Understanding Trifluoroacetic Anhydride: A Deeper Look at Its Evolution, Use, and Future
Historical Development
Chemistry never stands still. The advent of trifluoroacetic anhydride marked a clear shift in how researchers and manufacturers address the need for strong acylating agents. It grew out of the search for reagents that would both reliably acylate sensitive substrates and let chemists avoid some of the byproducts and hazards tied to older chemicals. Since the mid-20th century, labs with an eye on peptide bonds, protecting groups, and advanced organic syntheses have put trifluoroacetic anhydride at the table. Researchers watched as its adoption spread from small batch syntheses to broader industrial settings. Even now, its history keeps growing, shaped by the push for greener reactions and streamlined protocols. In my own work, the shift from less selective reagents to more finely tuned agents like trifluoroacetic anhydride signaled a turning point toward higher yields and cleaner separations, not just for specialty compounds but for large-scale pharmaceutical work as well.
Product Overview
You can’t talk about modern lab work without recognizing the reach of reagents like trifluoroacetic anhydride. It shows up as a clear, colorless liquid, easily spotted on a shelf where other acylating agents crowd together in glass bottles. What sets it apart is the way its trifluoromethyl groups drive reactions that most regular anhydrides struggle to complete. Reliability counts when you need to acylate alcohols, amines, or even water-sensitive intermediates. In my experience running multistep syntheses, it often delivers results where more temperamental reagents falter. That makes it a go-to choice for both classic transformations and the tweaking of cutting-edge reaction conditions.
Physical & Chemical Properties
Trifluoroacetic anhydride doesn’t hide its strength. With a boiling point just above room temperature and a strong, acrid smell that quickly reminds you of its reactivity, it gives clear notice to chemists. Its molecular structure, with two trifluoroacetyl groups jammed onto an anhydride, cranks up the electron-withdrawing power. That boost in electrophilicity lets it snatch up nucleophiles that might shrug off weaker agents. Handling it, I watch for the way it slices through organic layers and speeds up sluggish reactions. In a well-ventilated hood, its volatility demands respect—a simple spill can clear out a lab faster than any alarm.
Technical Specifications & Labeling
Proper labeling stands between smooth workflow and costly mistakes. My labs mark every bottle of trifluoroacetic anhydride with warnings that reflect its reactivity, volatility, and potential to harm exposed skin or lungs. Labels cover concentration, container material, and stability guidelines, all built on years of trial and error. Old timers in the lab tell stories about cracked seals and runaway exotherms—charts and safety icons reinforce those lessons for younger chemists. The details—batch purity, moisture content, recommended storage temps—appear as plain language guides, not just regulatory requirements. This helps keep everyone on track, especially when scale-ups raise the stakes.
Preparation Method
Producing trifluoroacetic anhydride usually involves either dehydration of trifluoroacetic acid with agents like phosphorus pentoxide or combinations of acid chlorides and bases. The raw materials themselves reflect a story of scale, with bulk trifluoroacetic acid trickling down from earlier fluorination work. In settings I’ve seen, the process calls for precise temperature control and skilled distillation—it’s too easy to lose yield or create dangerous byproducts without careful hands. Today, companies focus on improving yields, cutting waste, and finding safer dehydration reagents. These aren’t just environmental selling points; tighter methods mean lower costs and more consistent product quality for everyone down the line.
Chemical Reactions & Modifications
This anhydride’s appeal comes straight from the way it takes on acylation and activation reactions. It doesn’t just hand over an acyl group—it often changes the course of a synthesis by suppressing side products and allowing for milder conditions. In my own peptide chemistry projects, it handled both tricky N-acylations and the removal of protection groups that would choke under other conditions. The possibility to modify reaction pathways, especially for particularly sensitive pharmaceuticals or specialty polymers, makes trifluoroacetic anhydride a favorite for method development. Its predictability and force in the lab give experienced chemists the confidence to push boundaries in both published research and proprietary industry settings.
Synonyms & Product Names
Chemical work runs thick with jargon, and trifluoroacetic anhydride answers to several names, including TFAA and trifluoroacetyl anhydride. Each label brings its own audience—regulatory filings, catalog listings, or older literature. This spread of synonyms doesn’t show confusion; it simply reflects decades of research and commerce. In my shelves, I see both vintage glass stoppers from European suppliers and modern plastic drums labeled for high throughput, proving how widely the name travels and who depends on it.
