifications—whether with bases or in combination with Lewis acids—yield pathways to fluorinations, alkylations, and even tricky cyclizations. The versatility is hard to overstate.
Synonyms & Product Names
In journals and catalogues, triflic anhydride has a habit of multiplying its names: trifluoromethanesulfonic anhydride, TF2O, and the abbreviation Tf2O. These labels trip up newcomers, but for those involved in synthetic research, the interchangeable terms are second nature. What matters most is the unmistakable identity the compound brings to the bench—every time a reaction calls for high-gear activation, this is the flask that comes off the shelf.
Safety & Operational Standards
This reagent rewards careful handling and punishes carelessness. Skin or eye contact means a trip to the emergency wash, inhalation can damage airways, and mixing with water or incompatible solvents can create vicious exotherms. Labs with any sense keep this compound strictly in the fume hood; splash goggles, nitrile gloves, and face shields aren’t optional. Storage in tightly sealed ampules keeps atmospheric moisture away, protecting both chemical and chemist. In my experience, treating triflic anhydride with respect, never in haste, builds routines that keep everyone safe—especially new researchers unaccustomed to its reputation.
Application Area
Pharmaceutical labs translate triflic anhydride’s high reactivity to real-world impact. When a new drug candidate needs a unique backbone, or a metabolic tracer demands tricky halogenation, this reagent shows up again and again. Its speed and conversion rates mean fewer steps wasted, which adds up when screening hundreds of compounds. Materials chemists use it to build specialized polymers and create functionalized surfaces. Even in the growth of specialty chemicals for agriculture or electronics, triflic anhydride provides shortcuts that other reagents can’t reliably deliver. In my own projects on bioactive molecule synthesis, it shortened timelines and cut waste, offering a leap over trial-and-error with less consistent reagents.
Research & Development
The search for new reactions never stops. Scientists tinker with triflic anhydride—modifying conditions, pairing it with bases or transition metals, and chasing more selective transformations. This kind of R&D leads to cleaner reactions, new building blocks, and sometimes, true surprises in selectivity and yield. Much of the modern growth in heterocycle synthesis leans on triflic anhydride to install functional groups where other chemicals fall short. While academia often focuses on the “why” behind each transformation, industry adopts those lessons for scale-up and greener processes. Personal experience in method development reminds me: even a small tweak in triflic anhydride protocol can unlock a stuck route or boost a flagging yield, driving home just how critical this reagent remains in the chemist’s arsenal.
Toxicity Research
Safety doesn’t end at the fume hood. Scientists have dug into the toxicology of triflic anhydride, tracking its dangers both in direct exposure and environmental release. The compound reacts so fiercely with tissue and moisture that accidental splashes mean serious injury. Acute toxicity studies flag it as a skin and respiratory hazard. Waste treatment facilities and chemists working at the interface of bench and environment still debate best disposal practices, aiming to keep this fast-reacting compound out of public waterways. There’s no easy fix here—minimize releases, handle every drop with respect, and ensure everyone in the lab takes those safety briefings seriously. I’ve seen colleagues develop careful double-checking habits after close calls, a mindset that pays off over long careers.
Future Prospects
The future of triflic anhydride isn’t about replacing it, but about using it smarter—finding greener pathways, reducing waste, and integrating with automation. As sustainability goals in chemistry tighten, the drive is to tune procedures so reactions get maximum yield from minimum waste. Researchers push for catalysts that can regenerate spent triflates, for solid-phase uses that make separation cleaner, and for robotics that dose the reagent with surgical accuracy. In pharmaceutical pipelines where speed and efficiency drive decisions, triflic anhydride will continue shaping how discoveries make it to the next stage. Its unique mix of power and risk embodies the paradox of progress in chemical research—move forward with care, learn from missteps, and keep searching for new ways to unlock what’s possible.
Most people outside research labs have never heard of triflic anhydride, but its fingerprints show up in everything from advanced electronics to new drug discoveries. This clear liquid hardly ever appears in consumer glossaries, even though it helps scientists pull off chemical reactions that used to seem impossible. With its strong reputation, chemists treat triflic anhydride almost as a secret weapon when building complicated molecules.
Turning Simple Stuff into Useful Goods
My earliest introduction to this chemical happened during graduate school. A fellow student managed to convert a stubborn alcohol into an entirely different group by splashing in a dose of triflic anhydride. Watching that color change in the flask, I saw more than just a reaction—he opened the door to new molecules. That day, I realized that a small accessory reagent like this could shape how we treat disease, make new materials, or even shrink the size of tech devices.
