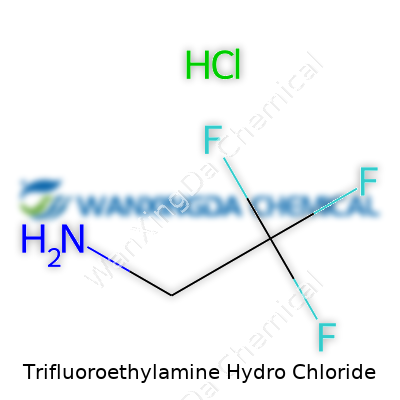
Trifluoroethylamine Hydrochloride: A Close Look at Progress, Practice, and Possibilities
A Walk Through Its History
Chemists began exploring the world of organofluorine compounds early in the twentieth century, running into the curious properties of trifluoromethyl groups as laboratory synthesis gained steam. Trifluoroethylamine hydrochloride later emerged as a chemical bred from these early efforts, bridging disciplines with its potent blend of reactivity and stability. People in labs, constantly searching for new scaffolds for drugs, materials, and catalysts, noticed how the introduction of –CF3 units often gave molecules new life—changing their boiling points, how they handled moisture, or their resistance to enzymes and breakdown. By the late twentieth century, steps to prepare trifluoroethylamine hydrochloride held the interest of both synthetic chemists and companies, who saw its value both as a building block and as a protected amine for assembly-line chemical processes.
Product Overview
Every time I’ve handled trifluoroethylamine hydrochloride, I've been struck by its utility. The powder, usually white or almost white, brings a pungent but not overwhelming odor; it taps into that familiar tanginess found in many amine hydrochlorides but carries a sharper note, which I later learned comes from the fluorines. The solid dissolves well in water and polar organic solvents, and once in solution, its ions rarely cause trouble, keeping reactions straightforward. What sets this compound apart for chemists is that it acts as a mild nucleophile with the trifluoroethyl group offering a balance—enough electron-withdrawing pull to tweak reactivity, not so much to make things unpredictable.
Physical, Chemical Properties, and Technical Benchmarks
As far as physical measurements go, trifluoroethylamine hydrochloride doesn't shock anyone: moderate melting point, good solubility, and a modest vapor pressure mean it stays out of trouble during routine shipping and handling. The hydrochloride salt stabilizes the otherwise volatile amine, and that simple choice makes life easier for whoever opens the bottle. Chemically, the compound stands up to most routine acids and bases; it resists oxidation better than less-fluorinated cousins, and in my experience, it rarely causes extraneous side reactions in multi-step organic synthesis. Most reputable suppliers ship it with detailed labeling, batch information, and purity grades; high-purity material offers few surprises in terms of residue or water content.
Preparation and Synthesis
Making trifluoroethylamine hydrochloride in the lab means working with a few common building blocks—often starting with trifluoroethanol or related derivatives, converting these through amination (sometimes via azide intermediates or by reductive amination techniques), and finally treating the resulting amine with hydrochloric acid gas or solution. The sequence needs careful attention to stoichiometry and temperature, but it remains within the reach of a skilled organic chemist. Scale-up introduces new wrinkles: batch reactors must be vented properly due to ammonia or other gaseous byproducts, and good containment matters, since both fluorinated byproducts and HCl gas can be rough on equipment.
Chemical Behavior and What Chemists Can Do With It
If there’s a charm to trifluoroethylamine hydrochloride, it’s in how flexibly it reacts. Once the hydrochloride group is liberated (a simple base treatment will do), the free amine partners easily in alkylation and acylation steps, or it can be fed into heterocycle-forming chemistry for substances that need a trifluoroethyl arm. Because the –CF3 group draws electrons, the amine isn’t as wild as many unsubstituted alkylamines; it behaves in a more controlled fashion with electrophiles. Fluorinated amines like this often work as precursors for specialty pharmaceuticals—including antiviral and anti-inflammatory drugs—or for fluorinated surfactants and specialty polymers. Amide coupling, urea formation, and reductive alkylations all take advantage of its properties.
Alternate Names and Product Synonyms
Over the years, I’ve seen trifluoroethylamine hydrochloride show up in catalogs under names like 2,2,2-trifluoroethylamine hydrochloride and TF-EA•HCl. Sometimes old research papers or catalogs from outside the U.S. refer to it by its CAS number or IUPAC designation, but the structure—CF3CH2NH2•HCl—always gives it away. Identifying the right compound depends on cross-checking the structure, not just picking the name from a list.
