Trifluoromethanesulfonyl Fluoride: Charting Its Journey and Impact
The Road from Discovery to Utility
Trifluoromethanesulfonyl fluoride didn’t arrive overnight in the labs and factories. Scientists in the mid-20th century explored fluorinated sulfonyl compounds, searching for tough, versatile reagents. Easy to overlook compared to more common chemicals, this compound carved out a reputation for itself thanks to its robust electrophilic properties. Its rise wasn’t just about theoretical appeal—practical chemists saw real results in their flasks. Evolving syntheses and practical demonstrations pushed trifluoromethanesulfonyl fluoride into an awkward but crucial niche of organofluorine chemistry, proving that persistence in the chemical community can bring rough-around-the-edges molecules front and center.
Why This Compound Draws So Much Attention
Anyone who’s handled trifluoromethanesulfonyl fluoride knows it by its sharp, penetrating odor and its powerful reactivity. The liquid sits crystal clear with a boiling point that sits conveniently under 30°C, so it stacks up well during distillations. Chemists like it for being both reactive and surprisingly robust under a variety of bench conditions, which isn’t a combo that turns up often in strong electrophiles. Trifluoromethanesulfonyl fluoride can bring out the best in a clever synthetic route, swapping its sulfonyl fluoride group for nucleophiles like an old pro. Its stability balances just enough volatility to keep things fast without flying off the handle.
Getting into the Technical Details
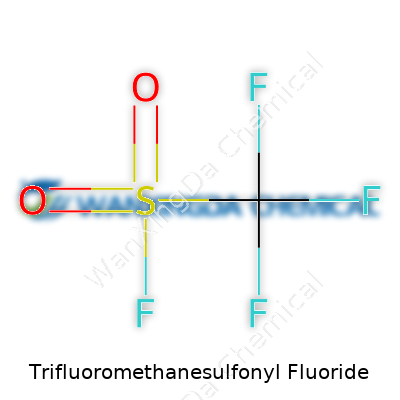
The formula, CF3SO2F, hints at its split personality—part hard-core fluorinated backbone, part unforgiving sulfonyl fluoride moiety. This composition brings about an aggressive, nearly universal capacity to transform nucleophiles, and its relatively low melting point keeps things practical for industrial processes. Physical handling means watching your fingers: trifluoromethanesulfonyl fluoride eats through moisture, reacts readily with amines and alcohols, and generates byproducts that deserve respect. Labeling often reminds handlers about these dangers, flagging both irritant and acute inhalation risks that aren’t just theoretical. Workers in industry and research labs both learn this fast, relying on good goggles and reliable glove selection to avoid trouble.
How Chemists Put It Together
Most methods start with trifluoromethanesulfonic anhydride or acid, pushing a halogenation or fluorination reaction over the edge with the right fluorine sources. Some synthetic tricks call for direct fluorination, but more accessible routes favor practical reagents like sulfuryl fluoride or antimony trifluoride. The process usually generates a foul mist and a persistent residue you don’t want in your lungs. Commercial production has dialed in those steps, balancing yield with the necessary engineering controls. Bench chemists piecing together small batches for research still find themselves stopping for a breath of fresh air and a double check on the fume hood.
Tweaking and Reacting in the Lab
Trifluoromethanesulfonyl fluoride shines brightest when asked to lay down a strong electrophilic punch. It reacts smoothly with amines and alcohols, introducing the prized triflyl group into molecules that might otherwise resist change. Syntheses of sulfonamides or sulfonate esters run easier and often cleaner, compared to older less-selective reagents. Beyond textbook group transfer, this sulfonyl fluoride lets chemists build up complex heterocycles, all the way into medicinal lead series and cutting-edge functional polymers. By itself, this molecule won’t give every answer, but in the hands of savvy researchers, it has already advanced both pure and applied science.
