Rethinking the Role of Trifluoroacetaldehyde Ethyl Hemiacetal in Modern Chemistry
Where Trifluoroacetaldehyde Ethyl Hemiacetal Comes From
Trifluoroacetaldehyde ethyl hemiacetal didn't suddenly pop up one day in a research lab. It has a history, and its development tracks closely with the rise of fluorinated compounds in the chemical industry. As organic chemists started harnessing fluorine’s tendency to reshape molecules, the trifluoromethyl group, in particular, drew lots of attention. Ethyl hemiacetals, with their mix of stability and reactivity, began popping up in specialty chemical manufacturing sometime in the second half of the twentieth century. My own first run-in with this molecule took place during a fragrance formulations project, highlighting how compounds like this keep wending their way into new research areas, from synthesis to applied sciences.
A Quick Glance at the Compound
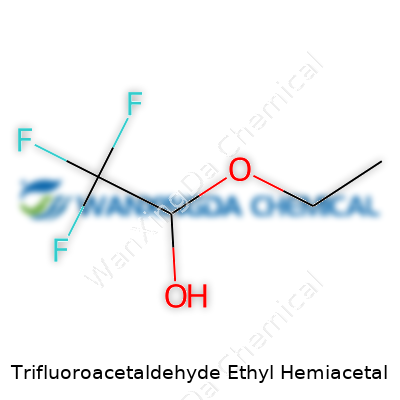
Trifluoroacetaldehyde ethyl hemiacetal balances stability and reactivity—a trait valuable for people trying to build more complex molecules. Its systematic name spells out the structure: you have an ethoxy group and a trifluoromethyl base, linked by an acetal functional group. Chemists often draw on this framework, finding that the tri-fluorination brings both chemical resistance and unique electronic effects. Folk in drug development or materials science spot these characteristics and explore the possibilities for new therapeutic agents, agrochemicals, or specialty solvents.
The Body and Behavior of the Molecule
Anyone who's handled this chemical can sense its subtle volatility. The clear, colorless liquid carries a sharp, ether-like odor, hinting at the presence of fluorine and the chemical’s modest boiling point. Density hovers north of one gram per cubic centimeter. Despite the seemingly innocuous appearance, one crack open of the cap in a poorly ventilated lab sets off both the nose and the warning systems. Most notable of all: trifluoroacetaldehyde ethyl hemiacetal resists water, thanks to the shielding from the three fluorine atoms, which also limit unwanted side-reactions.
Labeling, Handling, and Technical Standards
Labels often focus on flammability and volatility. If you've worked around halogenated acetals, you know personal protective equipment isn’t a mere suggestion. Gloves, goggles, and ventilation can’t be skipped, especially with regulatory bodies cracking down on fluorinated substances due to environmental persistence. Data sheets rarely underplay these points, since folks in labs and on factory floors depend on clear warnings. Disposal links up tightly with local rules, given the environmental controversy surrounding persistent fluorinated organics.
How This Chemical Comes Together
Preparative methods rely on classic organic chemistry: typically, an acid-catalyzed reaction between trifluoroacetaldehyde and ethanol does the job. Care in controlling water content proves crucial, since too much moisture swings the equilibrium and spoils purity. If you’ve ever tried it, you know fractional distillation sorts out the product, but even a slip-up with the temperature can mean extra hours chasing after clean fractions. Scale-up in industry never comes as easy as the beaker-scale version, so process engineers must tweak catalyst loads, temperature ramps, and cleanup protocols accordingly.
Chemical Reactions and Tweaks
This compound rarely sits still. The hemiacetal group waits for the right acid or base to shift the equilibrium toward either the aldehyde or the fully acetalized form. Reductive methods strip out functional groups, while oxidative routes open up possibilities for tiny cocktails of byproducts—including hazardous ones—under sloppy control. The robust trifluoromethyl portion blocks many typical hydrolytic attacks, yet under harsh enough conditions, you see the old rules of reactivity still apply. Modification experiments keep showing up in journals from synthetic organic chemistry to applied pharmaceutical sciences.
Alternate Names, Alternate Contexts
For anyone diving into supplier catalogs or research archives, synonyms often trip up searches. Expect terms like "ethyl 2,2,2-trifluoroacetaldehyde hemiacetal," or simply "TFAA ethyl hemiacetal." The business of chemistry keeps plenty of these aliases circulating, sometimes obscuring cross-disciplinary research or making sourcing harder for small labs outside the established networks.