Safety & Operational Standards
Every veteran scientist learns to treat trifluoroacetic anhydride with a blend of respect and vigilance. Even the smallest exposure brings risks—splashes on skin, harsh fumes in the air. Staff must stay current on handling protocols, from double-gloving to routine checks on ventilation. Fire suppression, spill containment, and clear evacuation plans aren’t luxuries, they form the backbone of safe, uninterrupted operations. I have seen what happens when training falters—minor incidents turning into major headaches. Investing in education, strict personal protective equipment, and prompt maintenance cuts both risk and downtime. I always argue for periodic drills over complacency, since this keeps safety part of the lab’s daily rhythm, not just a line in the manual.
Application Area
Trifluoroacetic anhydride enjoys a wide reach—its reactivity underpins the synthesis of agrochemicals, pharmaceuticals, dyes, and specialty polymers. Drug discovery teams often rely on it both for routine functional group manipulations and as a route toward new molecular scaffolds. Its speed and selectivity pay off in combinatorial chemistry, where time matters and waste adds up. In my collaborations with industry and academia, I’ve seen its use expand to areas like advanced material science and microelectronics. Researchers value how quickly it unlocks access to building blocks that feed into everything from anti-cancer agents to high-tech coatings. That range ensures its place across sectors, through both invention and day-to-day production.
Research & Development
Ongoing improvement marks the R&D story for trifluoroacetic anhydride. Teams keep searching for greener synthetic routes, better recycling protocols, and higher selectivity in challenging organic transformations. My colleagues always keep an eye out for process tweaks—greener dehydrating agents, energy savings from alternative distillation, and integration into continuous flow systems. Laboratory innovation translates into industrial advantage—a fact that shapes grant proposals and startup roadmaps alike. The need for sharper analytical techniques grows each year, as regulators and commercial customers expect both purity and documentation. Those who keep up with these trends tend to gain not just scientific, but also market leadership.
Toxicity Research
Toxicologists follow the trail of trifluoroacetic anhydride as it moves from lab bench to waste stream. Acute exposure can burn skin, irritate lungs, and leave health problems that linger longer than a spill. More subtle effects—especially from long-term, low-level contact—raise fresh questions. Institutions have invested in tracking metabolic breakdown, exposure thresholds, and the best techniques for neutralizing waste. Scientists busy with routine benchwork rarely see the full picture; sharing up-to-date findings with all users keeps real-world risks in focus. I’ve made a habit of checking toxicology updates at the start of every project, since new data sometimes means new safety steps, or even calls for substitute reagents in sensitive settings.
Future Prospects
Every year, the horizon for trifluoroacetic anhydride shifts, shaped by tighter regulation, broader applications, and a growing call for sustainable solutions. Green chemistry ramps up pressure to recycle reagents and drive reactions with less environmental impact. The search for drop-in replacements and biodegradable analogs moves forward, but so far, this anhydride holds onto a core role in synthesis for both research and large-scale production. Looking ahead, progress will depend on blending strong process safety, sharper analytical technologies, and smarter waste management. None of these advances work in isolation—only by pushing for improvements at each stage, from manufacturing to disposal, does this chemical keep its place in modern science and industry. My own hope is to see new approaches that let labs of every size enjoy the unique benefits of trifluoroacetic anhydride while steadily dialing back the health and environmental risks for the next generation of chemists.
More Than a Lab Shelf Staple
Step into a chemistry lab at a university or a pharmaceutical company, and trifluoroacetic anhydride grabs attention. Drawn from my early days in research, this chemical never goes undiscussed for long. Its role fuses the world of synthesis and the real needs of drug makers, crop scientists, and diagnostics developers. Laboratories reach for trifluoroacetic anhydride to push reactions to completion, speed up processes, and do so with fewer byproducts.
Driving Drug Discovery and Research
If you look at drug development, trifluoroacetic anhydride earns its keep. Synthesis routes without this reagent often stall, drag out, or spit out gunk that makes purification a nightmare. In peptide chemistry, coupling and protecting groups need split-second decisions—trifluoroacetic anhydride swoops in for these. Peptide chemists rely on it because it “activates” acids for easier bond formation. Cutting corners with other reagents can wreck yields or produce a grab bag of isomers. Frustration runs high when a six-figure molecule breaks down or stalls; with trifluoroacetic anhydride, reactions run quicker and cleaner.
Pushing the Boundaries of Crop Science
Fields of wheat and soybean benefit, too. Pesticide and herbicide developers leverage this chemical to build active compounds or modify existing ones. Agrochemical products regularly draw from small tweaks in functional groups to boost effectiveness or reduce toxicity. For those in the life sciences, trifluoroacetic anhydride shows its value in making molecular probes sharper, more selective, or easier to tag for detection.
