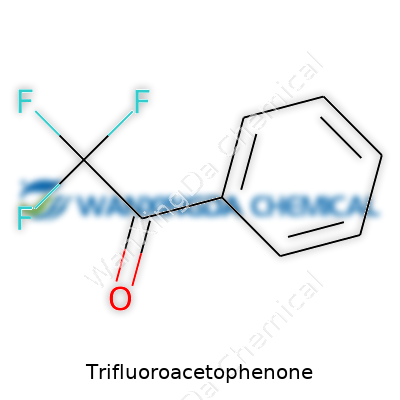
Trifluoroacetophenone: Why Chemists Keep Coming Back to This Key Building Block
Looking Back: How Trifluoroacetophenone Grew in the World of Chemistry
People working in laboratories have tinkered with trifluoroacetophenone for generations. Its story stretches back to the surge of interest in organofluorine compounds that took off through the twentieth century. Chemists searched for molecules that brought together useful reactivity and strong electron-withdrawing groups. The trifluoromethyl group, with its batch of three fluorine atoms, really stood out for its ability to pull electron density and unlock new behaviors in otherwise familiar aromatic frameworks. Trifluoroacetophenone rose up as an early example—offering both a classic phenyl ring and the punchy effect of the CF3 group right next to a carbonyl. Over the decades, its utility in synthesis, its unique reactivity, and its starring role in fluorinated research put it on the shelves and bench tops of many chemists I know.
What Makes This Molecule Tick: Physical and Chemical Properties
Take a bottle of trifluoroacetophenone off the shelf and you see a clear, colorless liquid, with a sharp, somewhat sweet odor that's hard to miss. It weighs in with a molecular formula of C8H5F3O—compact, but packing substantial chemical punch. With a boiling point near 144°C and a melting point low enough that it stays liquid at room temperature, this compound pours easily for measured reactions. Chemists keep an eye out for its volatility, which means losing some of it to the air is always a risk if the bottle sits out too long. The trifluoromethyl group really grabs electron density, shifting its carbonyl reactivity, boosting resistance to some reductions, and changing the ease with which it partners with nucleophiles compared to its acetophenone cousin. You see it showing different behavior in both standard organic synthesis and in the analytical lab, making it a favorite for reaction screening and method development.
Breaking Down Key Specs and Labels in the Lab
Labels on trifluoroacetophenone bottles always scream “Handle with Care.” Clear details about purity—often 98% or better—come standard. Labs check for water content and residual acids as even trace contamination can throw off sensitive reactions downstream. Chemists often find detailed certificates of analysis with spectroscopic data: an unmistakable 19F NMR singlet, sharp carbonyl IR absorption, and well-defined GC or LC retention times. Regulations push suppliers to track batch history and storage conditions because, like many fluorinated aromatics, this compound likes to stay dry, cool, and tightly sealed. No mistaking that whiff of volatility or the warning pictograms on the label—every researcher handling trifluoroacetophenone cares about more than just the reaction at hand; safety and documentation drive responsible use.
How Labs Prepare Trifluoroacetophenone
Chemists usually tackle the synthesis of trifluoroacetophenone through acylation routes. The Friedel-Crafts acylation sits among the classic methods, starting with trifluoroacetic anhydride or acid chloride slapped onto benzene or substituted aromatics, using a strong Lewis acid like aluminum chloride as a catalyst. The yields come high when the process runs dry and the equipment stays moisture-free. Once the acylation finishes, distillation or column chromatography cleans up the mixture. Mechanically, this type of chemistry feels approachable for most college labs yet scales well for industrial prep too. Sometimes, direct fluorination or other fluorine-introduction chemistry gets a mention, but most settle for the trifluoro group pre-installed, given the mess and cost of handling elemental fluorine. Year after year, the basic strategy changes little, underscoring the reliability of this method to deliver the goods for research and manufacturing alike.
