(Trifluoromethyl)Benzene: One Foot in the Lab and One in the Future
History and Technical Growth
Take a close look at chemical innovation over the last century and (Trifluoromethyl)benzene tells a much deeper story than its unfamiliar name suggests. In the early days, fluorinated organics mostly lingered in academic circles. Producing trifluoromethylated aromatics like this one required clever, sometimes hazardous chemistry; the methods often started with benzotrifluoride itself or used costly reagents. Industries gave these molecules a second look once advances in synthesis brought down price and raised purity standards. Researchers embraced this compound because trifluoromethyl groups alter molecular character in major ways. The rise of specialized reagents and improved reactors changed the landscape, taking (Trifluoromethyl)benzene out of obscurity and into mainstream research and industrial use.
What Sets It Apart: Product and Properties
Benzene gets most of the headlines, but swap in a trifluoromethyl group and the story changes. (Trifluoromethyl)benzene appears as a colorless liquid, packing a sweet, aromatic odor and boiling at higher temperatures than straight benzene. This change isn’t just trivia for textbooks — the trifluoromethyl group brings major electronic withdrawal to the aromatic ring, making the molecule less reactive in some electrophilic reactions but more intriguing for medicinal chemistry and materials science. Its density climbs beyond classic benzene, owing to the heavy fluorine atoms. The physical constants — boiling point, melting point, refractive index — show that a small substitution can shift an organic molecule’s character in surprising ways.
Sourcing, Standards, Labeling
Those who work with (Trifluoromethyl)benzene learn to pay attention to purity and trace contaminants. Impurities like unreacted benzene or side-products from the synthesis can disrupt downstream chemistry. Labs and industry alike scan certificates of analysis and lot reports for assurance that what’s in the drum matches the label. Transport and labeling need a tight grip on standards, since regulators treat aromatic solvents and fluorinated chemicals with justified caution. Correct labeling isn’t red tape — it’s a hard-earned response to real health risks and historical accidents.
Synthesizing The Compound
Getting to (Trifluoromethyl)benzene at scale takes modern chemistry. Traditional methods relied on chlorination followed by fluorination, which carried risks of hazardous waste and unpredictable side-products. Modern routes often use trifluoromethylation reagents developed in the last few decades, like those based on copper, silver, or palladium catalysis. Each method comes with its trade-offs: some offer higher yields, others produce less hazardous waste, a few run cleaner under continuous-flow conditions. This evolution in synthesis mirrors shifts across the fine chemicals landscape, as researchers balance process efficiency, safety, and sustainability.
Reactivity and Modification
Put (Trifluoromethyl)benzene on the bench and its reactivity profile shows both the power and the challenge of organofluorines. The trifluoromethyl group shrinks the aromatic ring’s electron cloud, making reactions like nitration or sulfonation sluggish but enhancing stability against unwanted degradation. Chemists push this molecule further with cross-coupling and directed ortho-metalation strategies, drawing new structures where biological activity or electronic performance hinge on subtle changes. Modifications like these build the backbone for new pharmaceuticals, agrochemicals, and specialty materials.
Other Names on the Bottle
In the wild world of chemical synonyms, (Trifluoromethyl)benzene also appears as alpha,alpha,alpha-trifluorotoluene or benzotrifluoride. Textbooks, catalogs, and research papers can’t seem to agree. Anyone hunting literature or chemical inventory runs into this naming dance, a legacy of different standards and evolving nomenclature. Recognizing synonyms matters for research, procurement, and safety — the wrong name on a manifest can mean serious trouble down the line.
Handling and Safety in Practice
Working with (Trifluoromethyl)benzene calls for respect. Vapor exposure affects the central nervous system, and long-term skin contact may lead to irritation. Stuffed in poorly ventilated stockrooms or spilled on warehouse floors, the risks aren’t theoretical. Many companies updated their protocols in the wake of stricter global chemical legislation. Standard practice now means chemical-resistant gloves, goggles, exhaust hoods, spill containment plans, and engineer-inspected storage. Proper training and an honest understanding of the material’s hazards separate seasoned professionals from disaster stories in trade journals.
Real-World Applications
Trifluoromethylated aromatics carved a spot in pharmaceuticals, where the -CF3 group improves metabolic stability and shifts biological activity. Drug designers often reach for molecules like (Trifluoromethyl)benzene to build blocks with greater resistance to degradation in the body. Pesticide and herbicide developers also find value; the trifluoromethyl tag often tweaks environmental persistence and effectiveness against stubborn pests. It shows up in specialty polymers and electronic materials as well, where tuning physical properties or breakdown resistance means big money for manufacturers. This one chemical sits at the crossroads where performance, longevity, and cost all matter.
