Trifluoromethanesulfonyl Chloride: Past, Present, and Prospects
Historical Development
Chemists started focusing on Trifluoromethanesulfonyl Chloride in the mid-twentieth century, right around the time organofluorine chemistry began turning heads for its role in synthetic innovation. Early research pushed this compound to the forefront because of its unique trifluoromethylsulfonyl group, which turned out to have exceptional reactivity when compared to typical sulfonyl counterparts. Before fluorine chemistry earned its complicated reputation for both usefulness and environmental scrutiny, Trifluoromethanesulfonyl Chloride found footing in labs that needed strong, reliable reagents. The real breakthrough came as folks noticed its ability to serve as a sulfonating agent in organic synthesis, giving rise to applications in pharmaceuticals, agrochemicals, and specialty materials. Over time, the approach to its preparation and application has grown more thoughtful, shifting from simple laboratory curiosity to a staple in both academic and industrial research routines.
Product Overview
Known to chemists for its tough, sometimes sharp odor and striking reactivity, Trifluoromethanesulfonyl Chloride (often called “triflyl chloride” or “TfCl”) stays solidly in demand. Specialists don’t overlook its hazards—it ranks among those chemicals you respect with proper gloves and goggles. Professionals count on it as a building block for transferring the trifluoromethanesulfonyl group. Its role extends across making sulfonamides, activating alcohols, and building fine chemicals that shape the backbone of modern drug development and agricultural solutions. As chemical manufacturing edges toward the future, this compound’s unique fluorinated makeup distinguishes it from more benign, traditional sulfonyl derivatives.
Physical & Chemical Properties
Trifluoromethanesulfonyl Chloride comes as a colorless to pale yellow liquid, giving off a noticeable biting smell and fuming on exposure to moist air—clear signs of its reactivity with water. Its molecular formula is CF3SO2Cl, and the weight sits at about 184.52 g/mol. Boiling point hovers low, around 32–34°C under atmospheric pressure, making it volatile even at room temperature. Swimming against the usual trend of hydrocarbon-based organics, this molecule packs a punch with its electron-withdrawing trifluoromethyl and sulfonyl groups, both of which give it sharp, often problematic reactivity with water and nucleophiles. Density checks in close to 1.6 g/cm³, so those working in the lab store it cautiously, knowing leaks evaporate and fume quickly—a trait that repeatedly complicates handling and storage. Chemical resistance offers a double-edged sword: high stability in anhydrous settings but swift, vigorous hydrolysis under humid conditions, liberating hydrogen chloride and trifluoromethanesulfonic acid.
Technical Specifications & Labeling
Strict labeling and packaging protocols guide its commercial handling, mainly because exposure risks exceed those of more mundane halogenated organics. Transportation falls under regulations for toxic and corrosive substances, both in Europe and North America. The United Nations assigns specific numbers to ensure clear risk signals right from the supply chain. Every shipment needs clear hazard pictograms—skull and crossbones for toxicity, corrosion symbols for the skin and eye danger. Any company shipping or using it closely follows these regulations to avoid accidents or regulatory action.
Preparation Method
Experienced chemists usually produce Trifluoromethanesulfonyl Chloride by reacting trifluoromethanesulfonic acid or sodium trifluoromethanesulfonate with chlorinating agents such as phosphorus pentachloride or thionyl chloride. Each route carries benefits and trade-offs: thionyl chloride often results in higher purity with fewer side reactions, while phosphorus pentachloride may appeal for cost or scale-up factors, though it brings greater side-product formation and environmental handling issues. Aqueous isolation rarely finds use, as the compound falls apart in water, so preparations happen strictly under anhydrous, inert gas conditions.
Chemical Reactions & Modifications
Trifluoromethanesulfonyl Chloride regularly stands out for its ability to act as a strong electrophile, granting it authority in creating sulfonamides and sulfonate esters. Its power to convert alcohols and amines into triflyl derivatives makes it a frequent tool in the synthesis of pharmaceuticals, protecting groups, and functional polymers. Alkylation, amidation, and esterification all work surprisingly well with this reagent, though its strength sometimes triggers unwanted side reactions, especially with less experienced hands. In my own experience synthesizing peptide derivatives, switching to triflyl chloride sped up protective group formation, but always demanded planning ahead for aggressive cleanup procedures and fast, efficient quench of residual reagent.