Safety Notes and Good Lab Practice
Anyone working with amines and hydrochloride salts has stories about strong odors, skin or respiratory irritation, and sometimes spill headaches. Trifluoroethylamine hydrochloride isn’t exceptionally hazardous, but it should never be underestimated. Gloves, goggles, and use of a well-ventilated fume hood aren’t optional. I make a habit of weighing and dissolving this amine behind a sash to dodge any airborne dust. The hydrochloride form cuts down volatility, a small mercy in long syntheses, but disposal requires careful management, especially since fluorinated compounds persist in the environment. Reporting accidental releases matters to environmental and health agencies, as regulations tighten around halogenated waste.
Real-World Uses and Current Research
Medicinal chemistry likely owes some of its progress on new small-molecule drugs to building blocks like trifluoroethylamine hydrochloride. The addition of fluorine can improve the metabolic stability and oral absorption of drug candidates. Some leading-edge antidepressants, antivirals, and enzyme inhibitors use trifluoroethyl groups to evade breakdown and boost activity. Beyond medicine, industrial chemists use the compound to help create specialty surfactants, textile treatments, and coatings with hydrophobic or oleophobic properties. The research angle has only grown—once high-throughput screening became standard in the pharmaceutical industry, new analogs with trifluoroethyl substituents showed up in library after library. Academic labs keep exploring how the unique size and electronics of trifluoroethyl groups shift biological activity or physical profiles, giving rise to patents and journal articles every year.
Toxicity and What’s Missing
Attention to the toxicology of fluorinated amines trails behind more common substances—a real weak spot considering their growing use. What we do know draws from animal studies and acute exposure reports: trifluoroethylamine hydrochloride irritates skin, mucous membranes, and eyes. Chronic data stay sparse, and the long-term fate of small organofluorines in the body or environment raises questions researchers still haven’t fully answered. Environmental scientists flag persistence as a shared challenge among fluorinated organics, warning that monitoring and waste management might need more rigorous oversight in manufacturing and laboratory settings. Calls for better metabolic studies, more granular exposure modeling, and harmonized handling standards keep ringing out in safety meetings and regulatory discussions.
What’s Next? Future Routes and Responsibilities
Fluorinated building blocks like trifluoroethylamine hydrochloride promise much with each tweak chemists devise. Future research points toward milder, more efficient preparation routes, greener alternatives to wasteful steps, and wider profiling of human and ecological impacts—a necessity as regulatory bodies sharpen focus on persistent organic pollutants. Pharmaceutical innovation continues pulling this compound forward, as new ways to block metabolic enzymes or shift solubility profiles keep researchers experimenting at both the benchtop and process scale. The big-picture challenge hangs over everyone using fluorinated small molecules: weigh the benefits of new chemistry against possible long-term fallout in the environment. As access and interest climb, tracking the lifecycle of these materials and staying committed to transparent research matter more than ever.
Some chemicals sound like they belong in science fiction novels. Trifluoroethylamine Hydrochloride might have that kind of aura at first glance. In reality, its role isn’t just technical; it has true impact on health, innovation, and the way research pushes medicine forward.
A Building Block in Modern Drug Discovery
Labs across the world use Trifluoroethylamine Hydrochloride for synthesis in pharmaceutical research. As drug makers look for ways to tackle tough diseases like cancer and resistant infections, they need building blocks that can add both stability and new properties to potential medicines. Trifluoroethylamine Hydrochloride helps create “fluorinated” molecules—a class known for improving how drugs act in the body.
Adding fluorine to a molecule often isn’t just for show. It can increase how long a drug stays effective, how easily it travels through the blood, and how it interacts with its target. Trifluoroethylamine Hydrochloride plays a part in this by reacting with other chemical pieces, helping scientists design drugs that aren’t easily broken down by enzymes. That means better doses, fewer side effects, and for some conditions, a shot at new therapies where old ones have failed.
The Power of a Small Change
I’ve watched research teams struggle with molecules that work in a test tube but fail inside the body. Weak stability in real-world conditions can end years of work. Fluorine atoms are picky, hard to fit onto molecules, but their effect is dramatic. Trifluoroethylamine Hydrochloride gives researchers a way to bring these atoms in, making it possible to fine-tune a drug’s chemistry without completely starting over.
Big pharmaceutical breakthroughs often come from small tweaks. Think about antiviral drugs that stick around longer in the bloodstream—many of those advances rest on clever chemistry, much like what Trifluoroethylamine Hydrochloride enables. Adding it at the right point can make or break a new treatment’s chances.