The power of triflic anhydride comes from its talent at activating otherwise reluctant molecules. It swaps out a hydrogen atom for a trifluoromethanesulfonyl group. This change turns a sluggish compound into a much more cooperative partner. It helps synthesize parts of cancer drugs, install sensitive groups onto DNA building blocks, and build custom polymers for devices. In pharmaceuticals, speed and control matter, especially when a single step can dominate the cost of a medicine.
The Trade-Offs No One Warns About
I sometimes hear students describe chemicals by their hazards alone. It is true that triflic anhydride stings eyes, burns skin, and reacts violently to water. That said, focusing only on dangers ignores why chemists still reach for it. With proper safeguards, it transforms stubborn building blocks, shortens complex syntheses, and can even boost environmental efficiency since fewer steps mean less waste.
I remember a project where colleagues tried gentler substitutes. Each alternative produced more byproducts or forced us to add extra cleaning steps. Eventually, the team returned to triflic anhydride because it accomplished the job swiftly. It cut costs and skipped two waste-generating stages.
Navigating a Chemical Balancing Act
Finding a balance between powerful reagents and ethical lab practice comes down to discipline. Safety training, tight ventilation, and chemical containment work together to keep risks low. There are groups now pushing for greener methods. Promising research explores solid versions that won’t splash or new catalysts that mimic its skills but use less toxic starting materials. Nature always rewards clever problem-solvers—those who find chemicals that do more with less harm usually land on the front page of scientific journals.
The next time you see a news story about new antibiotics, cancer treatments, or breakthroughs in OLED screens, remember chances are good that somewhere during development, a chemist reached for triflic anhydride. This chemical rarely makes headlines, but it shapes the breakthroughs we depend on every day. Chemists, engineers, and process experts keep refining how they handle it, always looking for safer or greener setups, but for now, triflic anhydride remains a driving force in ambitious laboratories around the world.
Triflic anhydride isn't something you want to treat as just another bottle in the cabinet. Anyone who's measured it out during a late-night synthesis remembers the way the bottle seems to radiate trouble. This reagent’s strength comes with real hazards, and chemistry students learn quickly to take every step seriously—for their safety and for those around them. I think about that first time our advisor hurried through the door after a rookie mishap with triflic anhydride. No one in the lab forgot that heated advice: respect the compound, or pay dearly.
Why Triflic Anhydride Demands Caution
Triflic anhydride cuts through water, skin, and gloves much faster than other acids. Personal stories from research groups echo warnings in the literature: contact burns, lung irritation, and surprisingly quick reactions with moisture. It releases toxic fumes, including hydrogen fluoride, when it reacts with anything wet. Many solvents—even those stored dry—can send up nasty vapors that chase you away if triflic anhydride splashes or spills. This story repeats across university departments and industrial labs alike. Ignore the risk and you might spend the day in medical care instead of finishing an experiment.
Real harm can come from complacency. There's a culture of bravado in some labs, and that’s where mistakes happen. When even seasoned chemists wince at a whiff of the reagent, it says a lot about its risk. Triflic anhydride’s corrosiveness beats standard acids. One drop on the skin does permanent damage. Breathing it in leads to burns in the nose and lungs, so doubts about ventilation or glove type don’t end well. If someone uses the wrong gloves or forgoes goggles, hospital trips aren't uncommon. It’s never worth it.
Practical Safety Measures Worth Following
Lab safety comes down to habit. I stick to nitrile gloves—double layered, changed after each session. Eye protection matters even more, since triflic anhydride finds a way past splash guards when people least expect it. Chemical fume hoods save lives; I don’t open the bottle outside of one. Squeeze bulbs and pipette aids keep fingers away from splashes. Once, I caught a spill with a fresh absorbent pad before it spread—luck played a part, but quick thinking matters.
Good ventilation and a prepped spill kit stand guard against the unpredictable. Hydrofluoric acid antidotes and calcium gluconate gel sit on my shelf next to the reagent. Training in chemical burns and first aid isn’t optional, either—no one ever credits luck in these emergencies. It comes down to community: teammates in the lab checking in, running drills, updating each other on near-misses. Information about symptoms or antidotes should never be buried in a binder. Signs and emergency numbers stay out in the open. Supervisors have to set the tone—no shortcuts, ever.