Known By Many Names, Used In Many Places
People in different corners of chemistry might call it triflyl fluoride, trifluoromethanesulphonyl fluoride, or just the blunt “TfF.” Whatever the name, it crops up in patent filings, research publications, and industrial brochures with equal weight. The chemistry behind the synonymy reflects the sheer spread of its application: a tool in organic synthesis for academics one day, an intermediate in specialty polymer production the next. Not many specialty chemicals straddle so many research and industrial divides with such confidence.
Staying Safe Isn’t Optional
This compound never gets handled on autopilot, whether in university departments or corporate pilot plants. Exposure spells trouble for skin, eyes, and especially lungs. Strict glove policies, full-face shields, and sealed reaction vessels add layers of protection. Safety data from peer-reviewed reports and regulatory agencies agree that trifluoromethanesulfonyl fluoride can do a lot more than sting; acute toxicity and the potential for irreversible injury put it under restricted-access status in many labs. Finished reactions need to neutralize residues quickly with aqueous bases. Facilities handling it ramp up air-handling and emergency response preparedness in ways that set it apart from routine solvents or less dynamic reagents.
Where the Work Happens
Industries dealing with electronic materials, agrochemicals, and advanced pharmaceuticals value trifluoromethanesulfonyl fluoride as both starting block and synthetic helper. The demand in recent years for “triflate” chemistry has soared, especially for building block synthesis, late-stage small molecule derivatization, and site-specific labeling of biomolecules. Its reach extends into the design of high-performance ion-exchange polymers and battery electrolytes, especially as manufacturers seek ways to combine stability with ionic conductivity. Research labs still rely heavily on this molecule for new sulfonylation strategies, pushing the edge in chemical biology and novel drug platforms. Every researcher with a toolkit for modern organic chemistry knows at least the basics of triflyl fluoride.
Pushing the Limits through R&D
Institutional skepticism about strongly electrophilic fluorinated agents persists, yet research groups keep finding new reactions that depend on precisely those aggressive features. Recent publications show this compound playing a role in cross-coupling reactions and structure-selective modifications of complex pharmaceuticals. Material science teams draw on the triflyl group’s electron-withdrawing muscle to dial in reactivity for next-gen polymers. Teams work to develop less hazardous analogs or more benign production routes, but none have yet matched the combination of utility and tunable reactivity. Graduate students and postdocs still treat every order of trifluoromethanesulfonyl fluoride like a minor rite of passage: handle with respect, exploit what makes it special, don’t turn your back.
Toxicity—Too Important to Ignore
This isn’t one of those substances you tuck in a drawer and forget about. Case studies, regulatory reviews, and decades of industrial adoption all stress careful handling. Inhalation produces intense respiratory irritation, and the liquid causes burns on direct skin contact. Animal models show organ-level toxicity at fairly low doses, making real-time monitoring and emergency response more than just academic habits. Any waste generated needs secure deactivation—hydrolysis typically does the trick, but cutting corners only invites accidents. Lab veterans watch the new students; plant managers run drills. The compound’s role in synthesis doesn’t excuse sloppy risk management.
Looking Beyond the Present
Research pushes on, anyway. Trifluoromethanesulfonyl fluoride keeps earning its place for both classic and contemporary organic synthesis; its impact on electronics and pharmaceuticals keeps it front and center in scientific planning and industrial forecasts. Drive to build greener syntheses, minimize byproducts, and devise less hazardous analogs builds every year, but no single substitute matches its versatility yet. As new frontiers open up in lithium batteries, advanced coatings, and high-value pharmaceutical actives, this molecule refuses to fade into obsolescence. Scientists and engineers keep chasing after new ways to tame its hazards while capitalizing on its singular chemoselectivity. Even after all these decades, triflyl fluoride makes a loud statement: challenge brings progress, and chemistry owes a debt to those who keep pushing the boundaries—carefully.
