Trifluoromethane Sulfonic Acid Zinc: Digging Beneath the Surface
Tracing the Road from Discovery to Modern Markets
Ask anyone who’s rolled up their sleeves in a chemistry lab—trifluoromethane sulfonic acid zinc doesn’t get the headlines, but it’s quietly threaded through significant innovations. Early work on perfluorinated sulfonic compounds dates back to the golden age of organofluorine chemistry in the later twentieth century. Scientists discovered that attaching sulfonic acid to highly electronegative trifluoromethane created a powerhouse acidic group. Chemists then found that combining this robust acid with zinc generated a compound that opened the door to both unique catalytic pathways and stable ionic complexes. Over decades, researchers experimented with preparation, improved purity, and teased apart the nuanced behavior of the zinc-triflate compound, giving industry's specialists an indispensable tool for everything from high-end organic synthesis to advanced materials science.
What Sets This Chemical Apart?
Speak to someone familiar with trifluoromethanesulfonic acid zinc and you’ll hear it’s recognized for its clarity—usually appearing as a white to off-white crystalline powder, far less intimidating than its chemical structure would suggest. Its physical sturdiness makes for easy storage, yet its chemical potency shouldn’t be underestimated. With high thermal stability and strong acidity derived from the triflate component, this compound doesn’t shy away from harsh environments. Unlike traditional zinc salts, the triflate group strips the zinc of its usual reactivity, transforming it into a much more active, finely-tunable agent for specific tasks. This makes it a go-to ingredient when you need both strength and subtle maneuvering in chemical reactions, especially where water-sensitive or non-coordinating conditions make other acid and salt forms unreliable.
Technical Specs Aren’t Just for Lab Rats
Technical labeling for this compound doesn’t exist in a vacuum—it matters for anyone who cares about reproducibility and quality in results. Commercial samples consistently boast high purity, and careful documentation addresses trace water content, which can make or break complex reactions. Chemists pay attention to melting points, moisture uptake, and precise molecular weights, since deviations spell headaches in downstream processes. Proper labeling matters, not just for regulatory compliance, but to keep workflows moving smoothly in research and manufacturing. Anyone who’s been forced to troubleshoot unexpected reaction glitches knows that detailed technical specs save hours of frustration and money.
From Raw Ingredients to Finished Compound
Manufacturing zinc triflate brings together classic synthetic rigor and contemporary tweaks driven by industry demand. The most reliable method involves reacting trifluoromethanesulfonic acid with high-purity zinc oxide or zinc carbonate. This isn’t a casual mixing job. The reaction usually runs in an inert environment to protect the product from water and minimize unwanted byproducts. Solvent choice is critical—picking between acetonitrile or dichloromethane impacts yield and safety. After the reaction, concentrated solutions undergo solvent removal, and the product dries under vacuum or in a desiccator. True to chemistry’s hands-on culture, iterative refinements in these steps have made production safer, greener, and more precise.
The Real Work: Chemical Reactions and Tweaks
Zinc triflate’s true value shows up in the reaction flask. It often acts as a Lewis acid catalyst, coaxing reluctant compounds to react, especially when working in complex organic transformations like Friedel–Crafts acylation or certain cyclization reactions. Its stability in non-aqueous media puts it ahead of traditional zinc salts in many cases. In my own years experimenting with triflates, their clear advantage has been their non-coordinating nature, which avoids unwanted side reactions that often plague less robust catalysts. Chemists also use the compound for selective deprotection steps and to generate subtle modifications on sensitive molecules. Recent years have even seen clever applications in cross-coupling reactions and as a facilitator for polymerization, showing that, while the base chemistry is well-understood, there’s room for novel exploitation.
Alias Game: More Than Just a Name
Trifluoromethanesulfonic acid zinc wears a few different hats—zinc triflate, zinc trifluoromethanesulfonate, to name a pair. This might seem trivial, but clear naming keeps accident risk down and catalog searches sane. Inconsistent synonyms have tripped up procurement and led to mismatches between SDS documents and product labeling, costing industry and academia both time and cash. Adopting consistent, globally recognized nomenclature translates to greater safety and more productive science.
