Trifluoroacetyl Chloride: Unpacking Its Role from Laboratory Curiosity to Industrial Staple
Historical Development: Turning Curiosity Into Utility
Chemistry evolves as people chase new molecules. Trifluoroacetyl chloride didn’t show up out of nowhere—it has a backstory that runs parallel to the pursuit of fluorinated building blocks in modern synthesis. In the early and mid-twentieth century, chemists looked for ways to bring the unique properties of fluorine into organic reactions. Trifluoroacetyl chloride offered both a reactive acyl chloride and the muscle of three fluorine atoms, bringing new chemistry and an edge that older reagents lacked. Over time, as industries needed more complex pharmaceuticals, agrochemicals, and specialty materials, this compound gained commercial relevance.
Product Overview: What Sets It Apart
Trifluoroacetyl chloride falls into the family of acyl chlorides, but its identity gets its punch from the trifluoromethyl group. This molecule carries a sharp chemical personality: it’s volatile, reactive, and unforgiving in beginner’s hands. Its use goes beyond just a reagent—this compound sneaks its fingerprint onto pharmaceuticals, crop protection agents, dyes, and polymers aided by the stability and electronegativity delivered by the fluorine atoms. Chemists turn to it when they want a strong electron-withdrawing group in their product.
Physical & Chemical Properties: What You Deal With
You notice its boiling point hovers just above room temperature, making storage and handling a constant concern. The pungent odor lingers even with proper ventilation—an instant reminder not to take shortcuts in the lab. The molecule weighs in at around 130 g/mol, with a nearly colorless to slightly yellow liquid appearance. Its reactivity goes beyond just hydrolysis; few can match its zest for easy reactions with water, alcohols, or amines, rapidly producing hydrogen chloride gas and releasing heat that can escalate without respect for inattentiveness.
Technical Specifications & Labeling: Navigating The Rules
Laboratories and factories face pressure to get the details right. Every bottle demands a clear warning: toxic by inhalation, reacts violently with moisture, and releases corrosive fumes. The flashpoint is low, which forces careful planning around storage. Modern rules require transport with secure containment and detailed labeling under nations’ chemical safety regulations. It isn’t just about ticking boxes; you owe it to your colleagues to give them a fighting chance in case things go wrong.
Preparation Method: Behind The Synthesis
Manufacturers usually go the route of reacting trifluoroacetic acid with common chlorinating agents—thionyl chloride, phosphorus pentachloride, or oxalyl chloride. These reagents deliver more than just a simple swap; side-products like sulfur dioxide or phosphorus oxychloride emerge, so ventilation and precise temperature control become non-negotiable. The process itself might sound simple, but keeping byproducts under control and ensuring a pure, stable product every time takes experience and investment in solid equipment. From my time consulting in custom synthesis plants, even minor temperature spikes threaten batch integrity or the safety of workers.
Chemical Reactions & Modifications: Powering Modern Synthesis
This compound does more than just sit on a shelf; it changes the game for synthetic chemists. Its ability to install the trifluoroacetyl group in amides, esters, and ketones creates backbones common in pharmaceuticals and advanced materials. The product’s high reactivity invites both careful planning and risk: side reactions, violent exotherms, and environmental issues from chlorinated waste challenge everyone from R&D chemists to plant engineers. Experienced hands have seen accidents from improper quenching with alcohols or water, ending with containment foam and emergency room visits.
Synonyms & Product Names: Reading Between the Lines
Sometimes the same compound shows up as trifluoroacetic acid chloride or TFA-Cl. Literature and catalogs might also display one or two trade names, but most researchers recognize it as trifluoroacetyl chloride. Anyone sourcing this chemical must check products lists carefully; confusion here risks purchasing the wrong reagent or misreading hazard information. Fluorinated chemicals travel under a host of names—mistakes in communication at the procurement stage lead to shipping or usage headaches later.
Safety & Operational Standards: Not for the Inexperienced
Handling trifluoroacetyl chloride in any environment requires rigor. No one should open a bottle without proper PPE—nitrile gloves, lab coats, and face shields are bare minimum. Even short exposure to fumes means immediate irritation and a sprint to the emergency shower. Fume hoods become mandatory, not optional. Emergency protocols must translate into real drills, not just forgotten posters on the wall. I’ve seen new hires skip the full changeover of contaminated gloves after a single drop lands; only a narrow escape shows them that the rules protect lives, not just reputations.
