Trifluoroethyl Methacrylate: A Closer Look at Its Journey and Significance
Historical Development
Trifluoroethyl methacrylate didn’t appear overnight. Its path traces back to the mid-20th century, when chemists turned their attention to fluorinated compounds for new performance materials. The introduction of fluorine atoms into organic frameworks opened a new chapter in materials science. Polymers derived from these monomers offered impressive durability, low surface energy, and distinct chemical resistance. By the late 1960s, as acrylic polymers had already changed the coatings industry and dental practices, researchers expanded their scope, finding the value of fluorine’s presence in methacrylates. People who worked in early labs remember the skepticism — why introduce such expensive atoms? In time, answers became clear: properties like stain resistance, hydrophobicity, and improved weatherability could not be ignored. This monomer gradually found its way into specialty applications, no longer just a curiosity for the lab shelf but a tool for engineers and chemists seeking solutions that mere methyl or ethyl methacrylates couldn’t provide.
Product Overview
As someone who has watched specialty chemicals enter mainstream markets, it strikes me how trifluoroethyl methacrylate (TFEM) moved from being a niche monomer to a staple for advanced coatings and dental adhesives. Unlike standard methacrylates, TFEM brought something new to the table. Its key feature is the trifluoroethyl group, adding both chemical stability and practical value. Companies turned to TFEM when weather and solvent resistance mattered, when ordinary polymers failed. Seeing the growth of this product in dental care, protecting teeth from stains, or in high-end automotive coatings speaks volumes about its adoption.
Physical & Chemical Properties
No one who has uncapped a bottle of TFEM will forget its sharp, almost penetrating odor — a sign of both volatility and the underlying methacrylate backbone. Its clear, colorless appearance belies the unique properties beneath. The addition of three fluorine atoms drastically drops the surface energy compared to most conventional methacrylates. TFEM forms flexible but tough polymers that resist dirt, oils, and water, and shrug off harsh environmental conditions. It boils at a lower temperature than side-chain alkyl methacrylates, and its density outpaces many acrylic monomers. Its vapor pressure and viscosity make it challenging but rewarding to handle for those aiming to synthesize copolymers or to formulate high-performance surfaces.
Technical Specifications & Labeling
Anyone handling specialty monomers learns quickly that clear labeling and strict quality control save time and trouble. TFEM typically comes with purity above 98 percent, and reputable manufacturers provide batch analysis to avoid inhibitors being left unchecked. Labels call out the need to keep this liquid cool and protected from light, since polymerization can kick off spontaneously if left uncapped or stored improperly. It’s not an ingredient you find in everyday household products, and there’s a reason safety information gets stamped on every drum. For those working with this monomer, reading a label is never just a formality; it’s part of the daily workflow.
Preparation Method
TFEM isn’t just whipped up with common reagents. Its preparation relies on methacryloyl chloride and 2,2,2-trifluoroethanol, in the presence of a base like triethylamine to mop up the resulting hydrochloric acid. This route delivers good yields, but lab workers know to keep a steady hand and steady nerves — methacryloyl chloride brings its own hazards, and the process needs to stay dry and controlled. It strikes me how advances in reaction engineering improved not only safety but also purity, meaning today’s monomer is a safer bet for research and manufacturing than it was decades ago.
Chemical Reactions & Modifications
TFEM’s true value often appears when it gets added into copolymers. Its methacrylate group lets it join free-radical polymerizations alongside regular acrylic or vinyl monomers, but the trifluoroethyl group sets it apart in the resulting materials. Chemists who spend days chasing hydrophobicity or seeking lower optical refractive indices often find success incorporating TFEM, as its structure can tune the performance of both hard and soft segments in the final polymer. Researchers in adhesives, dental composites, and even electronic coatings exploit this chemistry, modifying resins for their unique combination of resistance and processability. It’s no surprise that as green chemistry gains ground, people started looking for new catalysts and reaction conditions to use TFEM more safely and with less environmental impact.
Synonyms & Product Names
TFEM isn’t a household name, but it does turn up under a few identities. Chemists sometimes call it 2,2,2-Trifluoroethyl 2-methyl-2-propenoate, or simply TFE methacrylate. Catalogs list it side by side with other trifluorinated acrylate and methacrylate monomers, each promoting new ways of making surfaces repel stains or increasing the glass transition temperature of plastics. It’s not the kind of thing you stumble across outside a research or an industrial setting, but within those spheres, this monomer’s name carries certain expectations for performance and utility.