Chemical Reactions and Useful Modifications
In my own time working with trifluoroacetophenone, the real kick comes from seeing how dramatically the CF3 group changes everyday chemistry. That stiff electron-withdrawing pull paves the way for interesting enolate chemistry—much less nucleophilic than classic acetophenones, which matters when you reach for alkylation or condensation strategies. The compound serves as a launchpad for making heterocycles, plays nicely in pericyclic reactions, and can stand as an intermediate for pharmaceutical scaffolds. Strong bases pull protons with more effort, but the payoff includes increased selectivity or access to new target molecules where trifluoromethyl groups can dictate biological activity or physical behavior. Downstream, chemists also tweak trifluoroacetophenone via reduction, halogenation, or addition reactions, getting new flavors of substituted aromatics with surprising utility. My colleagues in catalysis and material science have found that the push–pull effect of the trifluoromethyl carbonyl combination offers properties that standard ketones or non-fluorinated aromatics can't match.
Names and Aliases: Decoding What’s on the Bottle
Run a search through chemical databases and you trip over a handful of names for this molecule. The most formal label you see is 1,1,1-trifluoro-2-phenylethan-1-one. Folks call it trifluoroacetophenone for simplicity, but sometimes phenyl trifluoromethyl ketone, or TFAK, pops up in research circles. Synonyms cluster around the same core—consistent enough so misidentification doesn’t often trip people up in procurement or publication. In every stockroom I’ve visited, a quick check of the structural formula clears up ambiguity. Anyone handling the compound in an international context would do well to double-check the CAS number to stay on the right page for their compliance teams.
Building a Safer Lab: Operational Standards and Safety
You only need one spill or inhalation scare to start respecting these kinds of fluorinated ketones. Labs set strict ventilation standards and keep PPE rules in place for all organofluorine work—chemical-resistant gloves, solid eye protection, lab coats that cover skin. People keep this ketone capped tight in a cool, well-ventilated spot, with spill kits at the ready. The real risk comes from inhalation or skin contact, which can bring irritation and, depending on dose and exposure time, more serious effects. Training and signage keep accidents to a minimum. For waste streams, on-site protocols tag this type of compound as hazardous, so solvents and cleaning rags see proper disposal through certified channels. Regular audits and the growing digitalization of inventory make it easier to spot expired or degraded inventory before it causes trouble. Most modern facilities treat trifluoroacetophenone handling as a model case to reinforce safe chemical management, both to protect workers and meet regulatory compliance.
Trifluoroacetophenone in Action: Where It Makes a Difference
Beyond the inner sanctum of chemical research, trifluoroacetophenone gets plenty of play in the pharmaceutical and agrochemical industries. Medicinal chemists, chasing after greater metabolic stability or fine-tuning drug-like properties, often lean on the CF3 group as a way to adjust bioactivity, permeability, or resistance to enzymatic cleavage. Trifluoroacetophenone lays groundwork for target molecules in the search for new antivirals, anti-inflammatories, and central nervous system agents. Crop protection chemistry borrows from this expertise, as fluorinated building blocks often lend environmental persistence and targeted activity. In the realm of material science, researchers eye modifications based on trifluoroacetophenone for designing new polymers, fine-tuning dielectric properties, or even building specialty catalysts where electron-withdrawing power matters. Analytical labs appreciate its strong spectroscopic signals when developing new quantification methods, particularly using 19F NMR. Some surface science teams experiment with this compound as an anchor for further functionalization, helping bridge the molecular world to advanced coatings and nanotechnology.
In the Trenches: R&D Pushes and Challenges
Researchers walk a tightrope with organofluorine chemistry—everyone wants the performance edge, but the methods can strain resources and patience. In university labs, eco-friendly routes to trifluoroacetophenone get a lot of attention. Renewable feedstocks, milder reaction conditions, and solvent recycling programs look for a cleaner footprint. Scientists test greener acylation approaches, swap out harsh Lewis acids for catalysts based on iron or enzymes, and track their environmental impact with increasingly sharper analytical tools. Pharma and agrochemical companies invest big to leverage trifluoroacetophenone for next-generation drugs and active ingredients. The arms race for intellectual property keeps motivation high, but synthetic intermediates must meet tighter standards for trace impurities, batch reproducibility, and toxicology. Digitalization and automation change the pace—robots dose reagents, in-situ analytics guide conditions, and AI crunches data for reaction optimization. The push to do more, faster, safer, and greener keeps the compound in the conversation whenever new chemical space is explored.