Every so often, a chemical compound ends up earning a seat at some impressive tables in research labs and industrial setups. Trifluoromethanesulfonyl fluoride, better known among chemists as triflyl fluoride or TFMSF, provides one good example. The name might sound intimidating, but this substance has become a bit of a workhorse across several corners of modern chemistry.
Why TFMSF Matters in Synthesis
Organic synthesis drives a huge share of today’s chemical breakthroughs. Drugs, materials for electronics, crop-protecting agents—these all depend on new molecules developed in labs. TFMSF has taken off as a key player in these efforts, mostly because it reacts with a wide range of different molecules in predictable and useful ways. Chemists love reagents that help them build complex structures without creating unnecessary byproducts. TFMSF checks this box.
Its strongest point comes down to its ability to act as a sulfonylating agent. By introducing the triflyl group into molecules, TFMSF helps chemists convert relatively simple starting materials into compounds that can do everything from interrupting disease pathways to displaying new kinds of conductivity. Sulfonyl fluorides, in particular, feature in several research fields aiming at drug discovery and biochemical probing since the triflyl group often makes molecules more stable and more selective in biological environments.
Emerging Value in Drug Discovery
Drug development grows more complex year after year. Modern medicines don’t succeed by luck; they rely on precision, selectivity, and stability. TFMSF gives medicinal chemists a way to introduce the triflyl group into candidate molecules. This small tweak can radically change how a molecule interacts with its biological target. Pfizer’s development of covalent drugs, for example, relies on sulfonyl fluoride chemistry, which borrows from the core chemistry behind TFMSF.
The reactions TFMSF makes possible often deliver cleaner results, meaning chemists spend less time separating useful material from useless side products. Cutting down on this “chemical noise” means more efficient use of resources and quicker paths to useful compounds, whether that’s in the lab or scaling up for clinical trials.
Helping Build Next-Gen Materials
Chemical manufacturing doesn’t just produce medicines. Batteries, sensors, special plastics—all of these depend on structures made for maximum stability and performance. Chemists like to use TFMSF to introduce robust triflyl groups into molecular frameworks, delivering greater resistance to breakdown and new functionalities. The electronics industry, in particular, seeks out novel materials that don’t degrade easily and can withstand heat and chemical exposure. The chemical backbone provided by TFMSF serves engineers and scientists looking for new solutions in this competitive space.
Addressing the Risks
No chemical comes without trade-offs, and TFMSF is both reactive and potentially hazardous. Its handling needs to respect health and safety protocols in laboratories and industries. Technicians learn to work with these kinds of reagents using proper ventilation, gloves, and chemical hoods. Regulators and safety officers work to make sure practices around TFMSF and its derivatives protect both workers and the broader environment.
What’s Next?
TFMSF’s impact only keeps growing. Its role as a transformative tool in both drug discovery and materials science looks stable for the foreseeable future. The push for safer practices in handling these kinds of chemicals should move hand-in-hand with the creative uses that researchers continue to uncover. With careful work, compounds like TFMSF keep opening up new possibilities while sparking vital conversations about safety and sustainability in modern chemistry.
Looking Beyond the Letters: What is Trifluoromethanesulfonyl Fluoride?
Chemistry tends to get tangled up in complicated names. Trifluoromethanesulfonyl fluoride, with the chemical formula CF3SO2F, may not ring a bell for most folks. That formula looks like a scrambled code, but there’s a story behind each letter and number. This molecule carries a trifluoromethyl group (CF3) attached to a sulfonyl fluoride group (SO2F). Picture a carbon atom linked to three fluorines, then a sulfur double-bonded to two oxygens, and joined to a fluorine. Maybe it sounds simple on paper, but packing this much chemical punch into a small structure isn’t easy.
Why Chemists Pay Attention to This Compound
Trifluoromethanesulfonyl fluoride never shows up in supermarket aisles or household cleaners. Instead, it finds its place in labs and industrial sites, where it takes on some specific roles. The compound acts as a strong electrophile, looking to react with molecules that offer up electrons. That quality makes it valuable in certain chemical syntheses, especially in the journey towards pharmaceuticals or advanced materials. Whenever chemists talk about “triflylation,” they mean reactions involving this very compound. They use it to install a triflyl group, changing how a molecule behaves or making it ready for the next step.