Synonyms & Product Names
Names shuffle depending on discipline and application. Chemists shorten it to “TfCl,” “triflyl chloride,” or occasionally “trifluoromethanesulfonyl chloride.” Catalogs sometimes call it “trifluoromethanesulfonic acid chloride,” though this can mislead those unfamiliar with its acid chloride versus sulfonic acid nature. Staying alert to naming conventions avoids confusion on the bench, especially during ordering or regulatory paperwork.
Safety & Operational Standards
Handling Trifluoromethanesulfonyl Chloride stands worlds apart from routine solvents. Even one forgotten glove or loose-fitting goggle leaves users open to corrosive burns or toxic inhalation. The chemical eats through nitrile gloves fast and demands heavy-duty alternatives. Air handling shouldn’t rely just on a sash: full containment or glovebox conditions offer real security for large-scale or high-precision work. Strict attention to ventilation and emergency neutralization remains non-negotiable. Accidents involving triflyl chloride, while uncommon, often require hospital attention due to the risk of hydrochloric acid burns and systemic toxicity. Most facilities drill spill and exposure scenarios often, because real-world mistakes unfold quickly with something as fume-prone as this compound.
Application Area
The main draw for Trifluoromethanesulfonyl Chloride lies in the breadth of its reach, straddling medicinal chemistry, electronics, crop protection, and specialty plastics. In pharmaceuticals, it props up the architecture for potent sulfonamide drugs and unique protecting groups. Crop scientists modify agrochemicals with it to bump up environmental stability or target new pest threats. The electronics industry leans on it too, since it paves the way for non-stick coatings, specialty polymers, and ionic liquids. Academic and industrial researchers trust it to introduce the triflyl group, leveraging its electron-withdrawing bite to manipulate reactivity in molecules that resist other transformations. Each of these industries rests on the predictability and punch offered by Trifluoromethanesulfonyl Chloride, though safety protocols always travel hand-in-hand with innovation here.
Research & Development
Current research looks beyond classic sulfonation, drilling deep into less established roles for the triflyl group. Cutting-edge work explores its utility in battery electrolytes, as researchers chase higher stability and conductivity through novel ionic liquids and solid-state platforms. Green chemistry researchers scrutinize both the efficiency of triflyl chloride transformations and ways to minimize environmental havoc from waste streams. Chemists target milder, more selective activation strategies to spare delicate molecules from collateral damage, and in my own recent projects, automation has started to play a larger role in precise, small-scale dose delivery, reducing operator risk and tightening up data reproducibility. This area moves fast, and you keep up by reading both the foundational literature and the newest preprints each week.
Toxicity Research
Toxicological studies don’t paint an entirely reassuring picture. Respiratory exposure triggers burning sensations and can swiftly escalate to pulmonary damage if not swiftly addressed. Contact with the skin or eyes eats away at tissue, driving companies and university labs to upgrade to more robust protection. Chronic exposure rarely sees the light of day since the hazards of acute, accidental exposure draw immediate attention and catalyze detailed internal reviews and retraining. I remember colleagues who suffered minor burns despite careful technique—the margin for error shrinks faster than with many other lab chemicals, especially in a crowded research bay. Regulatory authorities don’t let up on monitoring handling and disposal either, considering the persistent risk tied to fluorinated organics. The push for deep-dive studies into breakdown pathways and long-term ecosystem impact keeps gathering steam as society exercises more scrutiny over fluorine-based chemicals.
Future Prospects
Looking forward, two paths stand out: embracing new synthetic breakthroughs and reckoning with sustainability. More teams shift toward seeking safer, more efficient routes to triflyl derivatives—one approach fine-tunes the chlorinating reagents or swaps batch processing for flow chemistry, neatly shrinking emissions and improving yield. Green chemistry standards tighten up access and handling, which means future work will come from labs equipped with better fume handling and automated controls. At the same time, rising curiosity about fluorinated compound persistence in the environment brings Trifluoromethanesulfonyl Chloride onto regulatory radars, likely triggering more in-depth risk assessment and efforts to engineer biodegradable or less toxic functional analogs. As a working chemist, I foresee innovation springing less from sheer power and more from selectivity, safety, and environmental tune-ups. It’s an era where skill with this compound will only grow more valued, provided we keep safety, environmental, and ethical responsibilities front and center.
