Commentary: Sodium Trifluoromethanesulfonate—A Chemical with Staying Power
Historical Footprints in Chemical Synthesis
Talking about sodium trifluoromethanesulfonate, or sodium triflate as many researchers call it, means exploring over half a century of organic chemistry progress. Early academic literature points to this compound as a mainstay in laboratories that pushed forward organofluorine chemistry and superacid research. Most folks working in synthesis circles will recognize the way sodium triflate slipped into favor due to its stability and non-nucleophilic properties—a breakthrough compared to the inconsistent results delivered by earlier sulfonate salts. It didn't pop up overnight. Scientists in the mid-20th century searched for alternatives to weak, unstable leaving groups, and the discovery of perfluoroalkyl sulfonates paved the way. Sodium triflate, as the sodium salt of trifluoromethanesulfonic acid, soon became a cornerstone for anyone wanting high-performance ion-exchange and reaction media.
Why Sodium Triflate Matters in Laboratories
Speaking from countless syntheses involving nucleophilic substitutions and cross-coupling reactions, sodium triflate always delivers the reliability needed for consistent results. Its robust thermal and chemical tolerance means no nasty surprises during scale-up. Sodium triflate comes as a white crystallized powder, soluble in most polar organic solvents—something every chemist values when setting up multistep reactions. In my own work, a bottle on the lab bench usually goes empty long before anything else, thanks to its wide-ranging applications. For every synthetic organic chemist, sodium triflate represents a simple way to introduce triflate ions without resorting to volatile or corrosive reagents.
Physical and Chemical Features to Know
The straightforward, salt-like nature of sodium triflate gives it a practicality rare among specialty reagents. Melting above 250°C and highly stable under normal temperatures, it resists hydrolysis and maintains integrity in both acidic and basic conditions. This reliability comes from the electron-withdrawing trifluoromethyl group, which strengthens the S–O bond and reduces nucleophilicity. Unlike some more exotic salts, sodium triflate won't degrade during typical storage conditions, avoids caking, and dissolves cleanly. This isn’t some rare, delicate material—just a rugged member of the sulfonate family.
How Scientists Make Sodium Triflate
Most production processes start with triflic acid, itself made by fluorinating sulfuryl chloride in the presence of hydrogen fluoride or other tough reagents. Adding sodium carbonate or sodium hydroxide to an aqueous solution of triflic acid brings about nearly quantitative conversion to sodium triflate. The result precipitates as a solid, which researchers collect, wash, and dry. I’ve followed this protocol, and it offers surprising purity and yield compared to many similar salts. Simplicity matters in the laboratory, and sodium triflate’s direct preparation takes a lot of failure out of the equation.
How Chemists Put Sodium Triflate to Work
The list of chemical reactions that improve with sodium triflate reads like a greatest-hits collection for the modern organic chemist. In Suzuki and Stille couplings, using triflate as a leaving group allows for more flexible substrate choice. Electrochemists benefit from its electrochemical inertness and high ionic conductivity, so you find this compound in supporting electrolytes for batteries and supercapacitors. Peptide chemists tap into sodium triflate to activate carboxylic acids for amide bond formation; researchers developing ionic liquids count on it for its non-coordinating nature. In pharmaceutical synthesis, sodium triflate regularly performs in the background, helping activate molecules at stages critical for drug development pipelines.
Tuning Properties with Chemical Modifications
Chemists love sodium triflate for its ability to serve as a platform for further modifications. Reacting it with various electrophiles creates more complex triflate esters, and swapping the sodium for other cations opens new avenues—lithium, potassium, and even organic ammonium triflates have all carved out their own niches. I’ve seen process chemists tweak these counterions to control solubility, reaction rates, and selectivity, which reflects sodium triflate’s importance not just as a reagent, but as a cornerstone in custom synthesis.
Names in the Literature
Writers and vendors usually call it sodium triflate or sodium trifluoromethanesulfonate, but other names such as sodium trifluoromethanesulphonate or sodium triflyl also show up. Chemical catalogues list the CAS number as a standard reference, which makes tracking sources and purity straightforward. Keeping track of synonyms matters because mislabeling has tripped up more than one shipment or catalog order.