Safety & Operational Standards
Having spent time in wet labs, I’ve seen how fast a routine polymerization can turn into an emergency if inattentiveness seeps in. Handling TFEM comes with non-negotiable rules. The sharp smell isn’t just an annoyance; vapor inhalation can irritate airways, and skin contact risks burns or sensitization. Proper ventilation, gloves, and eye protection become second nature, and storage in tightly-sealed, inhibitor-treated containers prevents unwanted runaway reactions. It helps to remember that regulatory bodies look closely at occupational limits and disposal, requiring every user to document training and be ready for spill or exposure events. Those who ignore safety guidelines sooner or later wish they hadn’t.
Application Area
I’ve watched TFEM leap from a specialty monomer for research to a regular ingredient in dental adhesives, medical device coatings, and electronics encapsulants. Dentists, in particular, value its non-stick and stain-resistant qualities, helping composite fillings stay bright longer. In the automotive sector, TFEM turns up in clear coats where water, road debris, and harsh sunlight would otherwise dull that fresh-off-the-lot shine. Electronics manufacturers use it in advanced protective layers, fending off moisture and corrosion. The monomer’s unique package of attributes means it shows up wherever survival in difficult environments is non-negotiable. Each application tells a story of trial, error, and eventual success — boosting performance standards in industries that don’t tolerate failure.
Research & Development
It’s no secret that research teams worldwide keep testing new ways to use TFEM, pushing beyond simple surface coatings or dental resins. Copolymer formulations now aim for finer control over hardness, flexibility, and surface energy, opening possibilities for everything from touchscreens to long-lived medical implants. Advances in curing methods, greener production, and nanotechnology-based blends crop up in patent filings and journal articles, driven by the quest for safer, longer-lasting materials. Years ago, TFEM was just a promising addition to the chemist’s toolkit. Now, as these multifunctional materials mature, collaborative research between academic labs and industrial partners has picked up pace, sharpening the focus on applications that weren’t even on the horizon a decade ago.
Toxicity Research
Every chemical deserves a close look at its health and safety profile, and TFEM is no exception. Current studies point to relatively low acute toxicity, but prolonged or unprotected exposure might carry risks for respiratory and skin sensitization. Regulatory agencies in the US, EU, and Asia have reviewed the available animal and in vitro data as they update workplace safety guidelines. Even though TFEM has not shown persistent bioaccumulation, its breakdown products include small amounts of methacrylic acid and trifluoroacetic acid, both flagged for monitoring in wastewater streams. In my experience, no research director wants to get caught short on toxicology, and so larger labs include TFEM in their panel of routinely-monitored workplace chemicals. Gaps remain, such as comprehensive chronic exposure studies in humans, an area likely to attract further research as usage continues to rise.
Future Prospects
Reflecting on the growing need for better, safer, and smarter materials, TFEM’s trajectory seems secure. As renewable energy, electric vehicles, and medical technology demand materials with exacting standards, the role for fluorinated monomers and their polymers is unlikely to shrink. The biggest challenge lies in balancing performance with environmental concerns, especially as the world scrutinizes the persistence of halogenated compounds. Ongoing research into degradable fluorinated polymers and safer alternatives signals that the story of TFEM is still evolving. Regulatory pressure and public demand for sustainability will shape how researchers and manufacturers use these materials. Those with a stake in coatings, adhesives, or advanced polymers have every reason to keep watching — and contributing — to the next chapter.
Chemistry Shapes the Everyday
Chemistry likes to hide in plain sight. Trifluoroethyl methacrylate—a mouthful to pronounce—doesn’t get much attention from anyone outside specialty manufacturing or research. Yet pretty much everyone living in a modern home benefits from the stuff. I worked in a lab where polymers ruled the day, so I’ve seen how tiny changes in a chemical’s structure can transform entire industries. This compound carries a trifluoromethyl group which sounds exotic on paper, but really it just means new possibilities for plastics.
What Makes Trifluoroethyl Methacrylate Useful?
Since polymer chemistry took off decades ago, adding fluorine atoms to plastics became the shortcut to better performance. Water beads up and rolls off, nothing sticks, and materials last longer under heat or UV. People depend on these features in places they rarely notice: phone cases, coatings on medical devices, cable insulation, dental fillings. Trifluoroethyl methacrylate lets manufacturers bake in these upgrades by swapping it into their recipes.
The trifluoroethyl group brings low surface energy. I saw waterproof coatings made with it keep electronics alive in spills nobody remembers but everyone fears. In hospitals, equipment needs to resist sticking and bacterial growth; this compound shows up in finishes on surgical tools for exactly that reason. It improves chemical resistance, which keeps plastics from breaking down when scrubbed or exposed to harsh cleaning agents.