A Closer Look: Structure Brings Reactivity
The structure of trifluoromethanesulfonyl fluoride offers more than just a mouthful of syllables. Its arrangement puts three fluorines next to a sulfonyl fluoride group. Fluorine, as one of the most electronegative elements, likes to pull electrons towards itself. Combined with sulfur’s affinity for oxygen, the molecule creates a spot eager for reactions, especially with nucleophiles—those molecules that love donating electrons. In practice, chemists tap into this eagerness. They can transform alcohols, amines, or other groups, opening up new possibilities for chemical modification.
Importance Beyond the Laboratory Walls
Not many people see the value in a specialized compound like this, but its role stretches beyond theory. Take the pharmaceutical industry. Complex drugs often require precise chemical tweaks. The ability to add, remove, or convert certain groups on a molecule can mean the difference between success and failure in getting a new medicine to market. Trifluoromethanesulfonyl fluoride gives chemists a reliable way to introduce triflyl groups exactly where needed. This isn’t just about theory—it’s about unlocking medicines that treat real illnesses.
Safety Considerations: Not Just “Chemistry Stuff”
Practical work with trifluoromethanesulfonyl fluoride brings its own set of risks. As someone who spent time in research labs, safety training was a daily ritual. Compounds that contain fluorine or sulfur can bring toxic fumes or severe reactivity under the wrong conditions. Mistakes might cause equipment damage or require emergency medical attention. Proper protocols, good ventilation, and protective equipment matter just as much as the chemistry itself. Respect for these hazards keeps the focus on innovation rather than incident response.
Seeking Solutions and Best Practices
Every time new synthetic methods arise, scientists rethink the tools in their kit. There’s growing pressure to move towards safer, greener, and less toxic chemicals. In my own experience, alternative reagents sometimes offered milder conditions or created fewer byproducts. Nevertheless, nothing quite matches the efficiency or precision that trifluoromethanesulfonyl fluoride brings to certain reactions. The goal isn’t replacement for the sake of change—it’s about using such a compound thoughtfully while continuing to refine techniques and safety.
People throw the word "toxic" around a lot. It gets diluted from all the memes and office jokes. Step into a lab with chemicals like trifluoromethanesulfonyl fluoride, though, and the word snaps back to reality. This stuff punches above its weight — a single careless move brings real harm, not a funny story.
Real Hazards, Real People
Trifluoromethanesulfonyl fluoride, or triflyl fluoride, brings hostile chemistry to the table. Volatile, reactive, and highly corrosive, it can ruin your day, your equipment, and even your health. I’ve seen new researchers walk into the lab thinking gloves and a lab coat give superpowers. Sweat, bad eyewear, or a gash in your nitrile leaves you exposed. Triflyl fluoride especially makes this risk more than theoretical. It vaporizes easily and fume inhalation attacks nasal passages and lungs. Eyes feel like they’ve been set on fire at a whiff, and liquid contact leaves tough burns on skin, not minor irritations you can ignore.
No Shortcuts: Safety Kit Isn’t Negotiable Here
No chemical danced with as much arrogance as the person who thinks skipping face shields or double gloves saves time. Standard nitrile gloves start to degrade sooner than you think, so switch to laminated or thick butyl gloves, swapping them if contamination looms. Splash-proof goggles quickly feel essential, not just smart, after one close call. Face shields block more than drips; splashing mists drift and cross the boundary of protective eyewear, burning anyone who bets otherwise.
Personal protection only takes you so far if the workspace doesn’t help you out. A proper fume hood becomes non-negotiable. Even once, I watched a colleague rush prep using only an open bench to "save setup time." That episode ended with flaming nerves and a week of hospital visits — not from direct splash, but breathing vapors drifting across the bench.