Weighing the Risks: Toxicity Research Sheds Light
No one doubts that trifluoroacetophenone brings some hazards to the bench. Early studies linked similar fluorinated ketones to eye and skin irritation, and inhalation risks draw strict line in the sand. Recent research casts a wider net, probing longer-term effects and how this compound behaves in biological and environmental settings. Toxicologists have looked closely at doses relevant to laboratory and manufacturing spills—noting acute symptoms in animal testing but typically low persistence in living tissue due to rapid breakdown or excretion. Still, gaps remain, especially when you consider workplace exposure, environmental accumulation, or breakdown products. Regulators and safety committees encourage labs to track every milligram, reduce accidental emissions, and replace high-hazard solvents wherever alternatives work. As we learn more about the environmental fate of organofluorines, everyone agrees that careful stewardship and transparent reporting for the lifecycle of trifluoroacetophenone make up the best line of defense.
Where Next? Prospects for Trifluoroacetophenone
From where I stand, trifluoroacetophenone isn’t fading from relevance. Its blend of reactivity, stability, and diverse chemical potential keeps it a cornerstone tool for inventing the next batch of pharmaceuticals, agrochemicals, and advanced materials. Ongoing work sharpens the methods for safer, more responsible handling and pushes the envelope on greener synthesis. Digital tools and automation promise to squeeze more insight from every experiment, while deeper toxicity screening keeps the risks in view for the long haul. As the world asks for molecules that can perform in ever-tougher environments—whether inside a living cell or across an industrial process—trifluoroacetophenone keeps offering a powerful shortcut to somewhere new in the world of chemistry. Even as regulations tighten and green chemistry comes front and center, it stands to remain a key player wherever someone needs the unique magic only fluorinated organics can bring.
Digging Deeper Into a Peculiar Chemical
Most folks haven't run into trifluoroacetophenone outside of a chemistry lab. It sounds like the sort of compound that only matters to specialists in white coats, but the truth runs deeper. Stranger still, this molecule pops up behind the scenes in more places than many realize.
Sharp Edge in Organic Chemistry
During grad school, I found my hands stained more than once by chemical reagents that never leave lab benches. Trifluoroacetophenone often played a starring role in synthetic research, offering up its three fluorine atoms as a way to shape other molecules just so. Chemists like tools that leave a mark—trifluoroacetophenone gives reactions a push, tweaking the properties of resulting compounds by loading them up with a strong electron-withdrawing group. This makes downstream reactions behave in new ways, unlocking fresh paths for drug discovery. It’s both a mild-mannered bystander and a powerful influencer, and I’ve watched seasoned scientists light up as reactions clicked into place thanks to its involvement.
Pharmaceutical Experiments
Fluorine, especially in threes, can give a new medicine longer life in the bloodstream or block unwanted side effects. While few over-the-counter pharmacy bottles sport trifluoroacetophenone on the label, many experimental drugs trace their chemical roots back to it. It helps with structure-activity optimization, dialing in effects until the balance feels just right. Companies run millions of dollars’ worth of experiments built atop simple building blocks like trifluoroacetophenone. Whenever I chat with medicinal chemists, the race to tweak molecular properties always comes up, and their reliance on fluorinated building blocks explains how a humble chemical connects to bigger stories of health and innovation.
Shifts in Agrochemicals and Materials
Not every use orbits the medical world. Trifluoroacetophenone also figures into the development of crop protection agents. Adding that three-fluorine “tag” can help design smarter herbicides and pesticides, targeting weeds and pests more precisely. This spares crops and helps dial down chemical runoff. I remember listening to a plant biologist explain how tiny changes in chemical structure could mean fewer environmental headaches—trifluoroacetophenone plays a part in that mission.