Dental Work, Electronics, and Beyond
One place this chemical makes a surprising impact: dental care. Many white fillings use resins built on methacrylates. Adding trifluoroethyl methacrylate to the mix stops fillings from absorbing stains and swelling up, extending how long someone’s dental work lasts. After watching friends deal with cracked, leaky fillings, I pay attention to which resins dentists use now.
Electronics designers love fluorinated polymers for insulating wiring. Trifluoroethyl methacrylate finds its way into cable coatings where reliability matters—think medical devices, mission-critical sensors, or aerospace wiring harnesses. Because these coatings don’t melt easily or soak up moisture, sensitive electronics keep on working in humid, hot, or unpredictable environments.
Balancing Innovation with Safety
Of course, nobody should sprinkle new chemicals into products without thinking twice. Reports of environmental persistence with certain fluorinated chemicals raised alarms in the past. Over time, better guidelines have emerged for handling, tracking, and disposing of materials that could linger outside the lab. Regulation walks a fine line: keep people safe without choking off innovation.
Chemists now look for ways to combine performance with environmental responsibility—something I saw start to happen in labs a decade ago. Companies invest in safer production routes, adoption of green chemistry principles, and tighter rules on how materials get used. Academic labs chase biodegradable alternatives, but the right substitution doesn’t always show up overnight.
What Happens Next
Every year, more industries look for materials that do more with less risk. Trifluoroethyl methacrylate’s popularity grows because it helps companies deliver on those promises, as long as they use the safeguards we’ve learned are necessary. Tools get stronger, dental work looks better, and electronics survive rainstorms—not by accident, but because the chemistry behind the scenes keeps pushing forward.
Why This Chemical Commands Respect
Trifluoroethyl methacrylate may sound like a mouthful, but to those working in labs and industries, the name carries weight. To see it, you’d think it’s just another bottle on the shelf—clear, with little odor, unassuming. My experience working as a research assistant during university drilled into my head that these unremarkable-looking compounds usually demand the most careful handling. This one, with its reactive double bond and fluorinated side group, brings hazard with little warning.
Personal Protection: No Corners to Cut
Most people don’t reach straight for gloves and goggles at home, but in any lab, you learn it fast. A splash in the eye or on skin from acrylates like this can burn, sensitize, or worse. Trifluoroethyl methacrylate doesn’t offer forgiveness. Respect starts with proper gloves—nitrile over latex, since some chemicals cut through latex in seconds. Safety goggles feel bulky, but you only need a reminder from an accident in the news to snap them on every time. Lab coats, buttoned sleeves, sometimes a face shield if the risk of splashes rises. Not theater—just common sense.
Ventilation: Dilute the Risk, Don’t Inhale It
You can’t trust your nose. Fluorinated methacrylates put vapors in the air long before you notice. These vapors sting the lungs and irritate the lining of your airways. I remember a colleague who shrugged off working outside the fume hood; she ended up nursing a cough for a week. Always treat volatile monomers with real ventilation—fume hoods aren’t just for show. Keeping exposure below recommended limits doesn’t happen by guesswork; it comes from engineering controls working in the background.
Spills and Waste: Handle the Unwanted, Too
A spill can turn a busy morning into a circus. Fluorinated acrylates stick and spread, so rapid cleanup with dedicated absorbents beats paper towels. Dispose of waste in proper containers with tight seals. Leaving it in an open beaker on the bench out of laziness overlooks flammability and slow emission of fumes. Regular reminders in group meetings about waste protocols make a difference. People remember stories, not signs posted on the wall.
Fire Risk: More Than Just Flammable
Some chemicals burn hot and fast, and trifluoroethyl methacrylate fits that category. Once ignited, fluorinated chemicals sometimes create harsh combustion products. I recall one lab fire drill where we checked extinguishers for suitability; no one wanted to chase down a chemical fire with water and end up with the mess of toxic runoff. Labs need the right extinguishers, not just the nearest one.
Beyond the Rules: A Culture of Safety
No matter how detailed a safety protocol reads, building habits forms the real backbone of safety. From talking with experienced chemists, a consistent thread appears: they all had stories of near-misses, and each changed their daily routines because of them. It’s not the printed rules that save you, but the attitude you bring in every time you crack open a new bottle. Sharing those stories during group training goes further than any mandated online quiz. Safety sticks when it becomes part of the culture as much as part of the paperwork.