Trifluoromethanesulfonyl chloride, or TFMSC, doesn’t get splashy headlines outside professional chemistry circles, but it plays a bigger role in the products and medicines we use every day than most people realize. Looking back at my own work in academic labs, reactions involving this compound felt special. Not because TFMSC is particularly flashy, but because a bottle of it often sat on the shelf ready to jumpstart some pivotal transformation that simply wouldn’t work without it.
Chemistry’s Reliable Power Tool
TFMSC brings muscle to the table by transferring its trifluoromethanesulfonyl (triflyl) group to other molecules. That might sound dry, but it means TFMSC enables chemists to build structures they couldn’t build before. In practical terms, this reaction turns ordinary molecules into powerhouse intermediates. The real runway for TFMSC comes up in the creation of “triflates”—molecules that open doors to complex cross-coupling reactions. Synthetic chemistry leans heavily on those, especially when designing new pharmaceuticals, specialty polymers, or high-performance electronic materials.
When you see advances in OLED screens or next-generation batteries, the quiet work of compounds like triflates sits in the background. I’ve watched teams wrestle with finding ways to get the reactivity just right for a reaction. Sometimes, swapping in triflyl groups with TFMSC transforms the yield and purity, turning frustration into progress. That’s a real-world payoff most folks outside the lab don’t encounter when they see the latest tech gadget in their hands.
Used to Protect, Then Disappear
You’ll also find TFMSC as a protecting group reagent—chemists build complicated molecules by temporarily “hiding” reactive spots so they don’t interfere with other steps. TFMSC creates these shields swiftly, reliably, and then lets go cleanly later. This stepwise approach underpins so much of drug development and natural product synthesis. Trying to work around the selective reactivity TFMSC provides often results in dead ends or lower yields. It’s not glamorous, but it’s necessary.
Environmental and Health Concerns Need Attention
No chemical is perfect. TFMSC earns respect for its efficiency and reliability, but it comes with clear risks. It’s toxic and reacts strongly with water, releasing dangerous byproducts like hydrochloric acid. Many institutions require strict fume hood use and rigorous PPE when handling it. Safety training in our labs drilled these risks into everyone’s head, because one slip with TFMSC can mean serious trouble. On an industrial scale, responsible disposal and minimizing emissions become even more critical. There’s a broader challenge in making these powerful tools safer and more eco-friendly. Researchers, including several teams at leading universities, have been searching for alternative reagents or methods that could replicate TFMSC’s advantages without the hazards. It’s a tough nut to crack, since that combination of high reactivity and easy subsequent removal doesn’t come along every day in chemical synthesis.
Paths Toward Safer and Greener Processes
Turning the spotlight on TFMSC also means asking hard questions about green chemistry. The best minds in synthesis chemistry are pursuing catalytic or less hazardous methods that can achieve what TFMSC does—sometimes with mixed results. Strong demand for greener processes keeps that search alive. Some early-stage startup labs have reported on novel energy-efficient transformations that use milder reagents, but established approaches with TFMSC still hold on to their spot thanks to their reliability.
TFMSC may not trigger much recognition among those outside the chemical sciences. Still, its use shapes everything from pharma breakthroughs to the materials in the devices we carry. Better safety practices and greener alternatives remain priorities as the field moves forward. Like any workhorse reagent, TFMSC’s story reminds us of the complexity and responsibility behind every product we depend on.
Standing in front of a bottle labeled Trifluoromethanesulfonyl Chloride, people in the lab might think, “Wear gloves and goggles, like with any chemical.” That idea misses how quickly this stuff can cause trouble. I’ve worked enough with reactive chemicals to know: just because it sits quietly on a shelf today doesn’t mean it won’t come at you hard tomorrow. This chloride variant, with its choking fumes and tendency to eat through clothing, cuts down on casual mistakes.