Challenges and Responsible Handling
No progress comes without bumps in the road. Trifluoroethylamine Hydrochloride demands careful handling; its reactivity, while useful, also means mistakes lead to dangerous byproducts. Chemical waste and exposure risk aren’t problems for someone else to solve. I’ve seen responsible labs invest in good ventilation, solid training, and clear waste protocols—not just for the law, but out of concern for everyone nearby.
The chemicals that help push the boundaries of what’s medically possible can also spill into groundwater, seep from waste streams, or cause immediate harm if mishandled. Stronger local monitoring, regular hazard training, and updated handling guidelines help keep risks in check. These investments reduce accidents and make sure progress doesn’t come at the price of a technician’s health or a community’s water supply.
Looking Forward
Research won’t stall. Chemists look for even safer, cleaner ways to achieve the same breakthroughs. Green chemistry—an idea as much as a toolkit—drives teams to cut down on waste, swap out raw materials, and recycle wherever they can. Trifluoroethylamine Hydrochloride remains a valuable tool in making next-generation drugs, as long as those who use it remember both its promise and its demands.
Paying Attention to Safety in Chemical Storage
Anyone who has ever worked around specialty chemicals knows that storage isn’t a minor afterthought. I've stood in labs with shelves stacked far too high, with jars crowding each other, and it’s crystal clear that the stakes aren’t small. With a reagent like trifluoroethylamine hydrochloride, the game isn’t just about preserving purity—it’s about keeping people safe, complying with real rules, and protecting our bottom line from preventable losses. Mess up, and you’re not just looking at wasted material. You risk health hazards, trashed equipment, and liability that’s hard to stomach.
Why Storage Choices Matter for This Compound
Most folks who handle laboratory chemicals know that air and moisture are not friends with hydrochloride salts—trifluoroethylamine hydrochloride included. It pulls water out of the air, and as it does, it starts to degrade and clump, making dosing impossible and results unpredictable. That’s not a minor inconvenience; it throws off reactions, wastes time, and, if you run a regulated operation, opens the door to compliance trouble. I’ve seen projects go on hold for weeks because one shipment turned solid from poor storage.
Cold, Dry, and Secure: Simple Rules, Big Payoff
Getting this right doesn’t require complex tech. It starts with a tightly sealed container—preferably a glass bottle with a PTFE-lined cap. I don’t cut corners here, because the cost of using a subpar closure shows up sooner or later. Storage in a cool, dry place is key. By “cool,” that often means room temperature, away from direct sunlight or any heat sources. Moisture is a quiet enemy, so desiccators or cabinets with drying agents do more than just look serious; they serve a definite purpose. If the space feels damp or the walls sweat in summer, it’s time to find another spot.
Keeping the People Safe, Too
Experience has shown me that even the most stable-looking amine salts can turn nasty in the wrong hands. Gloves, goggles, and proper labeling are non-negotiable. If someone pops the lid in a humid storeroom and walks away, the compound pulls in water and starts to cake or, worse, decomposes. Then, what seemed like routine work turns into a clean-up headache or a call to poison control. I’ve learned, sometimes the hard way, that putting up clear signage and running quick training refreshers prevents most mishaps.
Getting It Right Isn’t Optional
Mistakes in chemical storage carry real consequences. The EPA, OSHA, and even local fire codes all have their say. Regulatory bodies publish guidelines for a reason—explosions and toxic releases didn’t get there by accident. Compliance isn’t just about avoiding fines. It keeps the peace in the lab and gives people confidence that their workspace won’t turn on them.
Solutions: Simple Steps for Lasting Results
Routine checks matter. Take a walk through storage each month and check seals, labels, and conditions. Assign responsibility—make it personal. Digital logs make things smoother and leave less to chance. Invest in good storage infrastructure—a well-designed chemical cabinet, backup power for refrigeration, and enough desiccant to go around. If the lab grows, rethink layout before disaster forces your hand.
In the end, chemical storage isn’t mysterious or glamorous, but it’s a foundation that every safe and reliable operation rests on. Trifluoroethylamine hydrochloride is just one example, but the habits built keeping it safe reflect directly on everyone’s wellbeing and peace of mind.
Understanding the Formula and Weight
Trifluoroethylamine hydrochloride shows up in lab orders and research projects with its own tongue-twisting name. Strip away the jargon, and it boils down to a compound with some unique chemistry behind it. The chemical formula is C2H5F3N · HCl. That tells us each molecule bundles together two carbon atoms, five hydrogens, three fluorines, one nitrogen, plus a chunk of hydrochloride. If that seems like a mouthful, it is—but each piece plays a part in how this compound behaves when mixed or reacted in a lab.