Lab Safety and Industry Standards
Anyone working with sodium triflate should know it doesn’t pack the hazards seen with many strong acids or oxidizers. That said, good sense must prevail. As with any fine powder, proper ventilation helps avoid inhalation, and skin or eye contact asks for a wash-up. Not all its precursors share such a mild profile—triflic acid is a nasty corrosive and needs rigid controls. Companies producing sodium triflate abide by current Good Manufacturing Practices and safety protocols, though users still have a role. I’ve found that consistent labeling, dry storage, and care during disposal tie together safe handling with environmental responsibility.
Foundations and New Frontiers: Where Sodium Triflate Goes Next
On the applied side, this compound keeps finding homes in energy storage, fuel cells, and advanced synthetic routes for pharmaceuticals and electronic materials. Researchers target new polymers and ionic liquids using sodium triflate as both template and reactant, pushing the boundaries in battery chemistry and solid-state electrolyte design. Demand keeps pace with new technologies: the rise of electric vehicles, portable electronics, and green chemistry has all nudged more chemists to return to sodium triflate’s solid reputation. Research programs in universities and industry alike look at how it can increase catalyst life, mineralize pollutants, or even mediate unusual coupling reactions. Over years in the lab, I’ve seen skepticism about “just another salt” give way to respect as sodium triflate consistently outperforms expectations in difficult projects.
Understanding Toxicity and Environmental Fate
Unlike some fluorinated organics, sodium triflate doesn't break down to persistent or bioaccumulative substances under normal use. Most studies point to low acute toxicity for handling environments, which is reassuring for frequent users. Regulatory agencies tend to keep an eye out for buildup of any perfluorinated compounds, but sodium triflate avoids the most severe red flags. Disposal through standard chemical waste channels keeps both water and soil risk minimal. Researchers and manufacturers should keep an eye on future regulations as more attention shifts to fluorinated chemicals’ long-term fate.
Looking Ahead: Value and Innovation Linked to Sodium Triflate
Reflecting on the role sodium triflate plays in laboratories, there’s a sense that its story mirrors chemistry’s larger evolution. While flashy new reagents pop up in published papers, sodium triflate quietly supports both routine experiments and cutting-edge research. To anyone starting out in synthetic or material-focused chemistry, getting familiar with its properties and applications lays groundwork for deeper understanding later on. As greener methodologies and resource efficiency drive changes in industrial production, sodium triflate’s non-nucleophilic, stable profile promises to maintain its standard status—maybe even inspire further innovation. Students, researchers, and professionals stand to benefit from keeping this powerful chemical in regular rotation, not just for what it does, but for the trust earned through decades of problem-solving in the world’s best laboratories.
A Little Molecule with Big Impact
Sodium trifluoromethanesulfonate might sound like something only a chemist would get excited about, but its place in today’s technology-packed world deserves more attention. This compound, also known as sodium triflate, often works behind the scenes where people least expect it, showing up in industries that carry a real punch—especially energy storage and advanced chemistry labs.
Why Batteries Need Sodium Triflate
What pushes innovation isn’t always visible. Sodium triflate stands as a silent partner in modern sodium-ion batteries. The search for efficient, reliable, and secure ways to store energy never takes a break. For years, lithium-ion batteries soaked up attention, yet the need for alternatives keeps growing. Some regions can’t access enough lithium, so researchers look to sodium as a more available alternative. Sodium trifluoromethanesulfonate comes into play as an electrolyte salt. Its chemical stability and ability to easily break up in solvents means it helps deliver ions from one side of a battery to the other, making it essential for safe and long-lived performance.
When I checked out recent research, groups everywhere seemed eager to swap out the old and bring in better options. Sodium triflate avoids some of the safety issues tied to traditional battery salts, such as those that can ignite or degrade. That means engineers can focus more on design and less on disaster prevention. Watching the battery industry shift its gaze from lithium to sodium has reminded me that sometimes small tweaks in materials can shake up a whole sector.
Chemists’ Favorite Tool in the Toolbox
Batteries grab headlines, but sodium triflate pops up in all sorts of labs. Its use as a catalyst makes it a favorite for organic synthesis, helping to speed up reactions without getting used up. I’ve heard stories from friends in labs who keep a stash handy for making complex molecules, especially those used in medicine and specialized plastics. The compound’s ability to carry positive ions and stability in different conditions helps chemists develop new drugs, materials, and coatings.
I remember back in undergrad, seeing a flurry of unexpected results clear up after adding sodium triflate to a stubborn reaction. It can open new pathways, making those long nights in the lab pay off for medical discoveries or safer materials on store shelves.