Low Margin for Error: Safety and Use Standards
Laboratory veterans respect that even well-behaved chemicals have rules. Trifluoromethanesulfonic acid zinc doesn’t have a notorious profile for acute toxicity, but its strong acidity and fluorinated backbone mean it interacts seriously with moisture and organics. Adequate ventilation, personal protective equipment, and adherence to handling guidelines matter—corrosive properties and reactivity with skin or mucous membranes demand vigilance. Disposal practices also require attention. Not treating waste with the right protocol can land even seasoned professionals in regulatory hot water. Companies and university labs enforce strict training to ensure nobody mistakes the relative calm of this compound for an invitation to complacency.
Where the Work Happens: Application Areas
Industrial chemists and researchers lean on zinc triflate across a spectrum that stretches from fine chemical synthesis to electronics. In organic synthesis, it’s a powerful catalyst for reactions that shape pharmaceuticals and specialty polymers. Its role grows in the push toward greener, solvent-less technologies. Experts in materials science rely on it to tweak the structure of polymers and ionic liquids, driving advances in next-generation batteries and supercapacitors. I’ve seen it earn its keep in bench-top explorations of novel battery chemistries, where stable, strong acids are essential for tuning electrode interfaces. Its use even touches medicinal chemistry, enabling clean formation of carbon–nitrogen bonds critical for drug candidates.
A Pipeline of Progress: Research and New Directions
Zinc triflate doesn’t sit static on chemical supply shelves. Ongoing research in academic and private labs keeps stretching its capabilities. Scientists continue to refine catalyst systems, boosting efficiency while reducing the need for excess or harsh reagents. Environmental concerns push for greener, less resource-intensive preparation methods, with some promising work using milder reagents and milder solvents, or even solvent-free approaches. The interplay of zinc with organic and inorganic scaffolds unlocks new applications in nanotech, where the ability to tether metals to diverse functional groups opens fresh territory for molecular design. Publications in leading journals keep hinting at ways to fold this compound into emerging fields like synthetic biology and advanced therapeutics, indicating we’ve yet to see its limits.
Not Every Story Is Bright: Listening to Toxicity Research
The fluorinated component’s environmental resilience raises questions, especially as regulatory scrutiny of persistent organic pollutants keeps intensifying. So far, acute toxicity tests show zinc triflate to be less hazardous than many cousins, but lingering concerns about long-term accumulation and breakdown products stay on the table. Researchers dig into its biodegradability, aquatic toxicity, and pathways to mineralization out of caution, not alarm, recognizing the reputational hits suffered by related compounds. Responsible use and diligent waste management become more urgent as applications broaden, with many in the field calling for stronger interdisciplinary work linking synthetic chemistry with environmental toxicology.
Looking Forward: Navigating the Next Chapter
Zinc triflate’s profile keeps rising across high-tech sectors and green chemistry endeavors. Complex problems demand sharper tools, so stress falls on scaling up production methods while shrinking environmental impacts. New regulation may well tighten acceptable discharge levels for fluorinated compounds, nudging chemists toward cleaner synthesis or recovery methods. Industry partnerships between suppliers, end-users, and regulatory agencies could make or break the next leap in technology relying on this versatile compound. The tension between performance and sustainability will only sharpen. Recognizing this, scientists and safety officers must collaborate to close knowledge gaps and safeguard both innovation and responsibility. By not losing sight of either side—technical prowess or environmental stewardship—future progress in materials, pharmaceuticals, and catalysis remains within reach, shaped by hard-won lessons in triflate chemistry’s history.
I still remember the first time I heard about trifluoromethane sulfonic acid zinc in a graduate seminar—everyone called it “Zn(OTf)2” for short. At that point, it sounded like just another mouthful among dozens of complicated chemicals in the pharmaceutical world. Only after working in a chemistry lab, watching how synthesis projects often hit frustrating roadblocks, did I realize just how key this salt has become for scientific progress.