Folks in chemistry measure things down to the last decimal, because a gram misplaced can throw off an entire project. The molecular weight of trifluoroethylamine hydrochloride clocks in at about 153.53 g/mol. That number doesn’t just sit in a textbook; it guides how much gets weighed out for reactions, influences yields, and helps determine how reactions scale up. The small decisions—measuring out the right amount for a flask, adjusting for purity—hinge on understanding this weight.
Why the Chemical Makeup Is More Than Trivia
Some would ask why this matters outside a lab. Chemistry doesn’t stop at the workbench. Take pharmaceuticals. Compounds like trifluoroethylamine hydrochloride form building blocks in new drug discovery. Accuracy in formula and mass helps dial in dosage and track side effects. One wrong digit can set a whole batch astray, bumping risks for patients or stalling a clinical trial. In industries dealing with agrochemicals or materials science, those numbers shape how well a product works or how safe it runs through production lines.
Public trust in new treatments or safe materials hinges on solid science. Every published result, every patent filed, leans on knowing exactly what sits in the flask and how much of it’s there. I’ve seen over-eager shortcuts backfire, with batches scrapped and time lost, all from skipping over step one: confirm the formula and the weight.
A Focus on Precision Brings Better Solutions
Leaping into big research goals without understanding foundational details usually leads to frustration. More often than not, labs see run-ins with contamination or failed synthesis because someone skipped rechecking chemical formulas or weights. I’ve spent time triple-checking molecular weights on shaky early-morning shifts, and that muscle memory pays off. A miscalculation on the scale means wasted reagents—sometimes costly, sometimes hazardous.
One clear solution stands out: focus on training and reliable sourcing. Any lab ordering trifluoroethylamine hydrochloride should crosscheck labels with trusted databases. Sourcing from reputable vendors keeps impurities in check, and digital tools now help verify chemical identities. Open dialogue between suppliers and researchers can also flag inconsistencies before they slow down progress.
Thorough documentation is another ally. Every researcher has stories of trying to reverse-engineer a reaction based on half-written notes. Smart labs keep records tight, and no one scoffs at logging the chemical formula and weight up front. This discipline supports everything that follows, from safety protocols to publishing.
The Takeaway: Small Details, Big Impact
At a glance, trifluoroethylamine hydrochloride’s formula and weight might seem like obscure trivia. Peel back the surface, and those details keep experiments tight, products safe, and discoveries moving forward. Building habits around careful measurement and data verification isn’t just nitpicking—it plants the seeds for trustworthy, repeatable science.
Looking Beyond the Chemistry Lab
People like me who’ve worked around synthetic chemicals know the drill: new compound, quick search, scan a safety sheet, move on. Trifluoroethylamine hydrochloride pops up on supplier lists all the time—usually with bland warnings about handling with care, avoid breathing dust, standard stuff. But chemistry can hide bigger problems in plain sight. This particular compound shows the ways we sometimes underestimate risk, just because the chemical name isn’t famous or scary.
The Real Risks Lurk in Use and Misuse
Everyone wants clear answers: is something toxic, or is it not? The catch is, danger depends on both the substance and what people do with it. Trifluoroethylamine hydrochloride contains fluorine, a red flag for reactivity and toxicity. Fluorine compounds connect to the chemistry that built Teflon, but they’ve also powered some of the scariest industrial accidents and long-term health problems, especially in manufacturing and research settings.
This chemical, in solid form, won’t leap out of the jar and hurt you. Add water, mix with acids, or breathe dust in a hurry, plenty could go wrong. Inhalation, skin contact, or eye exposure could irritate, burn, or worsen existing health issues. The stories that stick in my mind come from rushed labs: someone skips gloves, spills a few grams, gets a weird tingling or cough, and only later digs up papers connecting certain organofluorines to nerve, kidney, or liver damage in animal studies.
Regulation Drags Its Feet
People look to government labels for reassurance. Here’s the thing: smaller specialty chemicals often fly under the radar. OSHA and EPA don’t always have reviews or exposure guidelines for newer or less-common lab reagents. Europe’s REACH law keeps an eye on many toxic substances, but there’s no guarantee trifluoroethylamine hydrochloride made that watch list. Information gaps breed overconfidence—folks treat it as just another shelf item.