Safety and Best Practices
Chemists who've handled liquid fluorinated compounds recall cautionary tales: accidental spills that stubbornly refused to clean up, or headaches and nausea after an inadvertent inhale. Regulatory agencies continue adjusting standards, sometimes tightening up storage requirements and exposure thresholds based on new data about long-term toxicity and bioaccumulation. Safe handling isn’t negotiable in an environment where newer evidence points toward unknown risks. Industry veterans often rely on institutional memory: label everything, ventilate everywhere, and double-check before disposal.
Why Researchers Care
Interest in this chemical comes down to its usefulness as a building block. Organic synthesis outfits chase it for the ways it can introduce a trifluoromethyl group under gentler conditions than other reagents. Pharmaceutical developers probe its use in generating new candidates where metabolic stability depends on fluorination. Even outside the life sciences, materials researchers toy with this molecule, banking on its resistance to heat and oxidation in specialty polymers or surface coatings. Across all these areas, there’s a drive for scalable processes, sustainable feedstocks, and minimal waste.
Exploring Toxicity and Environmental Impact
Toxicity research stands out in recent years, especially as carriers of persistent organic pollutants make headlines. Early animal studies point to moderate acute toxicity with symptoms centered around central nervous system effects. Chronic exposure raises more questions than it answers, in part because the full persistence and breakdown pattern of highly fluorinated molecules remain under investigation. Regulatory attention increases every year, especially as environmental advocates spotlight the risks of "forever chemicals." For those of us who remember less-regulated eras, the warning signs ring familiar: today's miracle compound often ends up tomorrow's headline risk.
Where This Chemical Goes Next
Looking forward, the future weighs heavy on two things: improving synthetic efficiency and clarifying health/environmental risks. Industry demand points to greener chemistry, so a lot of ongoing research revolves around catalysts with less waste or the use of renewable ethanol as a feedstock. Big questions stay unanswered about long-term environmental fate, especially given the growing willingness of regulators worldwide to restrict “problematic” fluorinated molecules. Part of the path forward probably includes stronger cross-talk between toxicologists, process chemists, and product designers—to make sure that breakthrough molecules improve products without turning into lasting environmental headaches. Scientists, policy-makers, and the public must keep pushing for more data, smarter synthesis, and honest cost/benefit discussions.
Trifluoroacetaldehyde ethyl hemiacetal usually doesn’t grab headlines, but it holds a quiet spot in the world of organic chemistry. Most people have never seen it, never touched it, yet their daily lives might involve products developed with its help. My background in research labs gives me a close view of strange compounds like this—molecules that rarely leave the glassware, but make a difference every time they do.
A Niche Ingredient with Real Impact
Chemists hunt for creative solutions. In the drive to design new drugs and specialty chemicals, trifluoroacetaldehyde ethyl hemiacetal keeps showing up because its trifluoromethyl group delivers properties other chemicals can’t match. I’ve seen its value while developing small molecule drugs in the lab. Add it to the mix, and you can shift a compound’s metabolism or help it slip past enzymes that would otherwise break it down too fast. That extra resilience can turn an idea into an actual medicine, something that could give folks a better shot at recovery.
How Fluorinated Compounds Change the Game
Fluorinated intermediates like trifluoroacetaldehyde ethyl hemiacetal give medicinal and agrochemical chemists more options. That trifluoromethyl group packs a punch: it boosts lipophilicity, which in plain language means certain drugs or molecules can get into cells more easily. In my own work, switching in a trifluoromethyl group helped me dial in how a new molecule traveled through a biological system. Little changes in the lab sometimes mean big advances in the real world.
Making New Materials Possible
The reach of this compound stretches beyond pharmaceuticals. In the process of making advanced coatings, fluorinated intermediates offer durability and resistance. Labs have studied new polymers and specialty films that can shrug off water, oil, and even corrosion. Systems developed using materials made from compounds like this hemiacetal end up in electronics, airplanes, and even cookware. The applications aren’t endless, but they’re surprisingly broad, considering how obscure the starting materials look.
Factoring In Health and Safety
Chemistry offers tools, but it also demands respect. Many fluorinated chemicals stick around in the environment, and folks have rightfully raised concerns about their safety. In my years in the lab, every conversation about using a reagent like this comes with talk about ventilation, gloves, and careful disposal. Once you’ve seen what a little mistake can do—with toxic vapors or stubborn residues—it’s hard not to think about responsible usage. So the industry needs a sharp focus on reducing waste and finding safer synthesis routes, ideally limiting unnecessary emissions.