A Chemical That Demands Respect
One splash, even a few drops, can chew through fabric and reach the skin. I watched a sleeve smolder after an accident, and the lesson stuck fast: always check for breaks in gloves before opening the bottle. The fumes creep out quickly. If you open a container in a small space, your eyes, nose, and lungs will remind you. Some people say if you can smell anything, you’re doing it wrong. Best practice—the kind that keeps accidents rare—is handling it only under a chemical hood, with exhaust fans running, and checking every gasket and seal.
Gloves, Goggles, and Gear—No Shortcuts
Too many folks trust a quick pair of thin nitrile gloves. Not enough protection here. You want thick, chemical-resistant gloves, a real face shield over your glasses, and a sturdy lab coat closed tight at the neck and wrists. I learned early on: short gloves leave wrists exposed, and even tiny splashes burn, scar, and force time away from work. Layering up keeps skin out of range.
Storage and Transfer—Minimizing Hazards
Storage decisions with this compound go beyond just putting it on a high shelf. Cool, ventilated rooms lower the odds of fumes building up. Once, during a heat wave, I heard about a bottle bursting when room temperature crept up past safe limits—reminders like that get around fast in the chemistry community. All lids and caps need tight seals to keep moisture out, since contact with water or even moisture in the air starts a dangerous release of hydrochloric acid. Always check bottles for cracks or leaks before moving them, and never leave containers open longer than necessary.
How Experience Teaches Caution
Reading stories from labs worldwide, real harm comes not from ignorance but from speeding up, skipping steps, or taking tools for granted. Regulations around compounds like this didn’t pop up overnight. There’s a reason professional bodies like the American Chemical Society publish strict guidelines about fume hood use and PPE. Not taking those rules seriously ends careers, sometimes lives. My own mistakes burned these steps into memory—nobody needs to get hurt if the risks stay front and center.
Learning from Others—And Asking for Help
Everyone starts out new. Even graduate students can forget a step and pay dearly for it. I always check the Safety Data Sheet, ask more experienced colleagues for tips, and never handle this alone on nights or weekends. Accidents happen fastest when we’re tired or distracted. If something goes wrong, an immediate wash with lots of water, then a scramble to the nearest emergency room, gives the best odds of recovery. No one wants to have that story, so prevention wins every time.
Improvement Comes from Honest Attention
Chemicals like Trifluoromethanesulfonyl Chloride don’t forgive lazy handling or guesswork. Investing in safer storage cabinets, fresh PPE, and better ventilation cost less than a hospital trip. Listening to the stories—the near-misses as well as the hard lessons—builds good habits and keeps both people and work on track. In the end, respect for the risks means more than a compliance checkbox. It protects lives, careers, and research all at once.
Understanding Trifluoromethanesulfonyl Chloride
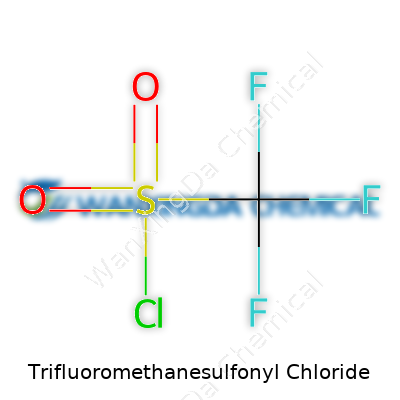
Trifluoromethanesulfonyl chloride, known among chemists as TfCl, shows up in plenty of conversations inside labs. The formula tells the story: CF3SO2Cl. This pattern packs a punch—the trifluoromethyl group connects to a sulfonyl chloride, giving both high reactivity and a certain edge that drives innovations in organic and pharmaceutical chemistry alike. If you look at the structure, it lines up like this: three fluorine atoms bonded to a carbon, all anchored to a SO2 group with an attached chlorine.
Anyone who has worked hands-on with organofluorine chemicals knows the special properties these compounds introduce. The trifluoromethyl group stands out thanks to its strong electron-withdrawing nature, which pushes reactions into new territory. In real-world applications, that boils down to more effective synthesis of pharmaceuticals and agricultural chemicals.