Push Toward Innovation and Safer Alternatives
Every new twist in (Trifluoromethyl)benzene research reflects broader trends in chemistry. Green chemistry has grown hard roots in production lines as companies hunt for process improvements with fewer emissions and less hazardous waste. The shift from hazardous starting materials to safer reagents marks a broad cultural change in the industry. Scientists also chase ways to make the molecule’s life-cycle less problematic; biodegradable materials, alternative solvents, and improved waste treatment all make regular appearances in development discussions.
Digging into Toxicity
Despite its benefits, the toxicological profile of (Trifluoromethyl)benzene triggers hard questions. Lab studies flagged risks for organ toxicity, especially at higher exposure levels. Regulatory agencies track any signs of chronic toxicity or environmental persistence, and rightly so: organofluorines sometimes become unexpected environmental hangers-on. Animal studies and cell assays feed into how guidelines set workplace limits or environmental discharge permissions. Transparency about exposure risks and ongoing research matter to protect not just chemists but end-users and bystanders.
Looking Forward: Challenges and Opportunities
As demand rises for elegant pharmaceutical scaffolds and harder-wearing materials, (Trifluoromethyl)benzene stands out as both a workhorse and a potential stumbling block. Pulling off safer manufacturing, clearer labeling, and robust toxicity profiling takes coordinated work across labs, regulators, and producers. Real progress means continued investment in clean synthesis methods, better green chemistry design, and technologies to track — and ultimately to reduce — environmental impact. Opportunities multiply where research groups break down the roadblocks between firmer safety practices and commercial advantage. For all its careful handling and strict monitoring, this molecule keeps disrupting old patterns, driving fresh thinking in science and industry.
Backing Up Science with Practical Needs
Chemistry often looks buried in complex formulas, but every so often a molecule steps out and makes an impact you wouldn’t expect. (Trifluoromethyl)Benzene stands out for both what it is and what it brings to the table in labs and industries. A ring of carbon paired with a trifluoromethyl group doesn’t just sound like textbook jargon. This compound has found homes in product development, high-tech synthesis, and analytical work—places where reliable performance wins out over novelty.
Where the Rubber Meets the Road in Pharmaceuticals
Most folks don’t think much about the ingredients behind their medication, but the medical field owes plenty to tricky chemistry. (Trifluoromethyl)Benzene shows up early in the game, working as a building block in drug synthesis. Adding fluorine atoms—like the ones found in this compound—to a molecule can change how the body absorbs, moves, breaks down, or even recognizes a medicine. That means stronger effects at lower doses or longer-lasting treatments without more pills. Major drugmakers have leaned on such chemistry to tweak existing drugs and shape new candidates, especially in cancer treatments and nervous system drugs.
Bringing Value to Crop Protection
Farmers deal with the relentless challenge of fighting pests and keeping yields high. Agrochemical companies eye (Trifluoromethyl)Benzene because its unique chemistry helps create stable, targeted pesticides and herbicides. The trifluoromethyl group resists breakdown from sunlight and harsh weather, which matters when thousands of acres depend on predictable results. Those improvements can cushion a farmer’s bottom line and help put food on the table worldwide.
Powering Up Materials Science and Electronics
In the hunt for cleaner tech and longer-lasting devices, engineers turn to fluorinated compounds like this one. Demand for energy-efficient displays and stronger insulation keeps rising, which pulls in the likes of (Trifluoromethyl)Benzene. Its chemical backbone makes it a reliable candidate for crafting materials with enhanced heat resistance and stability. When designers want displays that last in the sun or electronics that shrug off temperature swings, they look for solutions that start at the molecular level.
Sharper Analysis in the Lab
Reliable tools matter in research as much as in manufacturing. Analytical chemists often use (Trifluoromethyl)Benzene as a reference material or as a solvent for advanced testing methods like NMR spectroscopy. Its well-understood structure and sharp chemical signals help researchers spot impurities and measure purity, saving time and cutting down on costly errors. That’s not just value for academic labs—even industries testing for contaminants in food or water benefit from sharper, faster results.