The Push for Innovation and Responsibility
Real progress in specialty chemistry should pair invention with accountability. Teams keep looking for new ways to make, use, and recycle compounds including trifluoroacetaldehyde ethyl hemiacetal. Supporting these efforts means backing well-trained chemists, ethical guidelines, and proper oversight. After all, the goal is not just clever molecules, but smarter choices that balance performance with responsibility. Knowing the backbone of these essential chemicals—and treating them with due respect—keeps science moving forward without leaving harm in its wake.
A Glimpse at the Chemistry
Trifluoroacetaldehyde ethyl hemiacetal shows up in research settings far more often than it does in household conversations, but understanding this molecule highlights the value of organic chemistry and its ripple effects throughout science and industry. Taking a closer look at its chemical identity, the formula reads as C6H9F3O2. Scientists recognize its structure as a product formed when trifluoroacetaldehyde reacts with ethanol, resulting in a hemiacetal. On paper and in 3D models, this molecule brings together a trifluoromethyl group (CF3–) attached to a central carbon, and an ethoxy group (from ethanol), forming a linkage that creates the hemiacetal center. This “in-between” pattern isn’t just a chemical curiosity—it represents a useful checkpoint in many synthetic schemes.
How Structure Drives Behavior
My own experience with chemistry labs taught me to respect the role of every atom in a molecule. The trifluoromethyl group isn’t just a decorative add-on; fluorine atoms draw electron density away, making the central carbon more reactive. This quality shows up strongly when working within pharmaceutical or agrochemical development, where fluorinated compounds can resist metabolism longer, or change the way the molecule binds to enzymes or pests. The hemiacetal structure itself stands out for its lability. Under certain conditions, especially in the presence of acids or bases, hemiacetals revert to aldehydes and alcohols. This reactivity is useful if you want to trap or release the original aldehyde without harsh conditions—a trick that comes in handy during multi-step syntheses.
Why Should Anyone Care?
At first glance, trifluoroacetaldehyde ethyl hemiacetal may sound like the stuff of a textbook, but ignoring compounds like these skips over foundations behind much of the innovation in materials and drug development. Fluorinated molecules, often starting from simple building blocks such as this, turn up in things like MRI contrast agents, blood substitutes, refrigerants, and life-saving medicines. The chemical makeup and structure control everything from how the compound dissolves to how long it sticks around in the body.
There’s a lesson here for anyone pondering the influence of molecular tweaks: swapping a hydrogen for a fluorine, or tacking on an ethoxy group, creates ripples that reach beyond the beaker. Simple changes at the atomic scale can lead to safer, more effective medicinal compounds, or more targeted crop protection products. My years in the lab have shown that picking the right building block often separates a good solution from a great one.
Rising Challenges and Smarter Solutions
One issue worth considering is the safe handling and disposal of fluorinated chemicals. They tend to stick around in the environment, as tough-to-break-down “forever chemicals.” These compounds build up in soil and water, raising questions about long-term exposure. Researchers need to plan ahead with greener chemistry tools, designing molecules with the whole lifecycle in mind. At the bench, choosing conditions that avoid generating excess waste or hazardous byproducts keeps risks in check. Collaborating across chemistry, toxicology, and environmental science has sparked breakthroughs in breaking down tough fluorinated pollutants, but there’s plenty more work ahead.
Trifluoroacetaldehyde ethyl hemiacetal sits at a crossroads between innovation and responsibility. Chemistry drives discovery, but every new molecule demands fresh thinking about long-term impacts. The more people inside and outside the lab understand these foundations, the better prepared we’ll be to use them wisely.
Why This Chemical Matters
Working with chemicals that have long, complex names always makes me pause. Trifluoroacetaldehyde ethyl hemiacetal might not appear on every shelf, but in research labs and industry projects tied to pharmaceuticals or advanced materials, it finds its place. Such chemicals often end up at the center of new syntheses or specialty processes. Whenever I have handled reactive organofluorine compounds, one thing stands clear—slipping on safety routines for even one moment sets up everyone nearby for a bad day.