The Chemistry Behind the Name
Even folks who never worked in a chemistry lab have seen the impact of chemical building blocks like trifluoroethyl methacrylate. You may not realize it, but this compound plays a role in products you see every day, from dental materials to specialty coatings. Let’s break down what trifluoroethyl methacrylate actually is and why its molecular details matter for science and industry alike.
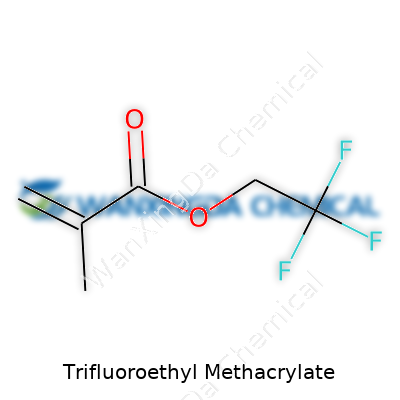
Getting to the Formula: C7H9F3O2
The formula for trifluoroethyl methacrylate is C7H9F3O2. Here’s what that means: the compound has seven carbon atoms, nine hydrogens, three fluorines, and two oxygens. These numbers do more than fill a spot in a database — they signal to chemists what’s possible for this molecule. That trio of fluorine atoms gives it a distinct character, making materials more resistant to water and chemicals. In my own lab experience, swapping a single atom, like trading a hydrogen for a fluorine, could push a polymer from ordinary to extraordinary in terms of durability.
What the Structure Tells Us
Digging beneath the formula, the architecture of trifluoroethyl methacrylate shapes its uses. Picture this: a methacrylate backbone, which has a double bond, links to a trifluoroethyl side chain. The double bond opens doors for the molecule to join up with others through polymerization, a key step in creating advanced plastics and resins. The trifluoroethyl group includes a -CF3 unit, known for making bonds tough and resistant to breakdown.
I’ve mixed plenty of monomers through the years, and every time, I saw how even the small trifluoro group could shift the way the final material repelled water or shrugged off harsh cleaning agents. In settings where you want a long-lasting dental filling or a slick, nonstick surface, a structure like this makes all the difference. Data from peer-reviewed studies confirms that just a few fluorine atoms lower the surface energy of finished polymers, which helps prevent things from sticking or staining.
Why This Matters for Everyday Life and Industry
When chemists introduce trifluoroethyl methacrylate into a product, it’s usually to add toughness or to reduce how much water it soaks up. That makes it valuable in biomedical devices, paints that last through rough weather, or coatings where resistance to solvents can be a dealbreaker. In my university project, I watched coatings based on this monomer stand up to acids that wrecked the control samples. Several manufacturers lean on these properties for medical adhesives, which rely on staying bonded under constant stress and moisture.
Looking Ahead: Innovation Without Compromise
The growing demand for materials with advanced capabilities means trifluoroethyl methacrylate has found a lasting place in the chemist’s toolkit. Still, just because something works doesn’t mean we should ignore the potential for environmental harm. Fluorinated materials linger in the environment, drawing attention from regulators and research teams. Many scientists, myself included, are interested in developing greener approaches to creating and breaking down these compounds. Biodegradable alternatives could preserve performance without leaving a permanent mark, and the race for safer chemistry remains a fixture of every good lab I’ve known.
Working with chemicals like trifluoroethyl methacrylate (TFEMA) always keeps me on my toes. This isn’t one of those materials you forget about on a shelf just because it looks harmless in a sealed drum. Handling TFEMA takes real respect for safety and practicality. Every supervisor I’ve known who spent any time near a polymer lab or coatings facility will have a story about what happens when someone shrugs off basic storage guidelines. Nobody needs a messy lesson about flammability or a ruined batch due to contamination.
TFEMA brings a double threat: it’s both highly flammable and reacts badly with moisture. Any time I’ve been around it, official safety standards weren’t just lines in a book — they kept everyone’s mind at ease. You want TFEMA in a cool, dry place, no sunlight creeping through the windows, and definitely no hot water pipes running overhead. Heat speeds up polymerization, turning shelf-stock into a concrete-like mass that can wreck expensive equipment and delay production schedules. The last time I saw a colleague slack off on temperature controls, we ended up tossing out a sealed container that ruined itself in storage.
Humidity poses just as much trouble. Trifluoroethyl methacrylate will react with moisture in the air, giving you byproducts nobody wants. Even trace water vapor floating in the room can compromise the quality of the chemical, so tight lids and proper seals aren’t optional extras. I’ve noticed some newer labs moving to nitrogen-blanketed cabinets for storage, because it’s easier (and a lot cheaper in the long run) to keep air and water away from the raw material than to reverse bad chemistry. That approach reflects stubborn experience — too many ruined shipments, and people start thinking ahead.