Smart Storage and Waste Disposal
Anyone storing triflic anhydride keeps it dry, tightly sealed, and labeled with hazard warnings clear as day. Refrigeration delays breakdown, but only in explosion-proof units. Old stock or contaminated bottles trigger cleanout protocols—even after years of use, containers never go into regular trash. Specialized waste jars, marked for acids, make a big difference. Waste contractors know what’s inside and never risk pickup confusion. Asking the department hazardous materials coordinator sounds obvious, but skipping one email leads to headaches down the line.
The best chemists I know don’t treat triflic anhydride like any other tool. Those habits keep careers—and lives—intact.
Facing Up to Triflic Anhydride’s Risks
In a lab packed with all sorts of chemical hazards, Triflic Anhydride stands out for the simple reason that it reacts fast and harshly with water and most organic material. I’ve worked in university research labs where everyone knew the consequences of a minor spill: just a few drops of moisture can set off heat and dangerous fumes. This isn’t something anyone can shrug off or recover from with a paper towel. The risks stretch from immediate chemical burns to even long-term health problems if the fumes sneak their way into the air. Even seasoned chemists pause and double-check the label before touching the bottle.
Suitable Storage: It’s Less About Tradition, More about Survival
Many folks new to the chemical world share a simple goal: keep everything dry and out of sunlight. This general rule doesn’t go far enough for Triflic Anhydride. The bottle doesn’t just belong in the dry cabinet. Think about a dedicated desiccator, built to keep moisture at bay. In most labs I've worked in, someone always double-bags the container with sealed plastic, then tucks it in a tightly-closed desiccator jar with a thick bed of drying agent. This isn’t overkill—it’s just smart practice. Leaving it anywhere near the regular flammable cabinet invites trouble. Most flammable cabinets stay at room temperature, and even the tiniest gap in a bottle cap can mean ruin.
Plastic, Not Glass—And Don’t Fool Yourself
Triflic Anhydride loves to eat away at glass over time. Years ago, I saw the aftermath of forgetting about this detail: a cloudy glass bottle that crumbled at the neck, leaking its contents and nearly ruining a bench top. Chemists who know their business only choose strong plastic storage bottles, clearly labeled and checked before opening. Long-term storage in glass feels foolish in hindsight, yet old habits still crop up in smaller labs. Switching bottles as soon as a shipment arrives becomes an indispensable weekly ritual. It’s one of those routines you wish everyone followed, as the alternative leads to nothing but cleaning up toxic messes.
Ventilation, Visibility, and Vigilance
No sense in preparing the perfect storage if you forget about the air. Triflic Anhydride turns nasty in a hurry when something goes wrong, so storing it inside a fume hood—not just for daily use, but for full-time storage—makes sense. In larger facilities, some managers rig up local extraction systems, keeping vapors from spreading. On top of that, clear labeling and routine inventories keep the risk of unnoticed leaks or corroded seals low. I remember more than once catching a cracked cap before it turned ugly, thanks to a simple weekly walk-through. Regular eyes keep labs safe.
Building Better Habits and Spreading the Word
Safe storage of Triflic Anhydride isn’t complicated by fancy gadgets—it just takes respect and follow-through. Sharing stories, like the time a storage blunder cleared out an entire lab wing for a day, works better than any poster on the wall. Passing along practical advice, like keeping an emergency spill kit at arm’s reach, helps everyone learn. Nothing replaces the real-world lessons earned through cautious storage and quick thinking. At the end of the day, knowing where and how to store dangerous chemicals keeps science moving and people safe.
The Chemical Formula of Triflic Anhydride
Let’s talk about a molecule that packs quite a punch in modern labs: triflic anhydride, with the chemical formula (CF3SO2)2O. Anyone who’s spent time in synthetic chemistry circles recognizes its reputation for making tough transformations possible, especially when classic approaches can’t cut it.
Why Chemists Care About Triflic Anhydride
Back in my university days, I remember those benches scattered with glassware, pipettes, and the familiar tang of something acidic in the air. We learned quickly that reagents with power often come with quirks, and triflic anhydride was no exception. Its chemical formula might look simple written out, but the stuff inside that bottle changes the way reactions happen.
Triflic anhydride acts as a top-tier activator for hydroxyl and amino groups. Add it to an otherwise sluggish mix and suddenly new pathways open up, making it easier to introduce leaving groups or boost reaction rates. Organic chemists often rely on this molecule to fine-tune molecules for drug discovery or complicated materials science projects.
What Makes Triflic Anhydride Special?
The unique power of (CF3SO2)2O comes from the triflyl group it carries. Introduce that into a reaction, and everything gets more reactive. Chemists use it to make sulfonate esters from alcohols or to turn amides into more versatile intermediates. What seems like a harsh reagent actually fills in gaps left by milder options that just stall out or give messy results.