Lab Safety and Future Questions
Like many specialized reagents, it brings risks—not every lab worker learns to handle chemicals with fluorine safely. Mishaps stick in your memory. Years ago, I overheard a scramble after an accidental spill. Proper training and respect for these tools stay important. Research journals and regulatory agencies keep hammering home the need to work safely as fluorinated organics grow more common in labs worldwide.
Looking Forward
As new drugs and cleaner agrochemicals top everyone’s wish lists, chemicals like trifluoroacetophenone will stay in demand. Transparency about supply chain, safe handling, and environmental impact matters. Chemists should keep asking tough questions, looking for smarter, greener process routes. The path runs straight through the crowded backrooms of research labs, where small molecules pull off big tricks—and trifluoroacetophenone remains one of those unsung players that keeps innovation humming.
Breaking Down the Formula
You walk into any college chemistry lab and see all sorts of glassware, aromas, and colorful liquids swirling around. One of the names that comes up pretty often in organic synthesis conversations is Trifluoroacetophenone. The chemical formula—C8H5F3O—packs a punch in its short string of letters and numbers. At first glance, it doesn’t sound flashy. But in practice, this simple formula signals a molecule that’s managed to make an impression in both classic research and specialized industry projects.
What Lies Behind C8H5F3O?
A formula like C8H5F3O doesn’t just exist on paper. It stands for a compound where a trifluoromethyl group (CF3) joins up with an acetophenone backbone. Anyone who’s tinkered with aromatic ketones knows this changes the whole behavior of the molecule. Replace plain hydrogens with fluorines, and suddenly you’ve altered the way electrons dance across the ring. These three extra fluorine atoms aren’t just for show—they pull on that cloud of electrons, making the molecule a lot less reactive toward certain attacks and a lot more helpful if you’re aiming for particular results in synthesis.
Why Fluorine Matters in Chemistry
There’s a reason chemists fuss about adding fluorine to organic molecules. Fluorine’s strong electronegativity makes the molecule tougher and a bit slippery for enzymes. Drug designers, for example, like using fluorinated groups to dodge the body’s metabolic buzz saws. With Trifluoroacetophenone, the CF3 tag acts like a chemical shield, stabilizing the core and opening doors to transformations that a plain acetophenone just doesn’t handle as well.
Where Trifluoroacetophenone Shows Up
I’ve seen this compound pop up during late-night literature searches, especially in projects on pharmaceutical intermediates and advanced polymers. Academic articles from journals like Journal of Organic Chemistry mention trifluoromethyl groups in candidate molecules for novel materials or therapies. Its use isn’t just theory; Trifluoroacetophenone comes into play in research labs stretching from Tokyo to Toronto.
When a colleague used this molecule in a fluorinated ligand project, they talked about its stubborn stability under rough reactions. That’s something researchers count on—especially when the stakes include whether your whole day’s work survives a final purification. Trifluoroacetophenone isn’t the star of a blockbuster drug, but it holds the door open for a complicated world of fluorinated chemistry.
Responsible Handling and Future Work
Working with fluorinated compounds calls for real care. Everyone in the lab knows fluorine tends to fight back if handled loosely. Personal experience taught me respectful handling policies—ventilation, gloves, and attention to what’s going down the drain all matter. The right protocols not only protect researchers but protect the wider environment from fluorinated byproducts.
Looking ahead, as green chemistry continues to push pharmaceutical and material industries toward safer and less wasteful processes, the fluorine legacy in molecules like Trifluoroacetophenone comes under sharp focus. Some labs are experimenting with routes that use fewer hazardous reagents or try to recycle fluorine sources. Change never happens overnight, especially with complex molecules that have earned their reputation for reliability in synthesis. But the discussion is there, and actions, even at the small scale, add up.
Recognizing the Risks in the Lab and Beyond
Most folks don’t cross paths with trifluoroacetophenone on a regular basis. For anyone who works in chemical labs or handles compounds for a living, seeing long names like this gets you in the habit of double-checking the label and respecting the risks. The name alone doesn’t say much, but experience dealing with chemicals that contain fluorine often means extra care. Trifluoro compounds have a reputation for being more than a little tricky if handled wrong.