Simple Steps for Safety
The basics make all the difference. Stainless steel or high-grade plastic containers don’t leach contaminants. Shelves rated for chemical storage beat out ordinary plywood, especially in buildings that get humid in the summer. Every good lab or warehouse puts clear labels on every drum, keeps incompatible materials on separated shelves, and enforces rules about opening containers only in ventilated areas. On top of that, grounding and bonding reduce static sparks, which matters in places juggling volatile liquids. Fires don’t care about intentions — one tiny lapse can bring out fire marshals and insurance adjusters in a hurry.
Personal responsibility shapes the safety culture. I’ve noticed that folks who get training in chemical hazards usually respect the rules. A regular safety refresher and posted reminders go further than a thousand-page manual nobody reads. I remember a plant manager who would quiz everyone about storage temperatures during team meetings; it was annoying at first, but it drove the point home stronger than a poster could.
What Needs Fixing?
If there’s room for improvement, it’s in the area of automation and monitoring. Temperature loggers that send alerts to your phone catch problems before they grow legs. Installing smart sensors for humidity gives a lab edge in busy seasons, especially if foot traffic and outside weather change room conditions. Costs may seem high upfront, but compared to the price of lost product or safety violations, automation almost always pays back its investment fast.
So, storing TFEMA is more than just following a protocol. It’s a habit, a mindset, and a team effort. Everyone from warehouse techs to lab supervisors shares responsibility for keeping things safe, efficient, and trouble-free. The stories I’ve witnessed remind me why nobody treats chemical storage as an afterthought.
Behind the Chemistry: Why Trifluoroethyl Methacrylate Matters
For those working with high-performance coatings, adhesives, and polymers, trifluoroethyl methacrylate isn’t just another name on an inventory list. This specialty chemical gives everyday products the power to resist stains, shed water, and outlast tough environments. Growing up around a small auto repair shop, I used to watch old car seat fabrics get slick with grease and stubborn stains. Cleaner and polish only did so much. Over the years, change swept through these industries. Now, products last longer, shed dirt, and make cleanup less painful—many times thanks to molecules like trifluoroethyl methacrylate quietly working in the background.
Turning Ordinary into Extraordinary: Key Industrial Roles
Trifluoroethyl methacrylate stands out in the world of specialty acrylics. Coatings made with this compound shield everything from electronics housings to aircraft panels. The fluorine atoms in trifluoroethyl methacrylate don’t just sound fancy; they turn a regular plastic surface into a slippery, easy-to-clean shield. This benefit doesn’t disappear after the first rainy day or cleaning with a harsh solvent. Factories making electronics casings or outdoor signs lean on these polymers to build products that won’t fade fast or crack at the first sign of stress.
Paints and varnishes pick up another trick here: when a formula includes trifluoroethyl methacrylate, water beads up and rolls off. This doesn’t just make cleaning easy—it slows down corrosion, especially on metal. As someone who’s seen rust chew through tools and outdoor railings, I can say these advances do more than keep things pretty; they save money and cut down on waste.
Modern Challenges Demand Smart Adhesives
Walk into any hospital or laboratory, and every surface fights an endless battle against microbes, stains, and harsh cleaning routines. Scientists add trifluoroethyl methacrylate to adhesives for medical devices and cleanroom equipment, where its non-stick power keeps tape from peeling and materials safer to touch. In dentistry, it gets used in fillings and sealants for the same reasons, letting people walk away with a stronger, longer-lasting fix. This comes from fluorine’s natural resistance to chemicals and stress—a small tweak on the molecular level making a big difference on the patient’s experience.
Solving Limits in Electronics and Energy
In electronics, where precision drives everything, surface static and moisture quickly spell disaster. This is where trifluoroethyl methacrylate steps up, building robust dielectric layers for circuit boards and flexible displays. Out in the field, solar panels collect more power and last longer by using coatings based on fluoroacrylate chemistry, shielding glass and plastic from damage. These tweaks sound simple, but they keep devices working as promised, even where weather and dust push materials to their limit.
Towards Better Chemical Choices
Trifluoroethyl methacrylate’s benefits come with questions, too. How do we balance the lasting strength of fluorinated plastics with environmental impact? Industries watch regulations, scrutinize chemical footprints, and invest in recycling programs to keep ahead. The next big step lies not just in making things tougher or easier to clean, but in finding ways to recover, reuse, or reinvent these advanced materials—ideas that could shape factories and laboratories for decades.