Risks Are Never Just on Paper
Many of us learn quickly that hazard labels only scratch the surface. This compound can release vapors that irritate the nose, eyes, or respiratory system. Some chemicals with similar makeup have shown unusual reactivity, and experience tells me that assuming anything “probably safe” can lead to real trouble. No one working with small molecules wants to trade a short task for a weeks-long cleanup or time lost to burns and headaches.
Safe Storage Is a Team Effort
Shortcuts in storage get people hurt. I have seen what happens to chemicals left on a sunny windowsill or in a damp storeroom: containers bloat, labels peel, and people start guessing. Storing Trifluoroacetaldehyde ethyl hemiacetal in a dry, cool place helps prevent unexpected degradation and pressure build-up. Standard chemical cabinets away from direct sunlight and heat sources work well in most labs. It’s not only about protecting the chemical itself—it’s about keeping fumes away from others, especially staff or students unfamiliar with organofluorine compounds.
Using sturdy, compatible containers keeps spills and leaks rare. Fluorinated solvents have a way of finding the one weak spot in old plastic, so glass or high-quality PTFE-lined containers make sense. I make it a habit to double-check seal integrity rather than trusting faded inventory notes. Anything cracked goes straight to hazardous waste—no argument.
Handling Never Means “Just Pour It In”
Pulling out a bottle of this chemical makes me think twice about opening it near any open flames or strong bases. Vapors from volatile acetals drift easily. Good ventilation pulls the invisible hazards away and gives everyone breathing room—literally and figuratively. Fume hoods and chemical splash goggles aren’t negotiable when dealing with compounds that can irritate eyes or skin. I’ve seen the difference that gloves and a meticulous approach make: one careful chemist averts a crisis while a rushed colleague sets off an alarm.
Every lab team should practice small spills and emergency protocols, especially for liquids that can vaporize quickly. Relying on absorbent pads, containment trays, and well-placed spill kits keeps minor slip-ups from ballooning into full evacuations. I once saw a newcomer wipe up a spill with a regular towel, not realizing the chemical could cut through it—instead of just cleaning, he set off fumes. Training isn’t a luxury, it’s a baseline.
Clear Communication Keeps Us Safer
Labeling every bottle with the full chemical name and its hazards gives newcomers and seasoned researchers alike a fighting chance at safety. It sounds basic, but during inventory checks, I’ve seen more than one unmarked container squeeze through because “everyone knows what’s in there.” No one knows everything, and emergencies don’t wait for the right person to show up.
For labs or storage rooms where organofluorine compounds appear, regular audits and honest conversations about near-misses build trust and help prevent accidents. A “see something, say something” mindset does more than a binder full of safety sheets.
Steady Vigilance Beats Any Manual
No amount of paperwork matches the value of hands-on respect for chemicals like Trifluoroacetaldehyde ethyl hemiacetal. Every sharp nose, steady hand, and honest conversation adds up to safer research and fewer regrets. It’s not just about checking a box for compliance—it keeps the work, and the people behind it, going strong.
Small Chemistry, Big Questions
Trifluoroacetaldehyde ethyl hemiacetal doesn’t pop up in everyday chat, but it makes its way into labs and sometimes into broader industry circles. Any chemical with “trifluoro” in the name deserves respect, since fluorine can change how molecules behave toward living things and the environment. Instead of bowing to complicated jargon, let’s break down what matters: Should anyone working around this substance worry about health, safety, or long-term problems?
What Stands Out About Its Toxicity?
Few mainstream sources hand out precise toxicity reports for trifluoroacetaldehyde ethyl hemiacetal. That’s part of the problem—if thorough studies haven’t been published, there’s less guidance for the real world. I’ve seen how rare chemicals get glossed over in small quantities, folks assuming there’s no real risk because the data pile feels small. That’s dangerous thinking.
You can’t ignore the legacy of related trifluoro compounds. Fluorinated chemicals often persist far longer in the environment than most people expect. Some, like PFAS, have made headlines for sticking around in water, soil, even human blood. Most experts urge caution for downstream effects, precisely because these molecules sometimes dodge the body’s natural defenses. It’s not just about immediate burns or coughs—it’s the potential for chronic exposure, unknown breakdown products, and unexpected health impacts over time.
Weighing Exposure and Precaution
The threshold for treating something as hazardous should come down to more than just a documented LD50 in rats. Many folks working in labs or handling obscure chemicals focus on short-term health symptoms. Skin redness, respiratory irritation, that sort of thing. Yet the story with many fluorinated compounds is written decades later, after repeated small exposures stack up. That’s not to say everyone using this hemiacetal needs panic, but it calls for a little skepticism about sweeping it under the rug as “generally safe.”