Challenges and Smarter Paths Forward
Handling fluorinated compounds isn’t completely risk-free. The environmental persistence of some byproducts has drawn attention from regulatory agencies. So, while industries unlock more value from (Trifluoromethyl)Benzene, they also need cleaner processes and sustainable chemistry. Some groups have started using milder catalysts and recycling waste products, reducing the impact downstream. Investing in greener approaches lines up with tougher regulations and with the long-term interest of protecting soil, water, and people.
Why Care About Chemical Storage?
If someone has worked in a lab or managed a garage packed with odd substances, it’s easy to overlook why clear rules for chemical storage even exist. With (Trifluoromethyl)Benzene, the reasons start with basics—fire risk, vapor control, and human safety. This aromatic compound doesn’t play by the same rules as water or everyday solvents. It’s clear, fragrant, and flammable as a dry lawn in August. You wouldn’t stash it anywhere crowded or toss the bottle among cleaning supplies. One spark and the result gets messy, fast.
Managing Volatility and Exposure
Small labs, cramped stockrooms, and corners of workshops can’t just slap a label and call it good. (Trifluoromethyl)Benzene wants a cool, well-ventilated area, far from ignition sources. Its vapor can make short work of a storage closet without air movement, so any high shelf near heat sources spells trouble. Over the years, chemistry has taught that solvents don’t just burn—some sneak away, vaporize, and collect in hidden spots before igniting. Fire codes and insurance agents know why the rules seem strict.
Material Compatibility
Plastic jugs? Maybe not. This stuff pairs best with glass or specific metals. Once, a careless swap to an ill-suited container left an oily ring and a ruined shelf. Next time, a chemical-resistant cap did the trick. No one wants to find a mystery leak weeks later. Don’t forget clear labeling, since colorless liquids all start to look the same, especially late in the day or during a busy shift.
Protecting Workers, Not Just Product
Anyone who’s worked around organic solvents knows the sting in your throat after bad ventilation. Long exposure can quietly wear health down, so safety is about more than just following rules. Respirators, gloves, and chemical goggles are worth the small hassle. Splash just a few drops and you’ll regret not taking five minutes to gear up. Fume hoods turn an annoying task into routine due diligence.
Keeping Fire Risks Under Control
Storing flammable liquids away from sunlight, heaters, and electrical outlets becomes second nature after figuring out how unpredictable high-vapor substances can be. Static discharge sparks faster than you think. Metal shelving that’s not grounded or plastic bins that create static—both introduce surprise hazards. It’s usually the lazy shortcuts that catch up first.
What Happens Down the Line?
Waste handling matters just as much as day-to-day storage. Pouring leftovers down the drain or tossing contaminated gloves in regular trash can create headaches for the whole facility, not just the person in charge. Specialized waste containers set aside for chemicals ensure nothing unwanted seeps out where it shouldn’t. Regular staff training helps keep old habits in check and prevents mistakes that only show up weeks later.
Smart Habits Save Money and Lives
The basics never go out of style: good labeling, proper ventilation, secure closures, and respectful distance from heat. These steps protect not just people in one lab, but an entire building full of lives and investments. Statistics from occupational health groups and fire departments back up how routine safety practices pay off. It’s never about paranoia, just realistic acknowledgment that chemistry punishes carelessness. Every bottle of (Trifluoromethyl)Benzene in safe hands is one less problem waiting to happen.
Understanding Trifluoromethylbenzene
The world keeps searching for new chemicals and molecules to answer industrial needs. One of the lesser-known names is (trifluoromethyl)benzene, often appearing in labs as a clear, colorless liquid. Anyone who’s handled weird-smelling solvents in a university lab probably rubbed elbows with something similar. This molecule sits at the crossroads of organic chemistry, used as a building block in making pharmaceuticals, agrochemicals, and specialty polymers. Few notice it lurking behind the scenes, but its use keeps growing.
Human Health Hazards: What We Know
Toxicity stories with volatile organic compounds rarely paint a warm picture. Breathing in strong vapors leads to headaches, dizziness, and throat irritation. Chronic exposure pushes risks higher. While researchers have studied the toxicity of many fluorinated aromatics, specifics for (trifluoromethyl)benzene remain thin. Data from neighboring compounds steer us to think about liver and kidney stress, along with risks to neurological health.
Skin contact brings out the usual warnings: dryness, swelling, and the possibility of chemical burns if left unwashed. My own experience running reactions with similar compounds—goggles fogged, fume hood humming—left me careful about ventilation and gloves. It may seem alarmist to bundle up when spending five minutes pouring a clear liquid, but the short-term discomfort isn’t worth the risk.