Significance in Chemistry
The reason folks care about trifluoromethanesulfonyl chloride goes beyond its technical formula. This compound serves as a heavy hitter for introducing the trifluoromethanesulfonyl (triflyl) group into organic molecules. As someone who has spent uncountable hours at a lab bench, I've seen chemists reach for this reagent again and again. It helps make sulfonamides and sulfonate esters—the backbone of many drug candidates and crop protection molecules.
Adding triflate groups can trick even the toughest reaction systems into working smoothly. These groups resist breaking down, making them dependable for building molecules meant to stand up to tough environments. The trick comes from the stable carbon-fluorine bond, which just refuses to budge. As a result, drug makers squeeze extra shelf-life and performance out of their products.
Hazards and Challenges
Behind every bottle of trifluoromethanesulfonyl chloride sit real safety risks. The chloride part brings strong reactivity, which can spell trouble for untrained hands. In my experience, just a splash or a whiff of its fumes reminds everyone in the lab why chemical safety matters. Robust fume hoods, gloves, and eye protection become non-negotiables. Beyond the individual’s health, accidental releases threaten both workplace safety and the environment.
Disposal brings its own headaches. A hit of moisture lets this compound break down, kicking off acidic fumes that corrode skin and lungs. Not every lab or company has the gear to neutralize waste on-site, and even in 2024, hazardous waste facilities remain out of reach for many communities. These practical hurdles often slow progress and give pause to smaller innovators who can’t offload these burdens easily.
Looking for Better Solutions
Chemists who care about both cutting-edge science and keeping people safe keep searching for alternatives or safer handling shortcuts. Education tops the list every time—hands-on safety sessions make all the difference. Manufacturers could develop stabilized versions or new packaging that cuts the risk of accidental release. On the regulatory side, clear guidelines and routine inspections ensure the rules work as intended, not just on paper.
For those designing the next generation of medicines or materials, moving toward greener reagents or recyclable by-products means fewer headaches in the long run. None of these changes feel small or quick, but the benefits ripple across the whole research and manufacturing landscape. From my own time in labs—clean, bright, and sometimes all too pungent with chemical smell—it’s clear that a steady push for smarter, safer chemistry turns out better science for everyone.
Why Careless Storage Costs More Than You Think
Trifluoromethanesulfonyl chloride doesn’t sit on a lab shelf like table salt or benign acetone. This compound reacts aggressively when moisture drifts its way, and it shows zero forgiveness for lax safety measures. I once saw a lab tech ignore a cracked bottle cap, trusting short-term luck. It ended with expensive cleanups, stressed-out staff, and a reminder that small mistakes around this reagent don’t stay small.
Understanding the Risks Before They Escalate
Anyone who’s spent time with strong chlorinated reagents knows fumes sneak up on you. Trifluoromethanesulfonyl chloride, with its volatile nature, means that even a single vapor leak can hurt lungs, eyes, and skin. News doesn’t often report minor leaks, but they add up — and occupational health statistics prove repeated exposure breeds chronic trouble.
On top of personal injury, moisture in the air triggers it to release corrosive hydrogen chloride gas. Left unchecked, this ruins valuable equipment, eats through metal fixtures, and spreads contamination through shared lab spaces. Laboratories end up pouring more into air filtration budgets and sick leave than anyone likes to admit.
Optimal Storage: Beyond Common Sense
Every chemical comes with instructions, but real-world safety depends on how tightly those are followed. Out of habit, I check seals and secondary containers every time I open the chemical storage cabinet. For trifluoromethanesulfonyl chloride, a sturdy, tightly sealed glass bottle delivers the baseline protection. Polyethylene containers break down too quickly, so glass or compatible fluoropolymer materials stick around longer.
Dryness matters more than ambient temperature swings. This compound’s temper flares when water’s near, so a desiccator or dry cabinet adds peace of mind. Room temperature storage works as long as the cabinet keeps out humidity and sunlight. Direct sunlight not only breaks down the chemical, but it raises pressure inside the bottle—risking a cracked cap or scattered fumes.