Application Area: From Bench to Factory
Trifluoroacetyl chloride shapes progress in more industries than most realize. In pharmaceuticals, it helps create molecules resistant to breakdown by metabolic enzymes, giving drugs longer lifespans. Agrochemical giants count on it to produce active compounds that target pests with precision. Beyond these, the world of polymers and specialty coatings benefits from trifluorinated structures, boosting performance in tough environments like electronics or medical devices. The reach of this single reagent extends globally, linking millions of end users to anonymous vats in industrial parks.
Research & Development: Always a Moving Target
Chemists push boundaries, and trifluoroacetyl chloride keeps showing up as a tool of choice for assembling new molecules. Every year, literature shows tweaks on reaction conditions—shorter times, safer solvents, greener pathways. People explore how to swap out legacy methods using dangerous chlorinating agents, seeking routes that deliver equal purity while cutting down toxic waste or energy usage. As technology moves forward, demand rises for more sustainable handling or even solid-state surrogates that deliver the same group with less risk. Development teams tasked with green chemistry mandates look at this compound and see a challenge: How do you keep the utility but slash the hazards?
Toxicity Research: Understanding Risks Beyond the MSDS
Regulatory agencies and safety researchers dig into what happens when humans or the environment meet trifluoroacetyl chloride. Acute inhalation or contact burns are easy to imagine, but chronic low-level exposure research takes longer to unfold. Animal studies show respiratory harm and delayed healing when tissue exposure goes unchecked. Waste streams containing hydrolysis byproducts—especially in water-rich surroundings—call for thoughtful neutralization and containment. People underestimate the risk of handling even small samples; I’ve watched complacency undo years of safe operations with a single careless moment.
Future Prospects: Meeting Challenge With Ingenuity
The future for trifluoroacetyl chloride isn’t set in stone. Regulatory pressures push for alternatives or safer handling systems as society demands sustainable operations and less environmental damage. Investors watch closely as new catalysts or milder substitutes emerge from academic labs. Companies investing in automation and closed-system handling reduce operator risks but face high upfront costs. Startups pitch greener routes to trifluoromethylation, aiming to capture market share with lower emissions or safer waste. While the importance of this compound will likely continue in globally connected supply chains, only careful balance of innovation, regulation, and real-world experience will secure its place for the next generation.
The Backbone Role in Synthetic Chemistry
Chemists love building things, from tiny drug molecules to complex crop protection tools. Trifluoroacetyl chloride sits in that crowded toolbox, doing a job few other chemicals can. It brings trifluoromethyl groups into molecules, making new stuff that behaves in unique ways—stuff that can survive harsh acids or resist breaking down in a body. That little -CF3- bundle packs properties modern chemists hunt for. You find trifluoroacetyl chloride pulled off the shelf most in labs that push out pharmaceuticals, specialty chemicals, and new materials.
Building Better Medicines, For Good and Ill
A lot of medical breakthroughs rely on changing the structure of molecules so they hang around in the body just long enough to help. Engineers of medicine use trifluoroacetyl chloride in their hunt for longer-lasting drugs, targeting cancer, mood disorders, and chronic illnesses. The trick lies in the way that trifluoromethyl group blocks enzymes from chewing up the molecule too fast. Yet with each new molecule built, someone has to make sure it proves itself safe, and that takes years—and people devoted to thoughtful chemistry.
More Than Medicine: Fields That Rely on This Chemical
Crop scientists and people working on pest solutions also reach for trifluoroacetyl chloride. Those trifluorinated building blocks let them create new molecules that don’t wash away in the rain and don’t break down in tough sunlight. These qualities keep pests at bay, but they also raise real concerns about what we leave behind in soil and water. The same resistance to break-down that helps farmers poses tricky environmental questions once the field's been harvested.
Environmental Trade-Offs and Worker Safety
Nobody jumps into using strong chemicals without thinking of risks. Trifluoroacetyl chloride is volatile and releases strong fumes. If it leaks or someone misuses it, exposure can cause real harm to people and nearby wildlife. This isn’t a chemical for rookies; handling it takes real training, good gloves, well-ventilated spaces, and sharp attention to waste. Accidental releases can turn a routine day at a lab or plant into a genuine emergency.
Changing How We Use Chemicals—And How We Make Them Safe
Companies and researchers looking to minimize harm have begun hunting for alternatives or safer processes. There’s excitement around “green chemistry” which gets designers to ask whether they can skip the harsh stuff or recycle what’s already in the pot. Governments hold producers accountable with regulations. Responsible handling means sharing information—not just locking it away in technical documents, but teaching workers and communities about risks. Only a few decades back, the norm involved strong smells and little discussion. Now, disclosure matters.
