Trifluoroacetylacetone: Real Impact From a Small Molecule
Historical Development
Chemists in the early twentieth century started tinkering with acetylacetone—a simple diketone with a knack for chelating metals. By the 1950s, interest grew in swapping out some hydrogen for fluorine, giving rise to trifluoroacetylacetone. The story highlights something familiar in chemistry: new tools get made because someone chases a specific need. Researchers wanted a more electron-withdrawing group, hoping to boost volatility and change reactivity, and fluorine showed promise. This isn't just chemistry for chemistry’s sake. As electron-withdrawing power shifts the molecule’s acidity and coordination, suddenly researchers used trifluoroacetylacetone in everything from metal extraction to catalysis. It kicked off decades of studies, giving the lab world a new chelating agent beyond the basics.
Product Overview
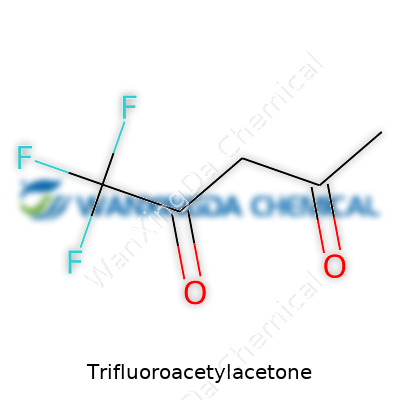
Trifluoroacetylacetone stands out with its transparent, colorless liquid form and a sharp odor that reminds anyone who’s handled diketones of their common backbone. Chemically, its structure’s simple: swap the methyl group from acetylacetone’s end with a trifluoromethyl group. This small tweak brings a huge effect on its chemical personality. In the lab, chemists appreciate how even a minor substitution throws acidity and reactivity in new directions. Behind that is the trifluoromethyl group—the reason for the molecule’s distinct roll in extractions and synthesis. Sometimes the usefulness of a chemical comes from what can’t be seen with the naked eye, and this is one of those times. Despite its volatility and vapor, the liquid remains in laboratories for its key coordination traits and unique role in organometallic chemistry.
Physical & Chemical Properties
Trifluoroacetylacetone carries a formula of CF3COCH2COCH3, boasting a boiling point far below its non-fluorinated cousin. The vapor pressure at room temperature makes storage and handling a practical concern. One immediate impact comes in its acidity: thanks to the electronegativity of those three fluorine atoms, the hydrogen at the methylene bridge sits more acidic than similar diketones. That changes not just the molecules it binds with, but also the sort of metals it drags out of solution during extractions. It’s hard to overstate how a shift in molecular electronics can open new doors for separation and analysis, especially in radiochemistry and trace element studies. The liquid remains stable in dry air but, exposed to moisture, it quickly hydrolyzes—another nod to the delicate balance in using reactive intermediates safely and effectively.
Technical Specifications & Labeling
While reading a label isn’t thrilling, the details can make a difference. For trifluoroacetylacetone, purity often exceeds 98%. In a real lab, details like water content, presence of residual acids, and clarity show up in paperwork. Researchers rely on trusted suppliers for a tight batch-to-batch standard, especially when the downstream chemistry is sensitive. The labeling sticks to chemical identity, CAS number, and safety flags—like warnings for volatility and skin irritation. I’ve stood in enough stockrooms to know genuinely good chemical labeling keeps everyone safer and gives back time that would have gone to double-checking properties. It helps that these breakdowns aren’t just bureaucratic hurdles; proper documentation avoids nasty surprises during syntheses or extractions.
Preparation Method
Creating trifluoroacetylacetone usually involves a Claisen condensation. Unlike the original acetylacetone, this version draws on the condensation of ethyl trifluoroacetate with acetone under basic conditions. Lab memories come back of slow additions and constant monitoring for exotherm. Purification, usually by distillation, demands precision since high volatility means material loss if heating is rushed or temperature swings happen. On an industrial scale, reactors take over, but the chemistry stays rooted in simple organic transformation. Handling alkali and managing hazardous vapors becomes part of the trade-off between running efficient processes and keeping people safe. This is where experienced chemists step up; a reliable process matters much more than squeezing out a few more milliliters of product.