Clear Results, Confidence in Quality
Labs can't afford high error rates. Pharmaceutical companies face pressure to produce compounds at scale, with safety and regulatory demands growing each year. In organic synthesis, reproducibility is king. My own experience reminds me that a single off-day with the wrong reagent can turn a whole batch unusable, wasting time and funds. Trifluoroacetic anhydride tends to do its job reliably, slashing mistakes and missing side products that could haunt downstream processes. The stakes rise when hard-won research grants or product launches depend on every batch passing muster.
Risks Demand Respect and Training
That said, trifluoroacetic anhydride doesn't play nicely with carelessness. Proper ventilation, gloves, and goggles are a must. Cutting corners leads straight to burns or worse when handling reactive chemicals. Chasing efficiency never justifies sloppy methods. Trained chemists keep risk in check, and quality controls back them up. Striking a balance between productivity and safety stays central, especially as labs push for faster development cycles.
Pushing for Greener Chemistry
There’s a push for greener solutions in modern labs. Trifluoroacetic anhydride, while effective, draws attention for environmental persistence. Waste management and alternatives rise up the list of priorities for younger chemists and regulatory bodies. Some labs explore enzymatic or water-based routes, though these don’t always match trifluoroacetic anhydride’s speed or selectivity. Funding research into replacements deserves more attention, as the industry faces tighter regulations and a changing climate.
Why It Matters Now
Chemistry doesn’t stay static. Each year brings tougher projects and fresh safety regulations. Trifluoroacetic anhydride sticks around because it works, but it also reminds us labs need both reliability and responsibility. Anyone working to bring new drugs, safer crops, or more sensitive diagnostics to market owes a debt to those tools that get the job done, but the future calls for ongoing review, innovation, and better stewardship—lessons I learned not from textbooks, but from time spent at the bench, sleeves rolled up and fume hoods humming.
Trifluoroacetic anhydride doesn’t sound like something most people run across every day, but in many chemistry labs, it’s about as familiar as coffee. This colorless liquid gets used for all sorts of useful reactions—especially when chemists want to tweak molecules just so. I’ve spent enough hours in research spaces to know its sharp, suffocating odor and the sense of urgency that comes whenever anyone cracks open a bottle.
The Right Storage Space: Practical Experience Speaks Loudest
Most accidents don’t unfold as a single event; they build up over time, usually from little lapses and overlooked details. In the crowded cabinet of reactive chemicals, trifluoroacetic anhydride has taught many a lesson the hard way. It reacts violently with water, including the humidity you barely notice on a muggy afternoon. Fumes seep out and corrode neighboring containers and anything metal. Once, I witnessed lab glassware turn cloudy and brittle after someone stored this chemical near sink valves, showing just how aggressive those vapors can be.
In any reasonable lab, you’ll find this chemical sealed up tightly in its original container, with a chemical-resistant cap and a clear hazard label. It rests on a dedicated shelf, far from acids, bases, alcohols, and especially water. Storage cabinets meant for corrosives—metal-free and lined with thick plastic—offer the only real peace of mind. Keeping it at a cool temperature (well below room temperature) slows down its propensity to decompose and lash out with fumes. A well-ventilated storage space keeps any stray gases from building up to dangerous levels.
Training Saves Labs and Lives
Proper storage isn’t just a line in a manual. Supervision makes the biggest difference. Rushed workers sometimes stash containers wherever there’s room. After a spill at my former research building, our lab manager started posting monthly chemical safety reminders and required everyone to mark the date bottles were first opened. The result—fewer mysteries about chemical age and a lot less stress when audits rolled around. Regular oversight doesn’t just protect property; it stops the don’t-touch-that emergencies before they start.
Asking users to always wear splash goggles, gloves made of fluorinated polymer, and a fitted lab coat still matters, even in storage. If you reach for a chemical in a hurry and the cap isn’t tightly secured, those fumes can cause burns or lasting damage to your airway in seconds. Fast access to an eyewash station and plenty of ventilation near storage helps reduce the risk of accidents turning into disasters.
Concrete Steps Toward Safer Practices
I’ve seen too many labs cut corners to save time. Still, spending a few minutes to double-check that trifluoroacetic anhydride stays in its place—dry, cool, ventilated, tightly closed—pays off. Encouraging a culture that values not only following, but understanding safety rules protects every person in a facility. Moving away from laissez-faire attitudes and putting in the effort to educate every lab member makes safe storage everyone’s responsibility, not just the job of one tired person at the end of the day.
The chemical’s utility doesn’t excuse ignoring its hazards. When science gets moving at full speed, a little diligence at the storage shelf keeps progress from turning into regret.