Striking a Balance: Science, Progress, and Care
The story of trifluoroacetyl chloride reveals the best and the challenges of chemical innovation. Powerful tools can fuel life-saving medicines and new solutions in farming, but they're best used with respect for the people working with them, and for downstream impact. Keeping progress safe means refusing shortcuts on safety and continuing the push for friendlier chemicals in tomorrow’s labs. This balance isn’t always comfortable, but it’s essential for trust—both in science and in the products that shape our lives.
Experience Teaches That Chemistry Isn’t Forgiving
I remember my earliest days in the academic lab, when the mention of “acyl chlorides” got a nervous chuckle from any researcher who’d been singed at least once. Trifluoroacetyl chloride brings its own brand of menace, not as familiar as some, but every bit as aggressive. Ask anyone who’s cracked open a fresh bottle; the sharp, biting odor hits first, chased by the knowledge that even a drop off the glove can spell trouble.
No Shortcuts With Protective Gear
Lab veterans don’t argue about the value of personal protective equipment here. Nitrile gloves, splash goggles, and full-length lab coats aren’t suggestions; they’re essentials. Fume hoods matter just as much. Trifluoroacetyl chloride fumes sting the eyes and cut through air with a cough-inducing punch. ScienceDirect notes the compound hydrolyzes on contact with moisture, which includes every exposed mucous membrane. Even in short exposures, the damage accumulates faster than many expect.
Understanding Real-World Risks of Exposure
Ask any seasoned chemist, and they’ll tell stories of rushed procedures going sideways. Direct skin contact brings acute burns. Eyes exposed to vapors risk serious injury, not just an afternoon of tearing. Vapors are heavier than air and sneak out of poorly sealed flasks. An overheated reaction or broken bottle quickly fills a small space, leaving little room for error. The CDC warns about the severe effects of chlorinated and fluorinated compounds on lungs; Trifluoroacetyl chloride doesn’t pull its punches here.
Preventing Accidents: It’s in the Details
Before opening any bottle, double-check the venting on the hood. Line all work areas with absorbent pads. Keep a bottle of sodium bicarbonate or a quenching agent within reach, not stored under the sink. We don’t just do this out of habit—quenching a small spill right away makes the difference between mild irritation and a full blown chemical burn. Every bottle deserves a secure cap and clear labeling, and no one should treat older partially-used bottles as safe to ignore.
Training Translates to Fewer Emergencies
Labs that prioritize real-world handling drills face fewer accidents. Practice opening bottles with pipettes, securing containers one-handed, and cleaning minor spills quickly builds confidence. I've seen high school students practice with colored water before handling dangerous acids. For trifluoroacetyl chloride, even trained professionals rehearse steps out loud: “Prepare the quench. Check the seal. Open in the hood.” Simple routines cut through nerves and muscle memory takes over when things get tense.
Reducing Risk for Everyone in the Building
Responsible storage goes a long way. Corrosives cabinet—never regular shelving—keeps reactivity in check. Sharing storage spaces in busy labs raises risks unless every bottle follows strict tracking and signage protocols. Waste needs dedicated containers, as mixing with incompatible chemicals or careless disposal creates new hazards. Data from occupational health reports show that chemical accidents cluster in spaces where people ignore these basics.
Building a Safety Culture That Lasts
No single rule keeps labs accident-free, but a healthy respect for what trifluoroacetyl chloride can do creates an attitude that spreads. Transparency about incidents—no sugarcoating—makes new researchers quicker to ask for help. In my experience, regular safety meetings focused on personal stories, not dry regulations, keep the risks real and everyone’s focus sharp, ensuring a lab where accidents stay rare and minor.
Why Chemists and Industry Care About Trifluoroacetyl Chloride
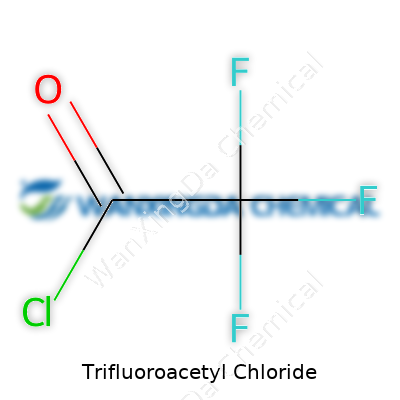
Ever walk into a laboratory, catch that whiff of sharp, slightly sweet chemicals in the air, and wonder what’s behind it? For folks who work with organic compounds, trifluoroacetyl chloride comes up as something more than just another bottle on the shelf. The formula—C2ClF3O—looks simple, but what it unlocks in synthesis says a lot about how chemistry shapes what society can build, from new drugs to specialty plastics.