Chemical Reactions & Modifications
The versatility of trifluoroacetylacetone comes alive in its reactivity. Chelation always gets top billing: the molecule wraps itself around transition metals, forming stable five-membered rings, which then serve in extraction of metals and even preparing volatile complexes for analytical chemistry (think atomic absorption). Beyond chelation, its own acidity means it protonates or deprotonates in controlled ways. Synthetic chemists turn trifluoroacetylacetone into ligands, derivatives, or intermediates—using those properties to shift selectivity in downstream chemistry. The simple methylation or halogenation steps morph the molecule further, and the strength of the trifluoromethyl group ripples outward, changing how reactive centers behave. For anyone in the trenches of organic synthesis, this is a tool with more sides than first appear.
Synonyms & Product Names
Colleagues ask for trifluoroacetylacetone under all sorts of names. In some supply catalogs, it goes by 1,1,1-trifluoro-2,4-pentanedione or TFAA. Labels often show a string of synonyms: 2,4-pentanedione,1,1,1-trifluoro-; trifluoro-2,4-pentanedione; and more. In many circles, “trifluoroacac” or “TFACAC” shows up in shorthand. Anyone working with multiple vendors has run across nearly every variant. Clear communication—both in the lab and in procurement—avoids mix-ups, as ordering a similar-sounding acetylacetone derivative can derail a whole series of experiments.
Safety & Operational Standards
Getting careless with trifluoroacetylacetone doesn’t end well. Its sharp odor signals both volatility and potential harm, and the exposure risk is real. Skin contact leads to irritation, inhalation of vapors can cause headaches or respiratory discomfort, and liquids splash unexpectedly. Modern lab practice demands fume hoods for weighing and transferring, and gloves every time. Training goes beyond reading a data sheet; people need to understand why goggles, aprons, and quick cleanup matter—one ruined day from a skin rash or chemical splash teaches faster than any poster on the wall. Spillage must be diluted and neutralized, and contaminated rags or waste belong in hazardous bins, not ordinary trash. These habits, built daily, stop small risks from turning into accidents. With environmental regulations clamping down, collection and disposal of spent material deserves the same discipline as everything upstream.
Application Area
Trifluoroacetylacetone’s biggest impact landed in analytical and inorganic chemistry. Its ability to form volatile metal chelates got it into atomic absorption and emission spectrometry work. Lab teams rushed to it for trace metal extraction, using the compound’s strong chelating and volatility profile to speed up sample preparation. In radiochemistry, its specificity gave selectivity in pulling out actinides and rare earths. Researchers in coordination chemistry often explore the family of ligands to tinker with catalyst design or study basic electronic effects. Some pharmaceutical syntheses rely on the unique electronic properties when a trifluoromethyl ketone is key to the route. This diversity comes because the molecule can be tailored or dropped into so many workflows, always leaving a mark with its acid strength and ability to hold metals tighter than simpler diketones. It’s a recurring tool at the experimental edge where other chemicals reach their limits.
Research & Development
Every few years, published articles add a new take on trifluoroacetylacetone. Method developers keep pushing the material in mass spectrometric sample preparation, trying to increase sensitivity or reduce prep time. Organometallic researchers often test new trifluoroacac complexes, examining their performance as catalysts or exploring metal-ligand effects in fine detail. Some teams adjust the synthesis route to scale up with fewer byproducts or greener reagents—a nod to both cost and environmental pressure. Meanwhile, computational chemists deploy modeling tools to predict what coordination complexes might do before they get made at the bench. For me, this cycle feels familiar—old tools become new again as people look at a problem from another angle. Lab partnerships across disciplines mean purification experts, spectroscopists, or process chemists all chime in, taking an old molecule down new roads.