Official data points to trifluoroacetophenone as an eye and respiratory irritant. It can cause coughing, watering, and burning sensations after fairly low-level exposure. No surprise, since many fluorinated aromatics have a bite to them. It’s enough to keep folks wearing gloves and eye protection every time a bottle opens. Breathing vapors, even at low doses, can leave your throat scratchy for the rest of the day.
The Gaps in Toxicity Data
People looking for a hard answer on whether trifluoroacetophenone is outright toxic don’t find a clear-cut story. There’s no famous “toxic disaster” involving it, and the available scientific research hasn’t mapped out every effect on the human body just yet. None of the standard public health agencies flag it as acutely hazardous on the level of some industrial solvents or pesticides. But the chemical structure gives a reason to hesitate. Many trifluoroacetophenone relatives—aromatic ketones with fluorine tacked on—have histories of causing trouble in high enough doses.
Years of lab work taught me never to trust chemicals just because big spills haven’t made headlines. Folks in research tend to treat any unknown as if it could do harm, at least until solid evidence says otherwise. Going by available reports, skin contact and inhalation are the two main routes to worry about. There’s not much evidence for serious chronic harm, but no one running a responsible facility wants to be the case study that discovers long-term trouble.
Treating Uncertainty with Respect
What gives chemicals like this their reputation is how fast they remind you when you cut corners. Just a whiff of the vapor drives home the importance of good ventilation. Spills hit fast, with headache, throat burn, and watery eyes. I remember a graduate student splashing a small drop on her glove, only to spend the next hour in the hallway, eyes streaming. Even basic mistakes leave big impressions.
The industry doesn’t put up with lax handling. Proper ventilation, gloves, safety goggles, and closed-toe shoes become non-negotiable habits. Emergency showers and eyewash stations in the lab aren’t just for looks. Training new chemists means drilling in the reality: treat trifluoroacetophenone and its relatives with the same respect given to more notorious chemicals, because the line between “irritant” and “lasting damage” can get blurry without warning.
Chasing Solutions in Training and Substitution
The clearest path to safety here comes down to vigilance and refusing shortcuts. Information gaps push research labs and companies to keep better records, invest in modern air monitoring, and push for quicker publication of toxicity data. Some companies look into safer alternatives, especially in processes where a different reagent can do a similar job with less risk.
Any time the science doesn’t settle the question of hazard, it’s the on-the-ground experience that shapes choices. Letting common sense guide every step tends to keep workers and students safe, even in a field where tomorrow’s information could change everything.
Simplicity Hides Risk
Trifluoroacetophenone stands out in many chemical labs because of its reliability in synthesis work and research. Its clear, almost unremarkable liquid appearance leads some folks to treat it lightly, but anyone familiar knows it brings hazards that call for respect. My early days in lab work taught me quickly—never underestimate a substance based on how it looks. Once, I saw a bottle left open, and the sharp, chemical odors spreading through the room had everyone scrubbing their hands and reviewing safety rules.
What Science Says About Safety
With trifluoroacetophenone, the main risks stem from its volatility and toxicity. Breathing in even small amounts can irritate the respiratory system. Skin contact may cause discomfort or even burns. The chemical’s vapors don’t just vanish, and spills present more than a minor inconvenience. A student I know wiped a drop off a bench without gloves and soon left the lab feeling ill. These stories stick, and they matter more than data sheets that nobody reads in full.
Find the Right Spot
Keeping trifluoroacetophenone away from sunlight and sources of heat isn’t just a suggestion. A dark, well-ventilated chemical storage cabinet always works better than a shelf near windows or radiators. Habit sometimes pushes people to tuck bottles wherever there’s space, but with volatile organics, those shortcuts can lead to leaks or even fire hazards. In some labs I worked in, anything containing strong fluorinated groups required a spot far from acids or bases—precautions that add a layer of insurance.
The Bottle Matters, Not Just the Label
Original bottles do more than advertising—they’re chemical-resistant and seal tightly, which limits evaporation. Some chemists switch over to unlabeled flasks or jars, but one spill drives home the lesson: use proper containers with clear hazard symbols. Over time, I’ve also learned to check storage bottles regularly for cracked seals or crusted lids. There’s no sense in risking a contamination incident because someone didn’t want to toss an old cap.