Driving Force in Organic Synthesis
Zn(OTf)2 pops up in labs where precise work is the currency — think about drug molecules, materials for new tech, and even agricultural chemistry. Chemists often lean on it as a Lewis acid. In regular words, that means it helps different ingredients come together in a controlled fashion. It keeps delicate reactions from going haywire, making sure results look eye-poppingly pure instead of turning into a goopy mess.
Take polymer chemistry as an example. The push for cleaner, greener plastics means scientists must shape big molecules with surgical precision. Zn(OTf)2 steps in to help stitch smaller pieces together almost like a conductor guiding the orchestra. More control means less waste — something the planet sorely needs. That's no small feat considering the amount of plastic and chemical run-off hurting our ecosystems.
A Game Changer for Pharma
Medicines demand rigorous standards, both in how researchers make them and in the purity that patients end up swallowing or injecting. Zn(OTf)2 plays a role behind the scenes in processes called selective catalysis. Building blocks of drugs, often called intermediates, get stuck or tangled during synthesis. With Zn(OTf)2 present, those building blocks smoothly rearrange or join without dangerous byproducts.
The knock-on effect? Faster lab times, safer products, and—maybe most important—affordable medicines reaching more people. Big organizations like the FDA and EMA have strict rules, so anything that helps meet these standards deserves some attention. But on the ground, I’ve seen young scientists light up when their reactions work the way textbooks promise, thanks to salts like Zn(OTf)2.
Tackling Environmental Concerns
Chemical waste has never been a popular topic around the dinner table, but it’s a reality in research and industry. Reagents that cut down on “side” reactions limit what’s sent to the hazardous waste bin. Zn(OTf)2 doesn’t solve every eco-problem, though. Its own production, and the care needed to handle strong acids, means safety gear and training matter a lot for anyone using it.
Regulatory bodies keep an eye on how substances like trifluoromethane sulfonic acid zinc travel through the supply chain. Having worked through a few audits, I know any slip can put entire projects on hold. The balance between innovation and safety becomes part of every decision in a modern lab.
Room for Growth
On the academic side, new fields like green chemistry are nudging chemists to experiment with less toxic metals or alternatives that do the same job. Some researchers look at ways to recycle spent Zn(OTf)2 instead of sending it straight for disposal—an idea worth watching as regulations tighten.
The story of trifluoromethane sulfonic acid zinc might not make headlines often, but its impact runs quiet and deep in how modern chemistry tackles some of society’s stickiest challenges—from cleaner reactions to safer drugs. Plenty of unsung heroes fill science labs around the world; this zinc salt just happens to be one of them.
Why Safety Really Matters Here
Working with high-strength chemicals like Trifluoromethane Sulfonic Acid Zinc isn’t part of most people’s day, but for folks in research and manufacturing, it’s a reality. I’ve experienced the rush of setting up a reaction with something corrosive, all senses on alert. This compound blends zinc’s usual reactivity with an acid known for being both tough and unforgiving. Anyone using it knows: any slip-up, even a simple splash, will cost more than just a stained coat. Missteps mean ruined lab work at best or serious injury at worst. Exposure risks include deep chemical burns and respiratory trouble—a trip to the ER no scientist wants to make.
The Basics That Should Never Be Ignored
There’s no shortcut to safety prep with this material. Before opening a single container, gear like splash goggles, butyl gloves, and a lab coat come out. Everyday latex just shrugs and lets the acid burn through, so going cheap or getting sloppy brings regret. Fume hoods aren’t optional—breathing in these fumes will coat the lungs with a burn you won’t soon forget. Keeping a fully charged eyewash and shower station nearby is a lesson learned from watching a fellow grad student race for the shower after a flask mishap. Speed mattered; time is tissue in these spills. Clean glassware, tight seals, and the right containers keep accidents rare but always plan for the worst.
