Trifluoromethyl Trifluoromethanesulfonate: A Closer Look at Its Journey, Impact, and Future
The Story Behind Trifluoromethyl Trifluoromethanesulfonate
Science and industry never stand still, and the chemical world thrives on a steady flow of fresh compounds. Trifluoromethyl trifluoromethanesulfonate, often known as triflic anhydride or by its more technical name, is one of those specialty reagents that has kept complex chemical transformations within reach. Chemists started working with sulfonate esters as far back as the mid-20th century to boost reaction speeds and convert stubborn substrates. The addition of a trifluoromethyl group gave researchers a reagent with more muscle, especially when reactions started calling for stronger electrophilic partners. Over time, this molecule found its place among a small club of activating agents, especially in research labs and the pharmaceutical industry that demands efficiency and reliability.
Product Overview and Properties That Matter
When you see trifluoromethyl trifluoromethanesulfonate up close, you’re looking at a clear, colorless liquid that doesn’t ask for attention on the lab bench. Yet under that placid appearance, lies extreme reactivity. With a boiling point slightly above room temperature and a knack for drawing water out of the air, this molecule doesn’t wait around in open air. Its dense, almost oily feel gives a hint about how different it is from water or most organic solvents. That density means every drop packs a punch. It acts as a strong trifluoromethylating agent and a powerful electrophile in organic synthesis. What makes it sing is the highly electronegative trifluoromethyl group paired with an equally electronegative sulfonate, creating a molecule that can trigger fast, clean reactions—just what synthetic chemists need during tricky transformations.
Rough Guide to Technical Specs and Labeling
Labels for this chemical remind you right away about strict handling. Most suppliers offer it with purity above 98%, which is plenty for most research and manufacturing applications. Bottles come in sealed ampoules or tightly capped glass jars since the reagent degrades with moisture or leaks. Technical sheets warn about incompatibility with common solvents like water, alcohol, and amines due to violent reactions or decomposition. Such specs are not just numbers; they set expectations for shelf life, storage temperature, and how much risk handlers must accept.
How It's Made: Bringing the Molecule to Life
Preparation of trifluoromethyl trifluoromethanesulfonate draws from the history of synthetic chemistry, where speed, selectivity, and yield reign supreme. Manufacturers usually work with trifluoromethanesulfonic anhydride and a fluorination step, or by direct reaction of triflic acid with specialized chlorinating or fluorinating agents. The process tends to produce harsh byproducts and relies on tightly controlled environments to avoid loss and waste. Each step needs careful monitoring to keep impurities at bay because downstream uses care about every stray atom.
What It Can Do: Chemical Reactions and Modifications
Chemists trust trifluoromethyl trifluoromethanesulfonate when they need a sledgehammer to unlock stubborn bonds in organic molecules. The reagent shows up often in the installation of trifluoromethyl groups—building blocks for pharmaceuticals, agrochemicals, and materials science projects. In my own experience, there’s a sense of relief watching a late-stage functionalization that stubbornly refused every other reagent finally yield when "triflic anhydride" shows up in the round-bottom flask. Its aggressive nature means new bonds form in minutes if not seconds, often at room temperature, minimizing the need for expensive setups or drawn-out heating protocols.
Other Names and Synonyms
Depending on the lab, literature, or country, this chemical travels under several names: triflic anhydride, trifluoromethyl triflate, and more arcane acronyms that pop up in old reference books. While names might change, the challenge and excitement it brings to organic synthesis don’t.
Keeping Things Safe: Operational Standards
Safe handling of this reagent comes down to respect and habit. Anyone who has ever spilled a few drops on a glove or lab bench remembers the aggressive corrosion and fumes that follow. Safety data sheets recommend heavy nitrile or neoprene gloves, goggles, and face shields with fume extraction on at all times. Every spill needs neutralization and containment without delay. Anyone working without proper training and supervision can land in trouble. The tough safety requirements help keep research moving forward without sacrificial mistakes—the sort nobody wants to learn twice.
Where It’s Used: The Big Picture in Application
Most of the trifluoromethyl trifluoromethanesulfonate shipped today finds purpose in high-end research: medical chemistry, specialized polymers, new materials, and some battery research. Companies in the pharmaceutical industry chase efficiency and high yield, so a single molecule that shaves days off a process or turns a failed reaction into a winner is worth every penny. I’ve seen projects scramble for a bottle when everything else fell short, especially for the installation of reactive functional groups on drug candidates or diagnostic agents designed to stick out in the bloodstream. In academic circles, the compound has delivered breakthroughs in trifluoromethylation, which help usher in new drug scaffolds or probe molecules resistant to metabolic attack.