Trifluoroacetyl chloride packs a punch because of its structure. Think of a carbon backbone with three fluorine atoms hooked right up. There’s a chlorine clipped to a carbonyl, forming a reactive acid chloride group. That might sound technical, but this structure makes the molecule both useful and a handful to handle. Fluorine atoms bring unmatched electronegativity, a bit of chemical stubbornness, so compounds like this one stick around longer and react in unusual ways. The carbonyl chloride unit speaks for its reactivity—it’s ready to leap into action given the right partner, which is exactly why folks reach for it when building new molecules.
The Science in Everyday Problems
In the pharmaceutical industry, researchers chase after drugs that won’t break down before they hit their target. The trifluoromethyl group, which this molecule can tack onto other compounds, often lends stability and can even change how a drug interacts in the body. If you’re a chemist hoping to slam a trifluoromethyl group onto an amine, trifluoroacetyl chloride offers a direct and time-proven route. It isn’t only pills on the line either—polymers used in coatings and electronics often need exactly this kind of chemical tweak to survive heat or harsh solvents. These tweaks fuel progress long after the reaction flask gets washed out and shelved.
It’s not all upside. Handling trifluoroacetyl chloride requires guts and skill. Once you open that bottle, there’s no room for guessing. This compound reacts fiercely with water, spitting out hydrogen chloride gas, which can burn lungs and skin. That risk demands respect. Labs and manufacturers need robust ventilation, solid personal protection, and strict handling protocols. Skipping steps is not an option. Incidents over the years—accidental releases, mishandled shipments—show what happens when safety practices lag behind chemical ambition. The lesson isn’t just for scientists: people who transport, store, and dispose of these materials form the fragile safety net for the rest of us.
Smarter Solutions For Chemistry’s Tough Challenges
Better solutions don’t just drop out of the sky. Some research groups look to swap out hazardous acid chlorides for milder alternatives. New reagents sometimes fill the same niche but with less risk. Training is at the heart of any safety push. I remember seeing young researchers learn the ropes, starting with mock drills before touching live chemicals. Clear documentation, fume hoods, and real accountability build a culture where people can push the limits of chemistry without gambling with their health.
Regulators also play a role. Decent rules on labeling, shipping, and using chemicals like trifluoroacetyl chloride have cut down on preventable incidents worldwide. Compliance is more than a line in a safety audit—it’s about recognizing the real dangers and accepting collective responsibility for what happens in and outside the lab.
Looking Ahead
Trifluoroacetyl chloride may not make headlines, but it’s part of the invisible machinery pushing innovation forward. Success in chemistry often means making tough calls about safety, utility, and environmental impact. Transparent conversation and constant training keep those who rely on this molecule safe and productive while opening the door to new science. As research moves forward, the hope is not just for better molecules, but for more people to go home safe after a hard day’s work at the bench.
Stories from work stick with you. The first time I handled a bottle of trifluoroacetyl chloride, the warning on the label made me skip lunch just to research what exactly I was about to touch. This compound earns its nasty reputation for a reason. It's corrosive, fuming, and has a habit of reacting with water to unleash toxic gases. Storage isn’t just a technical formality—it’s a matter of health, safety, and keeping things from going off the rails in the lab.
Why Proper Storage Can’t Be Skipped
One slip in storage isn’t just a missed tidiness point for the safety inspector. Trifluoroacetyl chloride releases both hydrogen chloride and trifluoroacetic acid vapors when mixed with moisture, both of which chew through metal and human tissue with equal enthusiasm. Fumes seep right through plastic wrap or thin gloves. Breathing that in? People have landed in the emergency room for less. I’ve seen metal shelving show pits after a small leak in a cap, and I’ve watched a new grad nearly faint after walking too close to a bottle stored near a sink.
Facts That Matter
There’s plenty of research on chemical storage, and the collective wisdom tells us to keep trifluoroacetyl chloride in tightly-sealed containers, preferably glass with a Teflon-lined cap. Ordinary plastic fails pretty fast. Humidity is the enemy, so the compound belongs in a cool, dry, and well-ventilated area—locked away from sinks, water baths, or anywhere condensation might form. Storing it above eye level means even a small spill puts a person directly in harm’s way. I learned from a lab manager who always insisted on storing reactive compounds below waist height, in trays that catch leaks before they spread.
Regulators have chimed in, and their advice holds up: segregate this stuff from common chemicals such as acids, bases, and especially anything reactive or flammable. The United States Occupational Safety and Health Administration and European Safety standards both treat it as a substance that calls for tight restrictions. Accidents involving incompatible chemicals can spiral in a flash—sometimes literally. Data on workplace incidents show that improper storage causes more injuries than accidental spills during use. Trifluoroacetyl chloride reminds us that handling isn’t just about wearing gloves; it’s about planning where the danger sits every night.