No Substitute for Smart Storage and Transport
In the shared spaces of a busy lab, it’s easy to forget just how reactive these chemicals can be. I’ve seen colleagues store acids and bases close enough to flirt with disaster. It’s a rookie move. Trifluoromethane Sulfonic Acid Zinc doesn’t get along with moisture or organics, so it demands a cool, dry, locked spot away from anything combustible. Containers need clear labeling—surprised faces happen when an unlabeled bottle gets mistaken for something harmless. Don’t move heavy bottles alone, and never rush across crowded benches carrying corrosives. Accidents spike when pressure mounts and corners get cut.
Planning for Trouble: What Really Saves the Day
Preparation goes past gloves and goggles. At one research job, new hires ran practice spill drills—no one rolled their eyes after we watched a demonstration that turned sugar cubes and water into a sizzling caustic mess. Small spills get tackled with neutralizing agents and proper adsorbents, not whatever paper towel is within reach. The experienced folks kept their Material Safety Data Sheets nearby, but relied on their muscle memory from past run-ins to react fast. Emergency teams knew the right chemical antidotes, but nobody wanted them to need it. Real safety never feels like a checklist; it’s a culture lived out loud every day.
Teaching Respect From Day One
Students and new team members learn quickly that chemistry goes wrong fast when arrogance seeps in. It isn’t about memorizing rules; it’s about earning a healthy respect for dangerous materials. Proper training keeps rare mistakes from turning tragic. The best supervisors I worked under refused to let anyone work unsupervised until confidence matched competence. Training videos and signage set the tone, but the older hands showing what happens if you cut corners bring the lessons home.
The Path Forward for Safer Labs
Upgrading ventilation, investing in more ergonomic equipment, and creating a real culture of respect for strong acids marks the path forward. One injury from complacency ripples out through a whole lab, shaking up routines and trust. Trifluoromethane Sulfonic Acid Zinc isn’t for the faint-hearted or the careless. With the right gear, solid habits, and shared knowledge, researchers harness its power without paying the price in scars. That’s the mark of a lab that doesn’t just talk about safety; it lives it.
The Chemistry Behind the Name
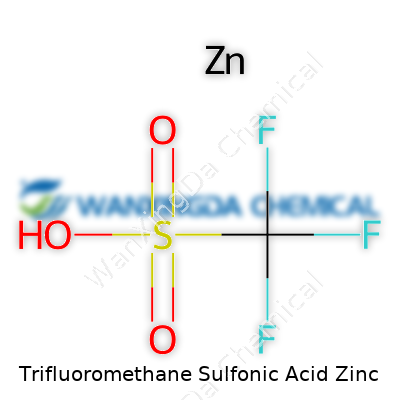
Trifluoromethane sulfonic acid, better known in labs as triflic acid, has a daunting name but an important place in advanced chemistry. When it meets zinc, things get even more interesting. The formula we’re talking about here—what folks call “Trifluoromethane Sulfonic Acid Zinc”—usually refers to a zinc salt made from triflic acid. Chemically, this compound carries the formula Zn(CF3SO3)2. That’s one zinc atom paired with two triflate ions. Anyone who has spent serious time around synthetic chemistry recognizes this mix as more than just a curiosity.
Why This Matters Beyond the Lab
Someone new to the topic might ask why a mouthful like zinc triflate deserves any attention. In the real world, most folks won’t encounter this chemical at the grocery store or on a farm. Still, this compound quietly helps in areas that ripple through industries, especially when it comes to making pharmaceuticals, running catalysts, and designing new materials for batteries or electronics. Zinc compounds like this one act as catalysts that drive otherwise sluggish or stubborn reactions. In my own experience doing work in an undergraduate chemistry lab, swapping out bulky catalyst systems for zinc triflate meant the difference between a failed experiment and a clean yield after hours of work. Industry depends on reliability, and zinc trifluoromethanesulfonate delivers in that department.