The world’s pharmaceutical factories count on selective, reliable reactions to churn out everything from painkillers to heart medications. Triflic anhydride gives process teams added flexibility, so smaller tweaks mean lower costs and fewer by-products.
Weighing Safety and Sustainability
Every time someone handles triflic anhydride, the safety gear gets double-checked. Years in the lab taught me that respect beats bravado each time out—especially with chemicals that fume, react with water, or release acid gases. Its high reactivity makes it both powerful and hazardous, so ventilation and cut-resistant gloves are standard.
Triflic anhydride doesn’t belong in waste drains. Environmental teams have raised the alarm about perfluorinated compounds. These molecules break down slowly, gathering over time in soil and water. Most labs collect and neutralize them, but the risk never fully goes away. Companies now look hard at their synthetic steps, searching for safer or greener options. The future leans toward alternatives that do similar chemistry without environmental drawbacks.
Looking Forward
Knowing the formula and power of triflic anhydride unlocks a toolkit for creative organic synthesis. The challenges lie in handling it wisely and searching for less burdensome options down the line. Some process chemists experiment with flow chemistry to reduce direct exposure. Others tweak older steps to sidestep the need for strong sulfonating agents. Safety and environmental concerns aren’t opposite chemistry—they live side by side, shaping how this molecule gets used across labs and industries.
The Risks Surrounding Triflic Anhydride
Triflic anhydride stands out in a lab for how it can make or break a synthesis, but it brings a laundry list of hazards alongside its usefulness. Chemists who work with it, myself included, know the sense of caution that sets in the minute this bottle lands on the bench. This isn’t just another reagent sitting in a cabinet — this stuff can burn right through gloves, skin, and even the airways if you’re not careful.
It only takes one whiff of the sharp, choking irritant smell to realize it has no business anywhere but a fume hood. Even small spills release fumes that rapidly attack the eyes and respiratory tract. I’ll never forget the time a careless transfer left a faint mist in the hood — and my nose burning minutes later. Goggles and gloves won’t always cut it; even a tiny splash or a touch can blister skin and cause intense pain.
Physical Hazards: Corrosive and Reactive
The corrosive nature of triflic anhydride doesn’t play favorites. Glass and plastic can both come under attack, and standard lab gear sometimes fails. Mix it with water, and you get a violent reaction, not to mention clouds of toxic gases. Triflic anhydride reacts hard with nucleophiles like water, alcohols, or even the ambient moisture in the air. The reaction can heat up quickly, spill out of control, and leave behind a mess that takes hours to clean up safely.
Fire’s not far away either. Many organic solvents don’t mix well with this chemical, and a simple error like using an incompatible flask can lead to unexpected exothermic events. Stories float around labs about close calls—heating gone too far, sudden decompositions, and even minor explosions. Each story comes with a lesson: nothing about triflic anhydride can be taken lightly.
Environmental Concerns
Waste left behind after a reaction often contains active triflic anhydride or its acidic fallout. Pouring it down the drain, which in itself sounds reckless, lets strong acids leach into water systems and threaten aquatic life. There’s a real cost to these mistakes. Waste treatment in most places can’t neutralize the level of acidity and reactivity triflic anhydride brings. I’ve seen labs double- and triple-check waste containers for leaks, labels, and compatibility before letting them out the door.
Knowledge, Training, and Responsibility
Working with harsh chemicals comes down to training and respect. Shortcuts—missing PPE, skipping the fume hood, leaving a bottle open—add up to a bad day or worse. High-level synthesis tasks shouldn’t land in the hands of new students or undertrained staff. Even for experienced researchers, refresher courses and open discussions about near-misses save lives.
Facilities that use triflic anhydride owe their safety records to regular drills, signage that leaves nothing to chance, and a culture where asking questions isn’t frowned upon. Peer review in the lab should include talking through what could go wrong. Real-time safety data, easy access to neutralizing agents, and clear, up-to-date procedure sheets matter far more than formal compliance checklists.
Looking for Safer Paths Forward
Research doesn’t always need to rely on the harshest reagents available. Green chemistry pushes for alternatives, and some new syntheses find ways to skip triflic anhydride altogether. The move toward less hazardous reagents isn’t just about comfort; it’s about sending more scientists home safe at the end of each day. Until the day comes that triflic anhydride gets replaced in most protocols, treating it with the same respect as the harshest acids or bases in the lab stays crucial.