Toxicity Research
Handling trifluoroacetylacetone comes with an undercurrent of caution that’s well deserved. Toxicologists have spent time tracking not just acute impacts, but what vapor or chronic exposure might do. Most data point to moderate toxicity: short-term exposure causes irritation to skin and lungs, while larger doses produce central nervous system effects. Animal studies back up these observations, though the compound’s volatility makes dose estimation tricky. Environmental fate studies show that trifluoroacetylacetone hydrolyzes in water, with breakdown products—mainly trifluoroacetic acid—raising their own set of concerns for aquatic life. In my experience, the way forward comes from tight procedural controls and clear emergency steps for exposure; relying on hope alone never works when chemistry gets risky. Ongoing monitoring in industrial and academic settings feeds fresh data into safety guidelines, keeping frontline workers protected as long as vigilance holds up.
Future Prospects
Chemistry doesn’t stand still; neither does the utility of a compound like trifluoroacetylacetone. Current trends lean hard into sustainability and precision, opening chances to reinvent old process chemistries with less waste and more targeted outcomes. As interest in advanced materials, especially transition metal complexes, grows for electronics or catalysis, researchers will keep tweaking trifluoroacac frameworks to eke out better function. Some labs already chase modified analogs for better selectivity or environmental persistence. Regulatory changes around fluorinated chemicals could push the field toward even cleaner versions or more rigorous containment, so anyone invested in production or R&D needs to watch laws and best practices evolve. This small molecule’s continued relevance hinges on careful stewardship, sharp science, and the willingness to adapt processes each time the field jumps forward. In academic, industrial, or government labs, chemistry rooted in practice keeps driving meaningful progress.
Trifluoroacetylacetone doesn't roll off the tongue like everyday words, but this clear, organic compound finds use in places most people never consider. Some years back, my chemistry professor held up a bottle of this stuff, grinned, and said, “This is how we catch metal — not in a net, but in a handshake.” I never forgot that lesson.
The Hidden Role in Metal Extraction
Chemists rely on trifluoroacetylacetone as a ligand. In plainer language, it acts like a hook. It grabs onto metal ions, helping researchers separate rare metals from tough mixtures. Take research labs digging into new materials — they're always chasing purity. By forming stable complexes with metals like copper, zinc, or iron, this chemical lets scientists pull out and study single metals with fewer steps, using less energy and fewer harsh chemicals than older methods. Real progress in clean-energy materials or electronics can trace its roots back to such processes. If you’ve used a device with advanced sensors or strong magnets, odds are someone in the supply chain leaned on chemistry like this to get the job done.
Catalysts and Industry
Trifluoroacetylacetone isn’t just about pulling out metals. It pops up as a building block for catalysts — substances that speed up reactions without getting used up. Companies blending new plastics or high-performance coatings often look for a catalyst that keeps working longer and gets more out of raw materials. The presence of three fluorine atoms in this molecule can boost catalyst stability or change how much gets made in a chemical batch. So, factories can run smoother, waste less, and still hit the quality mark, all thanks to a little trick from organic chemistry.
Backstage in Science
In the world of analytical chemistry, accuracy counts. When scientists need to see trace amounts of metals — imagine looking for a grain of salt in a swimming pool — they use trifluoroacetylacetone to form distinctive, easily detectable metal complexes. This helps in environmental monitoring, checking for pollutants, and making sure drinking water isn’t carrying anything that shouldn’t be there. My own summer internship left me hunched over instruments, and this chemical kept showing up in sample prep kits. The results shaped decisions about river clean-ups and waste disposal, giving communities confidence in their water safety.
Safety and Responsibility
Handling trifluoroacetylacetone calls for training and respect. It’s not meant for kitchen experiments. Like many chemicals, its vapors can irritate, and exposure adds health risks. Labs using it follow strict protocols, including proper ventilation and gear. Industry groups and regulatory agencies set exposure limits based on the best science available, since public health rides on responsible chemical management. Over the last decade, I’ve watched the bar rise for lab safety, and practices around handling tricky compounds like this have improved tremendously.
A Place in Tomorrow’s Technologies
What amazes me is how such a niche chemical keeps finding its way into new stories: more effective medicine delivery systems, next-gen batteries, and materials that stand up to harsh environments all depend on precisely those metal complexes this molecule helps form. As the world chases greener technology and safer products, the role of chemicals like trifluoroacetylacetone grows.