Experience taught me never to trust mere shelf labels alone. I saw new hires mistakenly place incompatible acids nearby during a rush. Acid and trifluoromethanesulfonyl chloride aren’t a duo anyone wants. Separating strong acids, bases, or reducing agents isn’t optional if you value safety. In shared labs, dedicated storage boxes with clear labeling help avoid near-misses.
Safer Habits Prevent Serious Blunders
Training takes priority. No newcomer should handle this reagent without hands-on instruction. Refresher sessions, spill drills, and clear procedures save more than rules stuck to a wall. Too often, poor habits slip in when teams get distracted, and those gaps lead to real harm.
Simple changes make a difference—vivid labels, laminated emergency instructions, and logging every access. Ventilated chemical cabinets with extraction points limit fume buildup. I’ve seen labs ignore these measures for years, then scramble after a minor leak prompts an OSHA inspection.
Moving Forward With Respect
Trifluoromethanesulfonyl chloride deserves the respect given to any unpredictable, corrosive chemical. Good storage practices aren’t just about ticking regulatory boxes—they protect people, budgets, and the integrity of hard-won research. A laboratory that values consistent, thoughtful handling stays productive and safe, setting the bar for everyone handling hazardous materials.
Everyday Chemistry and Human Health
Trifluoromethanesulfonyl chloride might not be a common household name, but it plays a starring role in labs and the chemical industry. This compound often crops up in conversations about organic synthesis, and its reputation as a useful reagent carries both respect and concern. As industries ramp up the use of complex chemicals, the conversation about health risks feels more urgent than ever.
Inhaling or coming into contact with this stuff isn’t something I'd recommend. My own stint in a synthetic chemistry lab taught me to respect any liquid with both “chloride” and “sulfonyl” in the name. Safety goggles and gloves quickly became the rule. Accidental skin contact triggers irritation. Even small spills have forced colleagues to evacuate lab spaces. The fumes sting in a way that’s hard to forget, signaling the presence of something that doesn’t belong in your lungs.
Chemicals that release hydrochloric acid on contact with water demand caution. Trifluoromethanesulfonyl chloride does just that. Irritation of the eyes, skin, and respiratory system takes only a few minutes of careless exposure. Safety data collected from multiple workplaces shows hospital visits linked to inflammation, severe coughing, or even burns. I’ve watched people underestimate even a tiny amount leaking from a flask and pay the price with days of discomfort.
Environmental Impact: Ripple Effects
Factories that use big volumes of this compound can’t ignore its environmental footprint. Spills happen. They don’t make the nightly news most of the time, but that doesn’t mean the local ecosystem shrugs it off. Reaction with water means nearby streams or drains might host a bit of hydrochloric acid if waste management slips. Acidifying a waterway threatens fish and disrupts water chemistry enough to make neighborhood biologists grumpy.
Regulations in countries with robust environmental oversight set strict rules for storing, transporting, and disposing of trifluoromethanesulfonyl chloride. International agencies classify it as hazardous. Some places even require permits for moving this compound outside the lab. These rules aren’t a bureaucratic headache—they reflect real risk. Accidental release means consequences for both people and local wildlife.
Solutions That Make a Difference
Managing the risks looks straightforward on paper. Closed reaction systems, decent ventilation, proper waste disposal: these are the basics. My old lab locked down storage to tightly controlled cabinets and always had neutralizing agents on hand. Training made the biggest difference. Knowing what to do in an emergency turned lab staff from would-be victims into the first line of defense.
Companies can turn to greener chemicals wherever possible, swapping out hazardous reagents for safer ones as technology advances. The call for more sustainable lab practices grows louder each year. Across the chemical industry, pressure builds to invest in better filtration, containment, and monitoring. Mistakes aren’t just human errors—they’re opportunities to do better.
Everyone in a workplace that sees bottles of trifluoromethanesulfonyl chloride needs to treat it with respect. Regulators nudge compliance along, but responsibility sits with every chemist, handler, and supervisor. Better safeguards mean fewer stories about accidents. The health of workers and the local environment often hangs in the balance, making careful handling more than just a box to check.