Ventilation and Storage: Details That Matter
Strong chemical smells in the air mean trouble, not quirks of the research world. Ventilation fans and sealed hoods aren’t just for show. One missed gasket or poor exhaust design invites toxins to spread, giving folks flu-like symptoms that don’t let up. If your exhaust setup doesn’t consistently pull all fumes away, ask for fixes.
Storage in small, tightly sealed bottles and away from moisture keeps the reactive fury at bay. Water gets involved, and those exothermic reactions won’t wait for oversight — sometimes generating dangerous fumes or heat. Closets for chemicals need ventilation, so vapors don’t build up in the dark.
Smart Habits, Real Prevention
Training matters more than fancy signage. Memorizing what to do in case of a spill, instantly recognizing the fire hazards, knowing exactly where the eyewash is, and having someone nearby who follows the same safety culture — all these cut accident rates. I’ve watched panic worsen incidents when clear protocols weren’t set or when people fumbled with emergency kits. Regular walk-throughs and practice go further than occasional training lectures.
Practical Solutions: Not Just for Scientists
You don’t need a doctorate to spot arrogance in a lab. Anyone who shrugs off chemical burns or claims “nothing bad ever happens to me” risks everyone nearby. Challenge hasty habits, reinforce double-checks with safety data, and report near-misses without shame. Engineers do more than design gear; they shape behaviors.
Trifluoromethanesulfonyl fluoride can’t ever become just another routine substance. Temperament, not just technical skill, separates those who go home safely from those stuck dealing with lifelong scars. That matters more than any progress report or data point. Safety gear and honest respect get your results — and bring you home at the end of the workday.
Storing chemicals with an unpredictable temperament isn't for the faint of heart. Trifluoromethanesulfonyl fluoride, or TfMSF, stands among the chemicals that make warehouse workers sweat and researchers double-check the lock on every cabinet. I remember the first time I saw a bottle of the stuff. The safety officer pointed at my open coat and said, “One splash and the coat stays here.” That told me everything about how to treat these materials—with respect, not just rules.
Hazards That Shouldn’t Be Brushed Aside
TfMSF brings a cocktail of dangers: corrosivity, reactivity, vapor hazard. Even years in laboratories don’t dull the sharpness of its warning labels. Inhaling even a little can leave you gasping, and splashes damage more than just clothing. It’s not a “set in the corner and forget” kind of thing.
Temperature control takes priority. TfMSF gets unstable when it's too warm. Many labs and storage facilities use refrigerators for temperature-sensitive compounds, typically aiming for around 2–8°C. At my last workplace, we designated an explosion-proof fridge, hauling it in after a near miss with an ordinary unit. The smell of scorched plastic still lingers in my memory. Not every facility can afford specialized setups, but even a decent chemical fridge beats nothing.
Airtight and Dry–For Good Reason
TfMSF reacts with moisture, creating toxic fumes. If the vial cap doesn’t seal properly, the situation quickly escalates from annoying to dangerous. We always checked seals twice: a missing gasket put everyone at risk. Silica gel packets inside the storage cabinet help pull traces of water out of the air—nothing fancy, just common sense handed down from older chemists who made their share of mistakes.
Segregation matters. TfMSF shouldn’t bunk up with anything flammable or reactive, even for a short time. A cluttered shelf becomes a layer cake of risk if something leaks or tips. I once saw a storage plan that put oxidizers next to solvents, and the boss nearly blew a fuse. Label everything, keep incompatible chemicals apart.
Access Limits and Accountability
Control isn’t just about locks. Fewer people with access means fewer accidents. I’ve watched experienced volunteers take their eyes off the task for a second and regret it for weeks. Most facilities keep a logbook with signatures, dates, and reasons for every retrieval—this isn’t bureaucratic overkill; it forces everyone to slow down and pay attention.