Looking Closer at an Overlooked Molecule
Ask anyone who’s spent time hunched over lab glassware and you’ll get a quick sigh at the mention of complex organic molecules. Trifluoroacetylacetone, with its molecular formula C5H5F3O2, stands out for reasons that hit home both in research and in practice. Unlike many buzzworthy chemicals, it doesn’t pop up in rookie textbooks. Folks in academia or industry end up bumping into it in metal complex synthesis, catalysis, or when hunting for ligands with a little extra punch.
Structure That Draws Attention
Take a pen and sketch it out—you’d find a backbone shaped by five carbons, laced with two oxygen atoms that look set for enol or keto action, and sporting a trifluoromethyl group. The chemical structure usually gets drawn as CF3COCH2COCH3. Here, that CF3 group swings the balance. The electron-withdrawing nature of fluorine doesn’t just tweak reactivity, it massively cranks up volatility and adds stability where it counts.
People in my world—old colleagues wrangling with transition metals, or students fighting through their first NMR spectra—can tell you the difference a few fluorines make. With trifluoroacetylacetone, chelation leaps forward. That means metal ions grab onto the oxygen atoms from both the keto and enol sides without letting go. The result: complexes that resist hydrolysis and tolerate nastier reaction conditions.
Why Its Role Matters in Everyday Labs
One reason trifluoroacetylacetone earns respect in chemistry circles: it keeps experiments moving in tough spots. Picture researchers building catalysts for polymer production. Using this compound, they dodge the headaches of metal instability, contamination, or water-mediated breakdown. That translates into cleaner runs and better yields. For graduate students running overnight reactions, that reliability means less anxiety when dawn rolls around and flasks are checked.
It doesn’t end with metal chelation. Analytical chemists put this molecule to work in spectroscopic techniques—think NMR and IR—since those fluorine atoms lay out easy-to-track signals. That clarity gives real confidence when you’re on the hook for pinning down subtle structural details or reaction outcomes.
Sustainability and Safety Concerns
Some worry about trifluorinated compounds sticking around in the environment. The fluorine-carbon bond doesn’t like to break, raising real worries for disposal and exposure. These issues played out in my own department where new protocols had to be written after a few near-misses with improper waste handling. Research groups and lab managers can’t just shrug off these risks. It pays to build safer collection and neutralization routines into regular tasks.
Real Solutions Come from Ground-Level Changes
Researchers can swap in greener alternatives if project constraints allow, or at the very least, invest in training around proper chemical management. Sharing best practices, open documentation, and realistic risk assessments foster a culture that balances progress with responsibility.
In the end, chemistry isn’t just about what’s on the chalkboard or hidden in catalogs. It’s the lived experience of every researcher who chooses, handles, and ultimately disposes of these molecules that gives them significance. Trifluoroacetylacetone, straightforward by formula but far-reaching in application, keeps challenging us to be sharp, responsible, and innovative with each new experiment.
Real Risks Behind a Familiar Laboratory Compound
Stepping into any chemistry lab, you quickly realize that safety isn’t just a box to tick. Each bottle on the shelf holds more than its label lets on. Trifluoroacetylacetone, with its long name and industrial uses, seems like just another reagent at first glance. People working in research, manufacturing, or even advanced education sometimes treat it like a routine tool. That tendency to gloss over real health risks keeps me on my toes whenever someone asks about chemical safety.
Hazards Aren’t Just Theoretical
Trifluoroacetylacetone carries more hazards than many expect from a seemingly run-of-the-mill compound. For starters, it’s a volatile liquid, releasing vapors that sting the nose and irritate the throat. These vapors don't just cause a sneeze; with enough exposure, they can lead to coughing and even chemical burns in the airway. In my own time around labs, I’ve watched folks develop headaches and dizziness from fleeting encounters—reminders that the body responds quickly to more than just dramatic spills.
Getting trifluoroacetylacetone on skin isn’t harmless, either. Direct contact leads to redness and irritation, and those who put off rinsing it off sometimes develop blisters. Eye exposure really amplifies the danger; watery, burning eyes often send people racing to the eyewash station. These aren’t rare reactions. Instead, they’re standard fare—seen by anyone who spends enough time handling chemicals.