Why Trifluoroacetic Anhydride Means Business
Ask anyone who’s spent time in a chemistry lab, trifluoroacetic anhydride turns even a routine synthesis into a high-stakes operation. It’s reactive, it’s unforgiving, and skin or eyes aren’t friends of this stuff. I remember the sharp, almost acrid smell that drifts up while opening a fresh bottle. That scent means business — chemical burns, breathing trouble, the sort of headaches you don’t shake off by stepping into the hallway. Safety isn’t red tape in this lab; it’s the only way home in one piece.
Gloves, Goggles, and More: Stop Shortcutting PPE
Working with trifluoroacetic anhydride, I learned that double-gloving with nitrile makes sense because this reagent can eat through regular latex in no time. Safety goggles are a must, but a splash-resistant face shield goes the extra mile — one misstep, and you don’t just lose your composure, you risk eye damage for life. I’ve watched newcomers underestimate the fumes, skipping lab coats or scrimping on gloves, and that isn’t just carelessness, it’s a gamble nobody wins.
Fact: Even a tiny spill can start to corrode metal benches, create toxic fumes, and may trigger violent reactions with water or alcohols left on glassware. Good labs run on discipline and habits: labeling secondary containers, never storing anhydride near acids or alcohols, and locking bottles away from sunlight and moisture. The safety data sheets don’t exaggerate this stuff. One wrong move, and you end up calling the environmental health team.
Airflow and Storage: Not Negotiable
I always check the fume hood before starting. Just hearing the exhaust whir doesn’t guarantee it’s moving enough air. Trifluoroacetic anhydride vapors don’t just smell bad — they can cause pulmonary edema, and there’s nothing theoretical about that. I only ever open or use this reagent with the sash pulled low, hands inside, and glassware checked for cracks. Every storage shelf gets a weekly wipe-down; I learned that leaks sneak up in summer when bottles sweat from temperature swings.
No matter how rushed a project gets, storing trifluoroacetic anhydride inside its original bottle with a good seal stops dangerous leaks and contamination. Leaving even a drop exposed risks a chain reaction with the moisture in the air. Combining this chemical with incompatible materials led to lab evacuations more than once. Experience teaches that proper containment isn’t fear, it’s respect for consequences.
Emergency Planning: Always Assume the Worst
Most lab accidents with this chemical happen to people who let their guard down. Training isn’t a box to tick; the person next to me could be on deck to grab the eyewash station or activate the emergency shower. We review plans, walk through spill protocols, and everyone knows where to find the backup gloves, absorbent pads, and respirators. Quick response makes the difference between a close call and a hospital visit. Local fire authorities remind us that trifluoroacetic anhydride releases toxic gases if it burns—so we never store it anywhere fire could reach easily.
There’s talk about better chemical engineering controls, self-sealing bottles, and automated syringe systems, but most safety hinges on ordinary people keeping their head. Ultimate protection comes down to preparation and a healthy fear of what a clear bottle can contain. After years at the bench, respect for trifluoroacetic anhydride isn’t old-fashioned, it’s earned.
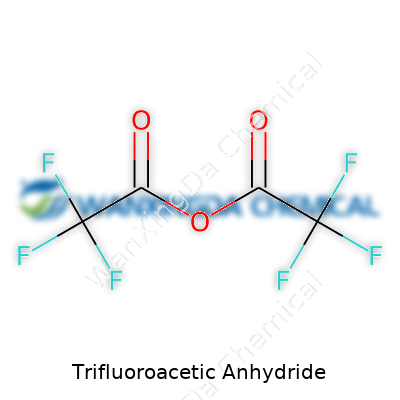
Understanding the Backbone: C4F6O3
Chemistry tends to scare off a lot of folks, but everything becomes a little less intimidating when you can break it down. With trifluoroacetic anhydride, you’re looking at a chemical with the formula C4F6O3. Each piece of that formula tells a story. Four carbons, six fluorines, and three oxygens—packed into a single molecule that serves all sorts of roles in both labs and industries.
Somewhere along the journey of learning about chemicals, I realized it pays off to really know what goes into the formulas we talk about. Trifluoroacetic anhydride proves that point. This compound doesn’t stay quietly on the shelf—its formula hints at its punchy reactivity. Chemists use it when they need to tweak molecules, especially when handling sensitive stuff. In particular, its popularity grows out of the strength those highly electronegative fluorines bring to the table. They don’t just play with electrons; they can shift entire reactions in different directions.