Environmental and Safety Considerations
Chemists and manufacturers can’t ignore the bigger picture. There’s real pressure now to look beyond what a compound can do inside a flask. Zinc is considered relatively benign compared to many heavy metals, but the triflate ion is another story. It’s persistent and doesn’t break down easily. Wastewater treatment plants don’t catch every molecule. Persistent fluorinated compounds, especially those with strong carbon-fluorine bonds, build up over time and may create headaches for future generations. That matters, from a scientific integrity standpoint. Researchers and regulators have started to turn a sharper eye toward per- and polyfluoroalkyl substances, or PFAS, which cover many chemicals like the triflate ion. Their nickname “forever chemicals” is no exaggeration.
Rethinking Chemistry for a Healthier Future
Solutions won’t surface overnight, but the chemistry community has learned the importance of circularity and accountability. Labs can focus on smaller-scale, highly controlled synthesis procedures—limiting the unnecessary use of long-lived ions. Industries using zinc triflate for large-scale production must develop protocols that capture, recycle, or destroy these ions before they reach waterways. Supporting research into fluorine chemistry, green chemistry alternatives, and efficient catalyst recovery practices makes a difference down the line. Funding for these efforts can change the landscape in a decade. Regulation matters too, but it works best when paired with a strong culture of safe, thoughtful lab practices.
The Road Ahead
A chemical formula might look simple—Zn(CF3SO3)2—but behind every set of symbols there’s a world of decisions shaping our future. Understanding the impact of compounds like zinc triflate moves science beyond textbooks and connects it to everyday lives. Buyers, researchers, policymakers, and students all have a stake in making decisions that lead to cleaner, safer, and more resilient chemistry.
Treating Potent Chemicals with Respect
Anyone who has worked with strong acids in the lab knows you can’t treat every bottle the same. Some go in the under-bench cupboard, others get shoved into a fume hood, and then there are those that demand a real double-take. Trifluoromethane sulfonic acid zinc falls into the group that deserves planning and a healthy degree of respect. This isn’t your everyday vinegar or lab salt. The presence of triflate and zinc brings both reactivity and potential danger, and storage plays a role in both safety and chemical stability.
Real Dangers, Not Just Theoretical Risks
No one wants to spend an afternoon dealing with a spill, much less getting caught up in corrosion issues or worse, a fire. Trifluoromethane sulfonic acid zinc is strongly acidic and it can attack certain materials many folks keep around in labs, from common metal shelving to glassware with chips. If it escapes from its container, it won’t just sit quietly. It reaches for water, eyes, skin, and soft metals. Many seasoned chemists know a shortcut stored for convenience can backfire years later.
Best Practices Come from Lessons Learned
The best storage advice often comes from researchers swapping war stories. I remember a hazardous materials training session where a chemist pulled out a bottle labelled only “TFMSA zinc.” He shrugged and said, “Kept it with the strong acids for a decade, then the cap corroded through.” That bottle’s slow leak meant hours of cleaning up a sticky, corrosive mess. A smart step involves storing this compound in airtight, corrosion-resistant containers. Polyethylene or Teflon-lined bottles can handle it. Metal lids or threads, on the other hand, don’t stand a chance in the long run. Factory-new containers aren't immune if the closure or seal gives out.
Humidity takes its toll too—chemicals like these will pull in water from the air if given a chance, which can change their properties, sometimes degrading them into a useless sludge. Dry cabinets with tight seals cut down that risk. Some facilities go a step further, placing acids like this in ventilated, fire-resistant cabinets designed for corrosive materials. These cabinets keep incompatible chemicals apart, offer a barrier in a fire, and contain spills if a bottle finally pops.
Smart Access, Smarter Separation
Mixing up storage can bring unexpected hazards. Many fires and near-misses start not with a single ingredient, but the wrong chemicals jumbled together. Grouping all strong acids in one section and keeping strong oxidizers elsewhere offers a practical layer of safety. Storing trifluoromethane sulfonic acid zinc away from bases and reactive metals is a rule that pays off. Some labs label shelves and storage bins by hazard type, giving newcomers a fighting chance to avoid mistakes.
Storage Is Everyone’s Responsibility
The real challenge isn't just choosing a shelf or a label. Lab safety builds up out of the small decisions made every day. Supervisors can set policies and buy special cabinets, but the folks doing the work have to follow through, keep their eyes open, and speak up when they spot something off. Mistakes happen when people get rushed or assume that yesterday’s setup still makes sense.