Simple Steps Go a Long Way
Each lab routine means double-checking seals and labels. If a bottle feels sticky, the safest move is not to wipe it and hope for the best, but to triple bag and replace it. Training new lab members to recognize the smell and appearance of leaks saves more trouble than any sign or poster. I like to keep a log—boring until something goes wrong—so everyone knows when it’s last been moved, opened, or inspected.
Backup matters, too. Proper ventilation makes all the difference. Fume hoods aren’t just for the dramatic reactions. Even small amounts of escaped vapor build up if the air isn’t moving. Emergency eyewash stations and spill kits with calcium carbonate neutralizer sit nearby. In the decade I’ve worked with these materials, I’ve found that the culture of vigilance, the daily habit of checking and re-checking, carries more weight than any top-down directive.
Building a Safer Mindset
Witnessing the aftermath of a storage failure teaches a blunt truth: shortcuts never pay off. Simple rules—dry space, sturdy containers, clear labeling, good logs—aren’t just about following a checklist. People rely on them to walk out of the lab at the end of the day with their health intact. A little care in storage today saves endless headaches and heartbreak tomorrow. That’s why every person who opens that storage room door should know exactly what’s inside, how it’s stored, and why those rules exist in the first place.
An Unfriendly Chemical in the Lab and Beyond
Trifluoroacetyl chloride sounds like something tucked away in a science textbook, but in some industries, it ends up playing a bigger role. You find it in chemical research, pharmaceutical production, and sometimes in the making of specialty polymers. Even those of us with decent chemistry grades didn’t learn much about its danger outside the classroom. Yet it carries some hefty health hazards, most people barely hear about.
Hazards More Than Skin Deep
Breathe it in, and the body pays the price. Trifluoroacetyl chloride has a strong, sharp smell, and it's not just an annoyance—it actively irritates the nose, throat, and lungs. Not long ago, I visited a colleague’s lab where this chemical was in use. Even from the entrance, my eyes started stinging, and I started coughing, despite the presence of fume hoods. The problem lies in the compound’s ability to release corrosive gases like hydrochloric acid when it reacts with water—including the moisture in the air or your own respiratory tract. That means the damage isn’t just temporary discomfort. It can lead to burns in the nose and windpipe, trigger bronchospasms, and cause persistent coughing and chest tightness.
Skin contact brings another set of dangers. Splash a little on your hand or arm, and painful blisters show up fast. Without immediate washing, the injury deepens. Eyes prove even more vulnerable. A small drop can cause serious burns, leading to permanent vision problems. The immediacy of this damage sticks with me—anyone who’s seen a chemical burn in person walks away knowing no lab procedure or shortcut is worth the risk.
Long-Term Worries and Hidden Dangers
Short-term exposure can be dramatic, but the long-term effects deserve concern, too. Health records and occupational studies point to the risk of chronic respiratory problems—wheezing, asthma-like symptoms, and increased susceptibility to infections. It’s hard to forget the faces of workers who thought a quick exposure was a small price to pay, but now live with nagging coughs that never seem to fade. While research is ongoing, the toxic load on the liver and kidneys raises even more red flags, suggesting repeated exposures could add up, with real consequences far down the line.
Why Preventing Exposure Matters
Facts tell a straightforward story: you can’t undo chemical injuries. Most of us in science or manufacturing have stories of shared safety goggles, torn gloves, someone skipping a safety check to save time. I’ve seen close calls myself. Truth is, prevention always beats treatment. Good ventilation, high-quality personal protective equipment, and strict procedural training make a real difference. It’s not enough to stick a warning label on a bottle and hope for the best. Regular refresher training turns chemical safety into habit. Facilities need active oversight, and management needs to emphasize health not just production quotas.
Replacing trifluoroacetyl chloride with safer alternatives where possible also makes sense. Some research points toward substitute reagents in pharmaceutical labs, offering similar results with less risk. Businesses should regularly seek out new practices, even if it calls for up-front investment. Cutting corners with hazardous chemicals only wins in the short term—long-term, the bills for healthcare, lost workdays, and legal action add up faster than most managers realize.
Put Health and Safety at the Center
Few of us think about obscure chemicals like trifluoroacetyl chloride, but for workers in the industries that use it, the risks are all too real. A commitment to health needs to run deeper than compliance checklists. It lives in the way we plan processes, train staff, and treat every near-miss as a lesson worth learning. People’s lives deserve that much respect.