This lopsided attention isn’t rare. Some fluorinated chemicals, like PFAS, draw headlines because they stick around in the environment and our blood. Others, like this amine salt, escape notice for years. Just because poison control centers don’t get flooded with calls about a chemical doesn’t mean it’s harmless. Risk travels slowly, through cumulative exposure and chronic effects that don’t get traced until much later.
Smart Chemistry Relies on Caution
A smart lab or responsible chemical handler doesn’t wait for a red star in a safety manual. What they do: use strong ventilation, gloves that actually resist organofluorines, proper disposal—never down the drain, never as regular trash. Every experienced researcher I know treats all new or rarely used compounds as potentially harmful until proven otherwise. That’s not paranoia; it’s pattern recognition learned from seeing enough “harmless” substances cause harm.
It helps to build a culture where asking about toxicology brings answers, not rolled eyes. Labs and small-scale manufacturers can partner with industrial hygienists, screen for skin irritation, and keep extra eye-wash stations in reach. Even simple practices, like running practice drills for spills or emergencies, make a difference. Sharing incident stories and updates on lesser-known hazards keeps everyone alert—important, since today’s niche chemical could start showing up in unexpected workplaces or products tomorrow.
Moving Forward With Eyes Open
No single chemical causes all problems. A safe workplace and clean environment depend on plain talk, personal responsibility, and solid, up-to-date information. Chemicals like trifluoroethylamine hydrochloride might not grab national headlines, but their hazards demand day-to-day vigilance, honest conversation, and care for both people and planet.
Purity Isn’t Just a Lab Concept—It Matters in the Real World
I’ve watched chemists scan product catalogs and datasheets, always pausing at one line: purity. When you talk about something like trifluoroethylamine hydro chloride, that number isn’t just a bragging point for suppliers. It tells a researcher or a plant manager whether they can start their work without extra headaches. Usually, the purity on the market sits at 98% or higher. This isn’t just a cherry-on-top feature—a high purity often signals fewer side reactions and less effort wasted in purifying the compound further.
Fluorinated amines—like this one—play supporting roles in drug synthesis, agrochemical development, and specialty materials. If you’re running a reaction that’s finicky about what goes in, a contaminant or two percent of "who-knows-what" can throw off your whole process. That’s money and time down the drain. I’ve met scientists who skip over options below 98% out of caution. One bad batch and a whole research cycle resets.
Packaging Sizes Reveal Who the Real Users Are
Watching buyers at chemical expos, I noticed how their questions about packaging sizes reveal what they’re really planning. Some only ask for small vials—the sort that hold 5 grams or less. Those folks are running tests, developing compounds, or working in tight university labs. Others want bottles, 25 or 100 grams at a time, sometimes more. These are scale-up specialists or those supporting pilot projects drifting toward larger-scale runs.
It’s pretty common to see trifluoroethylamine hydro chloride packed in moisture-tight bottles, sealed under inert gas, and labeled for safety and traceability. That’s not just for show. Even small leaks or decomposing over time can turn a promising project into a safety risk or a dud result. I remember a time when a friend’s team received an improperly sealed sample. Their whole shelf of experiments became question marks overnight, thanks to water creeping inside.
Real-World Impact and Why Choices Matter
Across pharma, crop protection, and other industries, trifluoroethylamine hydro chloride stands as one of those basic toolkit chemicals that needs to be right on the mark every time. Sourcing products with 98% or higher purity lets teams avoid chasing ghosts through complicated analytics just to check if everything is okay. It cuts down frustration and cuts out repeat work.
Some buyers lobby suppliers for even higher purities or guarantee batch-to-batch consistency. That comes with a price, of course. Juggling the budget and technical requirements becomes a negotiation. There are plenty of cases where a cost-conscious approach makes sense—grab the standard purity, check quality on arrival, and move ahead. For cutting-edge research or highly regulated pharma production, though, skimping on purity can spiral into project delays or regulatory problems.
Where Packaging and Purity Cross Paths
In my experience, packaging size isn’t just about the weight or volume. It’s about managing shelf life and knowing how much risk you can afford. If you open a big bottle and can’t guarantee a dry, inert environment, you risk degrading what’s left. That’s one reason suppliers often offer several smaller packaging choices. Smaller packs also mean fewer cross-contaminations and make it easier to store and track.
The story of trifluoroethylamine hydro chloride boils down to something that echoes far beyond a single chemical: buy for your workflow, not just your spreadsheet. Insist on the purity that fits your outcomes, pick packaging sizes that suit your use pace, and don’t treat supply details as an afterthought. It’s often the smallest contaminant or the simplest storage error that upends even the biggest projects.