Environmental Concerns: Fluorine Doesn’t Forget
Chemicals like (trifluoromethyl)benzene stick around. Fluorinated aromatic rings don’t break down easily—nature struggles to handle that tough carbon-fluorine bond. Streams and soil get exposed from spills or poor disposal habits. Over time, persistence in the environment raises red flags. Compounds with similar backbones end up in water and even accumulating in fish. Such contamination works its way up the food chain, echoing what scientists have seen with PFAS contamination in the last decade.
Long-lived fluorinated chemicals don’t just fade away. They drift in water, travel through soil, and climb into the diets of insects and small animals. Even when toxicity doesn’t stand out in the short-term, long-term presence creates concerns about chronic ecosystem stress.
The Road Ahead: Lessons From Yesterday’s Mistakes
Companies and researchers share a responsibility to minimize exposure and environmental return. Containment and thoughtful waste management should always come before efficiency. Strict separation of chemical waste means fewer surprises downstream. Labs and factories that clean up after themselves—triple-rinse, proper storage, and routine hazardous waste pickups—lower the risk of chemical drift into water systems.
Regulations still lag behind for newer compounds. That doesn’t excuse inaction. Each time a slick of unknown chemicals creeps into a river, communities pay the price. Experience with older fluorinated pollutants taught hard lessons—substitute chemicals don’t always solve underlying problems. Industry needs to test widely, report findings honestly, and keep looking for safer alternatives.
For anyone close to chemical use—whether researcher, worker, or neighbor—the best advice stays the same: avoid direct contact, push for better transparency, and expect rigorous assessment before expanding applications.
Sources of Trustworthy Information
Reliable voices come from agencies like the US Environmental Protection Agency and peer-reviewed literature. Keeping eyes open for new findings helps communities understand shifting hazards and opportunities. Science builds its reputation by self-correction, and open communication about risks protects people and places in the long run.
Understanding the Backbone
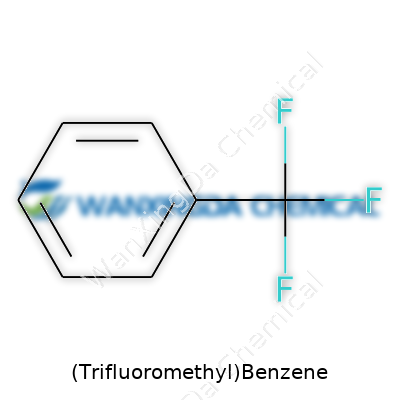
Years spent in the laboratory have shown one thing about organic chemistry: the devil hides in the details. In the case of (Trifluoromethyl)Benzene, chemists quickly realize they’re looking at a classic benzene ring that’s been given a little extra punch. The core structure remains the hexagonal ring of carbon atoms, common to all benzenes. Swapping in a trifluoromethyl group—CF₃—for one hydrogen sets up a compound with a unique fingerprint in both industry and research.
The Actual Chemistry
Chemists draw this molecule as a six-carbon aromatic ring, with a CF₃ group bonded directly to one of the carbons. Its molecular formula comes in as C₇H₅F₃. That formula packs quite a bit of meaning. In practice, the presence of those three tightly bound fluorine atoms in the CF₃ group completely changes how the molecule behaves. What seemed like a simple modification—the replacement of a single hydrogen with a powerhouse group—yields something far more intriguing.
Why It Matters in Industry and Research
Putting a trifluoromethyl group onto a benzene ring might feel subtle, but this tweak shakes up the properties of the compound in important ways. Fluorine atoms rank among the most electronegative elements. They draw electrons toward themselves, making the whole molecule far less reactive in some settings but incredibly useful in others. A chemist trying to create certain pharmaceuticals or polymers has likely run up against the quirks of trifluoromethyl groups.
Real-world pharmaceuticals routinely get more effective or stable when trifluoromethyl groups are present. Drug designers favor these compounds because the group resists metabolic breakdown, leading to better bioavailability. In the materials world, researchers insert trifluoromethyl groups to boost chemical resistance, making more robust paints, non-stick coatings, and specialty plastics.