Beyond Immediate Symptoms: Toxicity and Long-term Risk
Whether a substance builds up harm over time matters, not just in one-off accidents. With trifluoroacetylacetone, animal studies show liver and kidney stress after repeated exposure. This has to count for something. Workers who handle it daily face a level of risk that hasn’t always been respected. Some studies also report central nervous system effects after prolonged inhalation, which matches what many solvent-related compounds do.
Trifluoroacetylacetone’s chemical structure—two carbonyls and a hefty dose of fluorine—gives it the power to enter the bloodstream quickly through inhaled or absorbed routes. Once there, it tends to disrupt enzyme systems. That matters most to those who treat gloves and goggles as optional or who think good ventilation is just a comfort feature.
Prevention and Safer Handling Make a Difference
Working with hazardous chemicals has a way of encouraging better habits, especially when there’s a persistent threat of acute or long-term damage. Chemical fume hoods, gloves, and eye protection really change outcomes. Modern best practice also means closed storage and careful waste disposal—no shortcuts. I’ve witnessed younger lab workers skip the basics, convinced trifluoroacetylacetone is less risky because of its widespread use. Those blind spots keep emergency showers busy.
It makes sense to demand tighter workplace monitoring and reinforce training that shows how easily trifluoroacetylacetone’s hazards can become personal injuries. Peer-reviewed literature and regulatory data both support labeling it as a substance that needs respect, not just paperwork. It’s also worth pushing for substitutions in certain applications where safer alternatives exist; limiting cumulative exposure matters.
Respecting the Real Dangers
Calling trifluoroacetylacetone hazardous isn’t alarmist. The evidence lines up—irritation, toxicity risks, and lasting harm with repeat exposure. People who manage chemistry day-to-day deserve clear, direct guidance rooted in both experience and science. There’s no reason to ignore the facts or the protocols. By matching vigilance with up-to-date facts and simple habits, the number of close calls drops. That’s how any lab stays safer—no matter how trivial a chemical might seem at first glance.
Hard Lessons in Chemical Storage
Most people who have spent time in a lab have at least one close call they remember all too well. I still remember a day a glass bottle slipped from my hands — expensive reagents, warnings all over the label, and the whole place stunk of solvents for hours. Those warnings exist for a reason, especially when chemicals have a reputation for volatility, reactivity, or just plain danger. Trifluoroacetylacetone fits the bill. It comes with volatility, is flammable, and the vapors irritate like someone shoving a bleach-soaked rag under your nose.
What Sets Trifluoroacetylacetone Apart
This is not some everyday household cleaner or substance a curious teen slides into a science fair volcano. It’s used in advanced synthesis, often in coordination chemistry or material science labs. What makes this chemical stick out isn’t just its trifluoromethyl group lending extra volatility but the way it acts around air and moisture. Even when you handle it in small volumes, the risk sits high simply due to how easily it evaporates, spreads, and reacts.
From Storage Shelves to Safety Cabinets
Years back, the standard advice was a capped bottle on the shelf, maybe a label slapped on in faded ink. Now, anyone with sense keeps compounds like this in dedicated, ventilated storerooms, far from heat, sunlight, and any hint of a spark. Fireproof cabinets with secondary containment—these aren’t luxuries or overkill. They’re shaped by hard experience and updated regulations. Even a brief lapse, like leaving a vial in a warm room, can end with fumes in the air, alarms ringing, and everyone hustling outside. That’s not just theory—labs empty for reasons that could have been prevented by spending an extra minute at the storage cabinet.
Personal Experience: Gloves and Goggles Aren’t Optional
There’s a tendency to wave off using PPE with something you’ve worked around many times. That’s a mistake. Chemical-resistant gloves, safety goggles, and disposable lab coats go beyond box-ticking. A single splash can burn, and the fumes feel like breathing in ammonia and acid all at once. Ventilated fume hoods are the real MVP here. I watched a colleague once ignore the hood with “just a small transfer,” and it ended badly. Red eyes, coughing, and, days later, a strict policy in that section of the lab.