The hazard classification for chemicals often changes as better studies roll in. Governments like the EPA or ECHA often update their stances once environmental impacts become clearer. In this case, I haven’t found trifluoroacetaldehyde ethyl hemiacetal on major restricted lists yet. Lack of regulation doesn’t mean something is harmless—it can mean nobody has done the legwork to check.
The Safer Path
Personal experience in a busy research lab taught me the hard way that hazard assumptions make quick enemies. Gloves, eye protection, working in a hood—these aren’t optional accessories when dealing with compounds carrying fluorine. Company policy usually demands an updated Safety Data Sheet on hand, but public ones often miss recent literature or don’t dig deep enough. Leaning into a cautious approach protects people now, not just data for future study.
Better answers come from running actual studies: animal toxicity, environmental persistence, breakdown in water and air. The chemical community has a responsibility to get ahead, not wait for trouble to show up years later. It pays to treat trifluoroacetaldehyde ethyl hemiacetal with care, even if the full picture isn’t clear yet.
Moving Forward Without Blind Spots
Sometimes science offers half an answer, and the right call means not gambling on the missing half. Until clear, thorough research builds a solid safety consensus, nobody loses by respecting chemical uncertainty. Physical barriers, smart procedures, and regular reviews of emerging data won’t slow down meaningful work—they make sure the work doesn’t come at too high a cost.
Big Chemistry, Small Access
Plenty of folks outside of chemistry labs have probably never heard of Trifluoroacetaldehyde Ethyl Hemiacetal. This colorless liquid shows up where organic chemistry gets serious—especially for groups working with fluorinated compounds. In my time poking around both academic and industrial circles, I learned that the path to compounds like this one is rarely straightforward. While big name vendors display polished catalogs, getting your hands on something specialized like this has its quirks.
Picky Purity and Niche Supply
Purity always comes up early in the conversation. In the world of fluorinated chemicals, impurities sneak in almost too easily. For most reputable chemical suppliers, you’ll see listed purities touching 97% or higher, which counts as a decent minimum for research and some industrial runs. Look at Sigma-Aldrich, TCI Chemicals, or Alfa Aesar—these outfits maintain robust quality controls. I’ve seen researchers double check purity with their own NMR or GC/MS, especially for sensitive work, since even a one-percent contaminant can ruin a day’s project or throw off an entire study.
Barriers in Buying
This is not the kind of compound anyone clicks to buy on a whim. Strict regulations surround fluorinated chemicals—partly for safety and partly to keep track of what lands in sensitive research. Anyone looking to purchase will need a recognized lab or industrial address and the right documentation on hand. Some sellers go a step further and require end-user agreements or a letter of intent. As a chemistry grad student, I watched professors send emails back and forth, confirming uses and securing signatures instead of just adding to a cart.
What Matters to Buyers
Researchers need transparency—certificate of analysis, date of manufacture, lot number, and clear points of contact for technical questions. In my own lab days, even for compounds of less complexity, a paper trail made troubleshooting and troubleshooting much easier. Beyond stockroom ordering, support from a supplier—someone on the other end of the phone or email—makes all the difference when timelines get tight or shipments face customs holdups.
Smart Sourcing Steps
Anyone hunting this hemiacetal should start by listing university and industrial vendors with global reach, such as Sigma-Aldrich, Fisher Scientific, or TCI. Checking regional regulations keeps things smooth, especially given rules in the US, EU, and China that affect many fluorine-based chemicals. Trying to save money by searching obscure online shops usually opens doors to headaches—counterfeits, missed documentation, or flat-out scams. Every chemist I know has a story of a botched order from an unreliable source. Stick with vendors who back up purity with data and answer hard questions.
Long-term Fixes
Access and transparency go hand-in-hand. As more researchers push into fluorine chemistry, pressure mounts on suppliers to keep up quality and clear documentation. Universities, contract labs, and small businesses should work together and demand more open pricing, real-time inventory info, and better regulatory guidance. Having sat through more than a few supply hiccups, I believe patience and paperwork win the day, but the industry could ease the process by removing some of the red tape and jargon that scare off new innovators. If you play by the book, chemistry moves forward—and safety stays high.