Health and Safety Questions Still Remain
Widespread use doesn’t mean people should stop paying attention to health and safety. As more industries pick up sodium triflate, the responsibility grows to understand its side effects. Researchers haven’t fully mapped out its long-term environmental or health implications. Most signs point to low risk, but as stories about microplastics and persistent chemicals make the news, it makes sense to keep watching new data.
Efforts in green chemistry remind us that production and disposal both matter. If manufacturers can cut waste and recycle materials, that benefit reaches far beyond the lab. Clearer labeling, stepped-up worker training, and tighter emissions monitoring help build trust in chemical innovation.
Pushing for Smarter Chemistry
Sodium trifluoromethanesulfonate may never find the fame of gold or lithium, but it holds an outsized influence in the race for new technologies. A responsible path forward starts by connecting the dots between what goes into products and what comes out, at every layer of the supply chain. Smarter use looks at performance, price, and people’s well-being, all together.
Sodium trifluoromethanesulfonate might sound like something best left behind a chemistry lab's locked door, but you’d be surprised how often it crops up in advanced battery research and as a reagent in organic synthesis. Plenty of folks outside the chemical industry probably haven’t heard about it, yet those working with it day to day have a reason to pause and ask – are we handling something hazardous, or is it just another salt?
What We Know about Sodium Trifluoromethanesulfonate
The daily grind with chemicals, even those seen as fairly benign, has taught me to be cautious. You spot words like “trifluoro” often in fluorinated compounds and quickly learn to respect the amount of engineering that goes into making and using them safely. Sodium trifluoromethanesulfonate isn’t an exception. Its strong ability to move ions around makes it popular in the battery and electronics sector, and chemists turn to it for making tricky molecular swaps in the lab. It dissolves in everything from water to acetone, quietly getting the job done without much fuss.
Chemical handling guides don’t put it in the league of top-tier hazards. You’re unlikely to see people suiting up in full respirators unless it’s a big spill or an accident. Yet, I know that doesn’t mean you can just scoop it out barehanded. Just because a chemical doesn’t explode or stop your breathing in seconds, that doesn’t mean it’s harmless. The Material Safety Data Sheet for sodium trifluoromethanesulfonate notes irritation risk for eyes, skin, and lungs if dust clouds get out of hand. Swallowing or heavy exposure hasn’t been well-studied in humans, which points to a much bigger gap in our knowledge: labs and companies may be taking an educated guess on long-term exposure.
Known Hazards: Not Obvious, Not Ignorable
In my early lab days, I watched a colleague develop a rash only after several weeks handling “low hazard” powders. He wore gloves, didn’t cut corners, but constant exposure added up–nobody had flagged his chemical as dangerous, but that didn’t stop his skin from reminding him. The lesson stuck: irritation and chronic exposure don’t always show up on hazard labels. Science often lags a decade or more behind regular use. The Environmental Protection Agency, OSHA, and the European Chemicals Agency haven’t flagged sodium trifluoromethanesulfonate as a major toxicant or carcinogen yet, but a smart operator won’t depend solely on that kind of regulatory lag to stay safe. Dust inhalation, environmental persistence, and the downstream effects of disposal still raise questions in the absence of robust, long-term exposure studies.
Toward Sensible Solutions
Every academic and industrial lab should revisit their safety sheets every year. Don’t just skim the regulatory status. Look at real-world incidents for chemicals like sodium trifluoromethanesulfonate. If chronic exposure data is thin, lean into practical mitigation: use good local ventilation, keep containers tightly closed, and always protect skin and eyes with proper gear. Encourage reporting of minor symptoms, from skin irritation to coughing, and use those minor warning signs to rethink procedures. Choose waste disposal that ensures fluorinated compounds won’t wind up in waterways or landfills where they can persist.
Safety culture isn’t a matter of box-ticking but of respect for what we don’t know. Chemicals with few warnings today can become tomorrow’s concerns. Trusting experience and fostering open dialogue in the lab, along with strong basic protective measures for all materials, does more than any hazard label ever could to keep people safe.