Emergency equipment needs to stay in arm’s reach. Face shields, gloves, and eyewash stations aren’t decorative; they’re the last line of defense. We all heard stories of labs where someone skipped gloves “just this once.” The nurse on call always remembers those cases.
Seeking Solutions in Training and Design
Designing safer storage areas means more than concrete and steel. It means training people, posting reminders, and sharing stories of close calls. Regulators stress secondary containment—trays or tubs under bottles so leaks don’t travel. I’ve seen labs use repurposed baking pans and cat litter trays; it's the habit, not the brand, that saves property and lives.
It comes down to responsibility. Most accidents follow someone taking a shortcut or ignoring a small leak. TfMSF's nastiness keeps everyone on their toes. If employers cut corners, or if people don’t feel empowered to speak up, disaster finds a way in. Even one careless move becomes a warning others won’t soon forget.
A Molecule That Packs a Punch in Chemical Science
Trifluoromethanesulfonyl fluoride has a reputation that gets some people in labs pretty excited, while others stay cautious. Its structure is no secret: a trifluoromethyl group attached to a sulfonyl fluoride. This mix creates a molecule that doesn't just sit quietly—it has a strong impact, especially if you're talking synthesis or reactivity.
The Physical Side: A Faintly Pungent Gas with a Hidden Edge
Stories from the bench often highlight this chemical's gas phase at room temperature. You can't mistake its sharp, faint odor—some say it's a bit like strong acid, but it gives itself away before you even see it. Leaving it out isn't safe. Keeping this compound buttoned up in pressurized cylinders helps avoid both chemical mischief and headaches.
Temperature control plays a large part. The boiling point sits well below freezing, so even a light touch of warmth sends it drifting into the air quickly. Anyone who has handled this compound knows one slip in the cold chain leads to vapor everywhere. That’s why fume hoods aren’t optional. Every year, someone recounts a tale of a forgotten valve and a room in need of airing out.
Chemical Attitude: Reactive but Versatile
The real power of trifluoromethanesulfonyl fluoride shows up on the molecular battlefield. The sulfonyl fluoride group makes it astonishingly reactive with nucleophiles. In plain terms, it wants to shake hands with almost anyone holding an extra electron—amines, alcohols, even water. Synthetic chemists chase after it for this very talent. It offers a way to introduce the triflate group, and that change can flip the behavior of other molecules. It doesn’t just stand around; it stirs the pot in both industrial and academic settings.
It resists breaking down with most basic organic solvents, but bring it near water or a basic solution, and it goes from stable to steamed-up. I’ve seen more than one colleague joke that it’s like a dog with a favorite chew toy: give it the right target, and it just won’t let go. This means safety needs to match curiosity—nobody wants to launch an accidental hydrolysis and end up with a bigger mess.
Why Bother with Such a Feisty Compound?
The value comes from what it can add to other molecules. This isn’t just chemist-speak; pharmaceutical researchers and industrial labs both chase after reactions where trifluoromethanesulfonyl fluoride brings unique groups to life. Attaching a triflate group can crank up the reactivity, guide further transformations, or build molecules that resist breakdown—useful in drugs, protective coatings, or even electronics. The durability of the trifluoromethyl group—well-documented in scientific literature—gives rise to products that stick around far longer than many alternatives.
Handling With Respect, Not Fear
Experiences in the lab drive home the lesson that respect beats out carelessness. Protective gear, strong airflow, and serious attention keep things safe. If policymakers want to make this field safer, encouraging shared learning—incident reports, near-misses, and training—helps more than leaving folks to learn by accident. Integration of lessons learned from experienced hands keeps young scientists away from errors and improves long-term safety.
Trifluoromethanesulfonyl fluoride isn’t just another line in a catalog; it ties together potential and risk. Its story runs deeper than boiling points and reactivity charts. With proper respect, sharp minds can put this compound to good use, opening doors in science and industry that would otherwise stay closed.