Personal Safety Gear Isn’t Optional
Gloves and goggles have saved colleagues from messes more often than any high-tech tool. A single splash, even from a drop falling while closing a stopper, carries health risks. It’s not about paranoia—it’s about building habits that keep everyone safe. Proper ventilation, like using a fume hood, can turn a risky procedure into a manageable routine.
Practical Solutions for Safer Storage
Anyone working with trifluoroacetophenone benefits from periodic training sessions and quick-access spill kits nearby. Once, we staged a mock spill just to walk through what to do; seeing people move with purpose instead of confusion builds a sense of confidence. Inventory checks also help—nobody wants to stumble on expired or leaking chemicals in a forgotten drawer. For larger facilities, automated environmental monitoring lets workers catch storage problems before they grow.
Why Good Storage Habits Matter
Labs earn a reputation by how well they protect people, not just output data. Treating substances like trifluoroacetophenone with the respect called for by modern science keeps people healthy, protects equipment, and sets an example for the next generation of chemists. Being careful is not a chore; it’s the mark of someone who takes both their work—and their fellow researchers—seriously.
Understanding Its Substance from the Lab Bench
Working with trifluoroacetophenone, you get the sense that the stuff isn’t interested in doing what most simple chemicals do. Beneath its sharp, biting smell, there’s a clear story told by its volatile liquid form. In my experience, it’s a compound that transforms routine lab work into a game of patience and caution due to its flammability and reactivity with the air around it.
Why Physical Properties Matter Here
Pouring trifluoroacetophenone smells a bit like handling acetic acid, but the presence of those three tightly bonded fluorine atoms changes the whole dynamic. The liquid is colorless and behaves much less like an ordinary acetophenone, largely because that trifluoromethyl group increases its volatility. At room temperature, the pungent fumes announce themselves quickly, and the liquid evaporates fast if you don’t keep the cap on or work in a fume hood. Its boiling point hovers a bit higher than water, around 146°C, and that’s crucial for what happens during chemical reactions or distillation work. When heating it up, its vapors behave aggressively, reminding you that this isn’t a kitchen experiment.
Even its melting point—nestled well below freezing at about -23°C—shows that in the cold, this clear liquid stays liquid, which makes it manageable for many reactions but not pleasant to handle if you spill some on a frosty bench. It doesn’t have much patience for those who leave it uncapped near ignition sources, either: those volatile vapors will find a spark if they can. This makes storage and handling a daily concern. From hard-won trial and error, I’ve learned to respect the low flash point. One careless hand and you might end up on the receiving end of a chemical fire. Vapors carry a health risk, sending a burning tickle through your nose and throat, a warning not to treat it like common solvents.
The Role of Density and Solubility
Pouring trifluoroacetophenone, you’ll feel its density at about 1.3 grams per cubic centimeter. It sinks in water and forms visible layers, refusing to mix. This is the kind of behavior that demands attention during waste separation or transfer between vessels. If you spill it into water, recovery isn’t just as simple as dilution and forget it. You have to separate it with care, and sometimes there’s even more work involved in making sure none leaks into surrounding drains or soil. Solvents like ether or chloroform pick it up just fine, which is useful for chemists, though it can also make accidental releases harder to control.
Making Work Safer and Smarter
In the face of these physical quirks, good protocol goes a long way. Fume hoods, gloves, and goggles should be your best friends, but so should clear procedure and mindfulness. With climate and energy efficiency on everyone’s mind, chemists look for new ways to handle such volatile, hazardous chemicals with more respect for both safety and sustainability. Waste collection for halogenated organics isn’t just paperwork—it’s basic stewardship of the tools and resources we inherit from the past and want to pass along, improved, to those who come after.
Through years of practical exposure, the lesson with trifluoroacetophenone is to never let familiarity breed carelessness. The real property that matters isn’t just its density, boiling point, or irritant vapors. It’s a demanding chemistry partner, and dealing with it responsibly builds real trust and confidence in the lab.