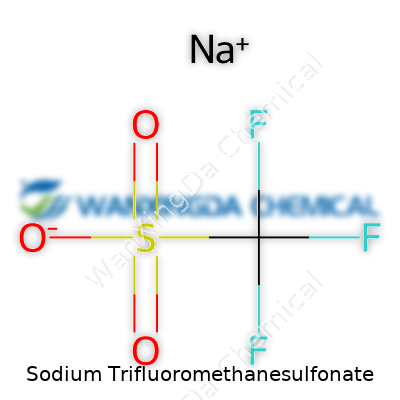
The Formula: NaCF3SO3
Sodium trifluoromethanesulfonate grabs the spotlight for anyone interested in applying advanced chemistry to real-world tasks. Its formula, NaCF3SO3, looks straightforward, but those seven atoms shape a versatile and high-value chemical. Looking at the structure, it carries a sodium ion paired with a trifluoromethanesulfonate anion, a combination that brings unique properties to the table. Knowing the formula helps uncover what it can do and why so many researchers and manufacturers give it priority.
Applications Influence Lives More Than They Appear
The power of sodium trifluoromethanesulfonate shows up outside the chemistry classroom. It’s not just textbook knowledge—anyone working with batteries, industrial synthesis, or specialty coatings bumps into this compound. It serves as an electrolyte salt in lithium-ion batteries, helping the charge-discharge cycles run smoothly and boosting stability. Better batteries in phones, laptops, and electric vehicles trace back to these kinds of molecules. People enjoying longer device lifespans or finding that their electric car runs more reliably benefit from research grounded in the correct chemical formula of additives like this one.
Beyond energy storage, the chemical also sees use in organic synthesis. As a reagent, it helps introduce triflate groups into molecules, letting chemists fine-tune pharmaceuticals, agrochemicals, or smart materials. These tweaks can mean the difference between a drug that reaches its target safely and one that doesn’t. By understanding what’s in NaCF3SO3, scientists predict behavior, control reactions, and troubleshoot when things don’t go as planned. It’s far from trivia—the formula holds the key to safer processes and better products.
Safety, Purity, and Trust
Handling chemicals demands accuracy. As someone who’s worked with a range of materials, I can say mislabeling or misunderstanding a molecule leads to trouble. The stakes get higher when compounds end up in batteries or medicines. Regular audits, rigid labeling standards, and staff who know what NaCF3SO3 stands for add layers of protection. These steps build trust in supply chains and protect workers, customers, and the environment. A single incorrect character in a formula could point to a completely different compound with a different safety profile. People rely on the right chemical identity to keep projects—and themselves—out of harm’s way.
Sustainable Chemistry Relies on Precision
It’s not only about current uses. The wider push for greener chemistry and better recycling puts even more spotlight on compounds like sodium trifluoromethanesulfonate. Efficient processes that reclaim battery materials or limit waste start with a solid grasp of what’s actually in the mix. Regulators and environmental scientists check precise formulas to set thresholds, inspect for contamination, and design cleanup measures. Getting NaCF3SO3 right means teams can innovate with confidence and respond effectively if something spills or regulations tighten.
Every element in a chemical formula plays its part far beyond the lab bench. For sodium trifluoromethanesulfonate, accuracy supports safety, innovation, and public trust. Anyone curious about how things work at the atomic level can appreciate why knowing NaCF3SO3 by heart isn’t a minor detail—it’s a building block for safer, smarter tech.
Simple Steps to Safer Chemical Storage
Working in a research lab has taught me a lot about keeping both people and supplies safe, and Sodium Trifluoromethanesulfonate provides a good example of why these routines matter. This salt pops up often as a reagent in organic synthesis, and its fluorinated nature brings benefits to reactions but makes storage more than a question of just tossing it in any old cabinet.
If you’ve ever opened a bottle of this compound, you know it’s not the sort of thing that enjoys getting wet. It clumps, cakes, and sometimes starts breaking down. Moisture in the air spells trouble for its usefulness. So, the number one priority is keeping the bottle sealed tight and far from humidity. I’ve seen too many chemicals lose their punch after sitting too long, and it’s never fun to realize a valuable reagent has turned useless mainly because the cap wasn’t closed or the container sat in a damp spot.
Control Starts With the Right Environment
I learned early on that the shelf you pick can mean the difference between good science and wasted budget. Like most powders with a bit of muscle, this one calls for a cool, dry space—think dedicated desiccator or a well-ventilated chemical cabinet, never near sources of water or fluctuating heat. In one lab, we had a shelf right above the sink, and anything left there soon took on an unfortunate texture or, worse, decomposed before its time.
It’s easy to shy away from the “extra” steps because they sound tedious in the moment. Yet every researcher and technician who cuts corners on proper storage ends up paying more for replacements. This isn’t just about finances; it’s about safety, too. Some fluorinated salts release hazardous fumes or become unpredictable when exposed to the wrong conditions. Even if Sodium Trifluoromethanesulfonate doesn’t seem volatile, storing it carelessly increases the risk of accidents.