Why Training and Protocols Aren’t Just Bureaucracy
Nobody wants another training session. But when it comes to chemicals with rapid evaporation and nasty vapors, training is education that saves skin, lungs, and sometimes lives. Where clear instructions and regular drills exist, accidents drop. Labels fade, instructions get lost, and old habits return without regular reminders.
Practical Solutions for Tough Problems
Beyond the usual precautions, investing in real-time chemical vapor detectors can give early warnings. Clearly marked spill kits near every workstation help staff respond fast. Separate waste containers, checked by designated staff, mean one less step is missed on a bad day. Digital inventory systems catch expired or degrading chemicals before they cause problems.
Bottom Line: Respect the Risks, Lean on Experience
Work with trifluoroacetylacetone long enough, and the stories stack up. Ventilated storage, PPE, regular training, and a culture of double-checking save more than reputation—they prevent injuries and keep doors open. Experience teaches that every step skipped might be the one that turns a routine day into a disaster. No chemical respect pays off more than with ones like this.
Behind the White-Coated Doors
Trifluoroacetylacetone might not get a mention in the college undergraduate textbook, but folks in research and industry definitely keep a bottle on the shelf. Anyone who has handled metal ions in a lab, be it for chemistry, physics, or materials science, has probably stumbled across this compound’s utility whether they noticed or not. The three fluorines on its backbone make it a far cry from the usual organic chemicals. That strong electron-withdrawing pull completely flips how it interacts with metals compared to ordinary acetylacetone.
Taking Metal Chelation Up a Notch
Researchers often need to grab metals out of a mix. Creating stable metal complexes isn’t just an academic exercise; it’s how people separate rare earths, purify catalysts, and get sensitive measurements. Because the trifluoromethyl group drags extra electron density away, metal ions like aluminum, copper, and lanthanides stick tighter to trifluoroacetylacetone than to its plain cousins. Less solubility in water, more stability in organic phases—these little tweaks keep things cleaner and more predictable in separation workflows. Plenty of new catalysts start life with a trifluoroacetylacetone-metal core before heading into polymerizations or green chemistry reactions.
Clean Surfaces, Better Devices
Semiconductor folks and materials scientists lean hard on trifluoroacetylacetone in the chase for pure, uniform coatings. In vapor deposition processes, starting with a metal-trifluoroacetylacetonate often gets around the problem of water sensitivity or messy side reactions. You want a smooth film for microelectronic layers or new solar panel surfaces? It helps when the volatile precursor leaves almost nothing behind except the metal oxide you’re after. It isn’t just academic: any shortcut that means fewer impurities on a chip helps push device performance past yesterday’s limits.
Sparking Up Analytical Chemistry
Trace analysis of metals, especially in environmental work, has a throughline running back to metal chelates. Analytical chemists throw trifluoroacetylacetone into the prep when they need to boost sensitivity or clean out noise. By forming a strong complex, the metal becomes less likely to disappear in the mess of extraction procedures. Chromatography and spectroscopy techniques, whether for soil testing, food monitoring, or water safety, get a boost in reliability. The method has stuck around because it works, even as technical names and machines change over time.
Not Just for the Mega-Lab
Practicality matters in the real world. Labs and factories often look for reagents that combine reliability, a reasonable shelf life, and predictable results. Trifluoroacetylacetone sticks out for holding up under harsher conditions and opening up options where more sensitive chemicals fail. While it is not all-purpose or the safest thing in the cabinet, institutional know-how goes a long way in limiting risks. I remember colleagues drilling lab newcomers on venting, storage, and clean-up just because a steady hand here means smoother experiments down the line.
Where the Road Leads
The chemical industry and research world will always see new challenges, from cleaner electronics to counting atoms in underground water. Reagents like trifluoroacetylacetone aren’t just legacy holdovers—they enable scientists to push into new spaces, try bolder separations, and save time on laborious purification routines. If anything, its role shows that behind every breakthrough, some of the most reliable helpers don’t make it into the headlines.