Impact and Challenges
This molecular tweak also invites some headaches. Working with fluorinated compounds brings up environmental concerns. Fluorine-containing byproducts resist breakdown, and chemists across the globe now weigh the benefits of materials like (Trifluoromethyl)Benzene against potential environmental persistence. Disposal, containment, and greener synthesis matter more now than they did a decade ago—at least for anyone who takes sustainable chemistry seriously.
In my experience, the conversation about trifluoromethyl chemistry centers just as much on innovation as on caution. A small lab can’t easily set up all the safety and filtration needed for large-scale reactions involving fluorine, yet avoiding these key groups shuts the door to new discoveries. Balancing creativity and stewardship means more than just following regulations. It demands honest discussion about the trade-offs from university lecture halls to boardrooms.
Finding a Better Path Forward
Chemists now prioritize selective synthesis, trying to cut down on unnecessary waste at every step. Catalysts that reduce hazardous byproducts, or new routes that sidestep the toughest reagents, represent a smarter attitude. Publicly funded research helps drive these breakthroughs, but private industry has a stake, too. Companies making cutting-edge drugs or coatings have every reason to join efforts to minimize harm. The chemical community holds more responsibility than ever before.
Final Thoughts
Every structural quirk in organic chemistry brings fresh challenges and fresh opportunities. (Trifluoromethyl)Benzene's structure and formula tell a story of innovation and the burden that comes with it. Attention to safety, new greener pathways, and open sharing of techniques let chemists get the best from these molecules without sidestepping their environmental footprint.
Flip open any undergraduate chemistry textbook, and there’s a section about aromatic compounds: simple rings like benzene, jazzed up with attachments. Tweak one attachment, and you never know how it changes a molecule’s game plan. Take (trifluoromethyl)benzene, or α,α,α-trifluorotoluene—not exactly something anyone splashes on pancakes, but chemists pay attention because a trifluoromethyl group on a benzene ring brings a punch both in the lab and where medicine or material science intersect.
Tough to Boil, Easy to Spot
Growing up, I always heard "oil and water don’t mix," but that’s only half the story. Toss a trifluoromethyl group onto benzene, and you get a clear, colorless liquid that holds its ground a little differently. Compared to plain benzene, the boiling point gets a boost up to around 103°C. That’s higher than benzene’s mark at 80°C. The reason? The trifluoromethyl group throws a tiny wrench into the molecule’s snug fit, so these molecules don’t pack as efficiently. More energy—heat, in this case—is needed to push them apart and turn the liquid into vapor. This makes (trifluoromethyl)benzene especially useful in settings where benzene would literally vanish into thin air.
Doesn’t Freeze Up Easily
On winter days in the Midwest, I’ve watched puddles freeze fast. Not the case for (trifluoromethyl)benzene: its melting point hovers around -29°C. So, you can stash a bottle in a standard laboratory fridge, and it stays liquid, ready to be measured or mixed. Low melting points offer an advantage for anyone crafting reactions that run at chilly temperatures—think of chemical transformations that need to avoid any accidental crystallization.
This Molecule’s Attitude Toward Water Is Clear
If you work with fluorinated organics, you don’t expect them to chase after water. (Trifluoromethyl)benzene fits right in with that reputation; it’s practically insoluble in water. The molecule’s structure, heavy on carbon and fluorine, just doesn’t mesh with water’s desire to bond with itself. This trait isn’t a drawback; chemists often use such compounds in extractions—pulling organic molecules away from water-based solutions cleanly.
What Makes Any of This Matter?
Lab safety demands attention. Benzene’s carcinogenic risks are well documented, so substitutes show up whenever possible. The trifluoromethyl version delivers similar performance in solvent roles but still calls for care—its vapor can irritate, and it’s not friendly to inhale in closed spaces. These physical properties—how it boils, freezes, and reacts to water—help set the guardrails for good science and safe handling.
Fluorinated aromatics like (trifluoromethyl)benzene swing open many doors. Pharmaceutical chemists tune drug candidates for better metabolism or less toxicity—adding a trifluoromethyl group shakes up a drug’s profile, often making it more stable or more effective. Material scientists find value in these molecules for coatings or electronics, chasing after properties like chemical resistance or thermal stability.
Making Smart Choices In and Out of the Lab
Every property, from boiling point to water solubility, ties directly into how a molecule gets used, handled, and disposed of. Beyond textbooks, small changes in chemistry like these keep research rolling forward, often making processes safer, greener, or more efficient. For a compound that looks simple, (trifluoromethyl)benzene keeps teaching new lessons about how to think bigger by just tweaking the smallest piece.