Labeling, Tracking, and Accountability
Good habits go beyond the right closet or storage box; labeling and inventory tracking count just as much. Knowing exactly what’s inside each container and checking for degradation makes return visits to the storage shelf less like a game of chance. Once, my team lost a batch of this salt because a previous group forgot to update an inventory sheet and didn’t date the bottle. We learned from that and now use clear labels, dates, and frequent checkups. It takes effort, but I’d prefer that over scrambling in the middle of an experiment because a container turned to mush.
Common Sense Goes a Long Way
The chemistry world depends on both the science and the human practices swirling around it. Open sharing, routine safety checks, and a willingness to talk about mistakes help everyone improve the odds. People tend to look for a high-tech fix, but in my experience, solid storage—like keeping Sodium Trifluoromethanesulfonate away from moisture and heat, with readable labels—is about straightforward diligence, much like double-checking your math before publishing results.
Anyone handling specialty salts in a lab or storeroom can benefit from just a bit more attention to storage details. Controlled environments pay back by making chemicals last longer and work better. That turns into safer workdays, fewer ruined experiments, and less money wasted on spoiled stock—outcomes everyone can appreciate, on both sides of the bench.
Sodium trifluoromethanesulfonate might not sound like an everyday item, but researchers and manufacturers know it’s a critical material for a range of chemical processes. Whether you’re trying to develop better battery components, fine-tune an advanced polymer, or push pharmaceutical research forward, this compound plays its part. Still, tracking down a reliable source for sodium trifluoromethanesulfonate isn’t like picking up aspirin at the local pharmacy. The process opens up big questions about safety, regulation, and the way specialty chemicals move through global supply chains.
The Hunt for a Trusted Supplier
Those who’ve spent time in a lab recognize the frustration that comes from inconsistent supply of specialty reagents. It’s tough to maintain momentum in research when shipment delays or quality control issues slow everything down. Sodium trifluoromethanesulfonate tends to be available through large chemical distributors aiming their services toward scientists and industrial technologists. Names like Sigma-Aldrich, TCI, and Alfa Aesar pop up frequently in online searches, but not everyone has a purchasing account or the required credentials to place an order.
Access isn’t just about money or credit cards. Suppliers want buyers to prove affiliation with research institutions or companies that understand chemical safety and compliance. This gatekeeping doesn’t always sit right with new or small-scale inventors—yet there are good reasons it works this way. Specialty chemicals, including sodium trifluoromethanesulfonate, sometimes carry risks if handled poorly. Accidental misuse or improper shipping can spark dangerous situations for people and the environment. Proper documentation and traceability lower the odds of accidents and misuse.
Regulation and the Shadow Market
Rules tighten every year around specialty chemical sales. Regulatory bodies in the United States, European Union, and other regions keep a sharp eye on who buys which chemicals, especially those with dual use potential. Chemical terrorism and illicit drug manufacturing drive part of this vigilance, but environmental considerations play an important role as well. For sodium trifluoromethanesulfonate, documentation around shipping, handling, and storage requirements keeps both buyers and sellers on their toes. Sourcing from informal or “no questions asked” online sellers opens the door to impure, mislabeled, or outright fake materials, which can throw off experiments or cause real hazards.
Having spent time struggling with rogue sample suppliers, I’ve learned there’s rarely a real bargain involved. Vetted chemical companies protect your health and the integrity of your research. Documentation like a certificate of analysis, safety data sheets, and transparent customer support build confidence. That’s why so many scientists keep going back to suppliers with solid reputations, even if the price feels steep.
Improving Access Without Sacrificing Safety
The challenge isn’t just about finding sodium trifluoromethanesulfonate—it’s about finding it in a way that makes sense for safety and research integrity. Universities and research organizations can do more to support junior researchers by sharing access to procurement systems and pooling purchasing power. Industry could help by building more direct partnerships with academic labs. Contemporary regulations do serve a purpose, but policymakers should look for ways to recognize legitimate educational and small-business needs without lowering the bar for safety and compliance.
Progress thrives where safety, trust, and open access meet. As chemistry grows more complex and cross-disciplinary, finding ways to balance these needs means looking beyond just the next purchase order or finished compound. For those seeking sodium trifluoromethanesulfonate, the path forward depends on building relationships with trusted suppliers and recognizing that responsible sourcing safeguards both people and discovery itself.