Beyond the Textbook
Due to its unique formula, trifluoroacetic anhydride stands out as a tool in organic synthesis. I remember my first real exposure to this compound happened during graduate research. Making the leap from textbook knowledge to hands-on chemistry highlighted how a precise formula makes the difference between a successful experiment and a total flop. Its formula means it acts as a powerhouse acylating agent, turning mild reactions into efficient, clean workflows. Industries dealing with pharmaceuticals or advanced materials keep it in rotation, not just for its reactivity but for the predictability that formula brings.
Ignoring what each atom brings to a compound increases the risk of unexpected side effects—think of runaway heat or even severe corrosion. Three well-placed oxygens make it quite reactive toward water, generating trifluoroacetic acid as a byproduct. This byproduct can be a blessing for some reactions, but generate headaches in others. Being aware of the formula lets chemists plan out their entire procedure, right down to the last clean-up step. Safety considerations pop up everywhere, but risks really shrink when you start with the right information.
Pushing Toward Transparency and Responsible Chemistry
I’ve watched the field of chemistry push for more transparent communication about what’s in a bottle. Precise chemical formulas cut through misunderstandings and let professionals compare notes across the world. That shared language lays the groundwork for responsible use and disposal, especially with active reagents like this one. People depend on clear rules and tested protocols, but those rules rely entirely on understanding the basics—starting with formulas.
Education around compounds like trifluoroacetic anhydride should push students to connect formulas to real-world impact. Interactive lessons, clear labeling, and direct demonstrations all play a role in building confidence and skill. As we look toward a future with advanced therapeutics and greener technologies, the next generation of chemists need solid building blocks to keep moving forward. C4F6O3 doesn’t just sit on a label; it calls out for careful planning and a respect for what those atoms can do.
Anyone who’s mixed up a reaction with trifluoroacetic anhydride knows the feeling of anticipation. This strong acylation agent pushes reactions forward with real punch, and that’s exactly what makes its compatibility a big deal. The thrill fades fast, though, when something in the flask doesn’t play nice. Trifluoroacetic anhydride, or TFAA for those who work with it regularly, tends to clash with many chemical classmates.
Why Compatibility Matters Here
Most organic chemists run into TFAA at least once. It’s a staple in making complex molecules, beefing up an acyl group, or tweaking a structure in pharmaceutical research. Mixing chemicals isn’t like making soup, though. Every drop poured together can send things sideways if they don’t get along. TFAA tears into nucleophiles—alcohols, amines, even water—sometimes too quickly. It reacts with moisture in the air, snapping up protons, and then forming up trifluoroacetic acid. You store it sealed tight for a reason.
There’s a reason you’ll never find TFAA next to basic solutions on a bench for very long. Bases make it decompose, and strong nucleophiles suck up TFAA’s acyl groups so fast the reaction can get out of hand. You want control, not an explosion of byproducts. Acids won’t actually stabilize it much either, since water makes this stuff fume and break down. More than one person has learned the hard way that a wet flask means wasted reagents and a ruined experiment.
Drawing from the Lab Experience
Mixing TFAA with the wrong kind of halides, like sodium chloride or potassium iodide, can create side products or even hazardous gases. I’ve seen the aftermath of poor choice—brown gunk coating glassware and yields dropping off a cliff. One time, I watched someone add TFAA to a leftover solution that still had a strong amine present. The heat kicked up so fast we had to dunk the flask in cold water, and the fume hood next to us filled with a sharp, almost choking odor. That’s not just inconvenient—it’s genuinely dangerous.
Still, TFAA shines when coupled with carefully chosen substrates. It’s a smart tool for making esters or activating carboxylic acids. But the key is using dry, neutral solvents like dichloromethane or acetonitrile, and cleaning up every trace of water before starting. People running peptide synthesis or tweaking sensitive molecules can use TFAA’s reactivity to their advantage, pushing reactions that would otherwise stall. Even then, you avoid common bases, steer clear of moisture, and double-check everything for traces of contaminants before going ahead.
Respect and Responsibility in the Lab
With chemicals like TFAA, overconfidence can create critical problems. Training and careful habit pay off much more than shortcuts. Good lab notes save time in the long run, preventing accidental repeats of failed mixes. Institutions push for regular safety training on things like TFAA because oversight can have real consequences. In some labs, proposals for new synthetic steps must run past a safety check before heading to the bench, and that’s saved more than one researcher from a costly cleanup or a hospital visit.
Using TFAA safely doesn’t call for paranoia, just consistent respect and clean work. Know its strengths, watch for dangerous pairings, and keep water and strong bases far away. Chemistry is about creative solutions, but also learning where not to push your luck. That’s where compatibility has meaning: not just in the flask, but in every routine and piece of advice passed down from one scientist to the next.