If I’ve learned anything working in shared lab spaces, it’s the importance of periodic clean-outs and inspections. Inventory checks sound boring, but forgotten chemicals turn into the worst surprises. Finding a leaking bottle before it ruptures beats reading about an accident in a safety bulletin.
Simple Steps Beat Complex Systems
Close the bottle tight. Use the right container. Never store acids like this above eye level, and don’t crowd the shelves. If a cabinet gets full, it’s time to reevaluate inventory, not start stacking. Good housekeeping is one of the quiet heroes of lab safety. Trifluoromethane sulfonic acid zinc demands respect, a dry, separate storage space, and just a little extra vigilance each time a hand reaches for the bottle.
What Purity Means in Practice
Anyone who has worked with chemicals like trifluoromethane sulfonic acid zinc knows the questions that come up before ordering: How pure is it? Which grades do labs and industry lean toward? I remember working in an academic lab, and getting stymied more than once by impurities that crept in, causing reactions to stall or send the GC traces in wild directions. Just a tiny bit of contamination in a strong acid can set off a minor catastrophe, especially when you’re counting on getting clean results.
In my experience, high purity almost always sits above 98%—sometimes labeled as “analytical grade” or “reagent grade.” Most suppliers list this right at the top of their product info. Jumping past 99%, you run into premium pricing, but also an assurance: you’re less likely to hit snags from trace metals or other unknown things mixed in. In research settings and electronics production, that extra one percent makes a real difference. For casual or educational applications, folks sometimes go with lower purity (closer to technical grade, a little below 98%), especially if the risks from impurities fall inside margin of error or tolerance. That’s not what you do when critical outcomes hang on every decimal place, though.
Why Such a Fussy Approach? Safety, Results, Reliability
One batch I worked with claimed high purity, but a cloudy solution tipped us off to leftovers from the synthesis process. Turned out the batch packed nearly 1% of an unknown byproduct, and our samples failed quality tests. That day, purity wasn’t just some bureaucratic number. It was the shield between clean, trustworthy results and a wasted week in the lab. High-grade acids like this treat surfaces, help make sensitive catalysts, and clean up electronic components. If the acid brings extra metals or moisture along, it ruins the finish or poisons the catalyst. In microelectronics, one part per million sometimes means the difference between a reliable component and a shorted pathway.
Why Certification, Audits, and Documentation Matter
Folks talk a lot about third-party testing these days, for good reason. Without independent verification, you’re taking someone’s word for it that their trifluoromethane sulfonic acid zinc actually comes as pure as the label says. Labs that care about traceability look for certificates of analysis from each batch, checked by outside labs. That matters more now that regulations for chemical handling keep tightening. Countries with strict import rules—like the United States or those in the EU—often demand proofs of purity as part of customs paperwork. Working without these puts you and your work in the crosshairs of regulators, and risks rejected shipments or failed compliance audits.
Better Purity: Not Just for Show
Purity levels speak to trust. They keep teams focused on the actual chemistry, not repeating failed reactions or solving mysterious contamination problems. Some companies step up to pharmaceutical or microelectronics grade, which can shoot the price up but safeguard entire production lines from heartache. I believe demanding a robust certificate isn’t bureaucracy run wild. It’s how you keep science and manufacturing honest and productive. Tough standards on things like water content, trace metals, or organic residue translate into solid results and fewer surprises.
Keeping the Bar High
Pushing for top-tier purity takes commitment from suppliers and buyers. Regular batch testing, clear labeling, and honest communication make it easier for everyone down the chain. Shortcuts here translate into frustration later on. Above all, asking tough questions up front means fewer spirals of blame when experiments go sideways, and that’s something any scientist or manufacturer can appreciate. The lesson stays the same, whether in a teaching lab or a semiconductor plant: don’t let purity slip, and your work stands a much better chance of holding up to scrutiny.