Current Research and Development Efforts
This molecule inspires research into greener synthetic methods and safer manufacturing routes. Chemical journals brim with new reactions that coax even better performance out of the trifluoromethyl sulfonate group or tweak its structure for less waste or milder conditions. Some teams focus on recycling byproducts or taming the harshness without sacrificing efficiency. The quest for “softer” electrophilic reagents keeps driving advances, with some alternatives looking to retain reactivity but drop the hazards by changing ancillary groups. So far, nothing matches this compound’s blend of speed and selectivity, but every tweak brings possibilities for broader use or less environmental impact.
Digging Into Toxicity Research
Toxicity remains a red flag for widespread adoption outside specialist circles. Short-term exposure brings acute effects—respiratory and dermal injuries happen swiftly, and medical staff remain wary about long-term risks with frequent exposure. Animal research confirms organ damage at higher doses, and accidental release causes alarm due to persistent breakdown products. Most chemical plants limit access and keep only small volumes on hand to reduce incident risk. Regulations in many countries now require reporting and monitoring, which creates pressure for safer handling and clear public communication about risks.
What’s Over the Horizon: Future Prospects
Future growth for trifluoromethyl trifluoromethanesulfonate rests in chemistry’s eternal struggle for safe, sustainable innovation. The molecule’s future hinges on how industries balance risk and reward. As medicine leans on more fluorinated drugs for hard-to-treat diseases and as electronics look for ever more stable materials, demand for reactive fluorinated reagents stays high. Improved handling equipment and better training help manage some hazards, but the next leap may come from designing similar molecules that soften the negatives. Green chemistry and process intensification offer hope that manufacturers can make and use this chemical with less waste and energy. Researchers worldwide keep looking for ways to stretch its potential without stretching safety thin.
There’s a certain thrill in the world of chemistry, especially when a substance like trifluoromethyl trifluoromethanesulfonate shows up in the story. This tongue-twister of a chemical, often called the “triflate,” comes up when reactions need a push that the usual suspects just can’t give. It's not a household name, and that's probably for the best—this stuff asks for respect in the lab. The real question is: why does this compound matter, and how does it shape the work that goes on behind closed chemistry doors?
A Demand for Strong Tools in Modern Chemistry
Chemistry likes to throw curveballs, especially in pharmaceuticals and materials science. Scientists often want to add the trifluoromethyl group—a cluster with a punch, famous for making molecules behave in new ways, like cranking up their stability or letting medicines resist breakdown in the body. Trifluoromethyl trifluoromethanesulfonate shines here, acting as a no-nonsense source of trifluoromethyl groups when others just fall short or make too much of a mess. The power of this compound comes from how eager it is to jump into reactions—chemists call it a strong electrophile, which just means it’s ready to grab onto other molecules fast.
Big pharma companies chase these trifluoromethyl groups for a reason. More and more drugs benefit from this chemical badge, helping pills survive stomach acid or slip through biological barriers. Fluorine atoms, which this compound brings in spades, make a real difference in how active compounds behave. Data from the last decade shows an uptick in new medicines landing on the market with these groups. These aren’t just numbers in a brochure—they point to more treatment options and, sometimes, drugs that actually stick around long enough in the body to help.
Organic Synthesis Gets a Lift
In research labs, the triflate turns up as a workhorse. It brings an edge when chemists want to build up exotic molecules, especially when other pathways get bogged down by too many steps or old-school reagents. With its solid ability to transfer the trifluoromethyl group—without fuss—it saves time and cleanup. For chemists like me who have spent afternoons skimming off failed reactions, that means less frustration and more success stories. The satisfaction comes from finding a route that works, not just going in circles with low yields.
Price, Hazards, and Responsible Use
Here’s the hitch: trifluoromethyl trifluoromethanesulfonate doesn’t hand out favors for free. It’s expensive, and it demands attention to safety. This is a compound that isn’t friendly with moisture, and it has a tendency to react so vigorously that a distracted moment can spell disaster. Big labs with solid ventilation and strict protocols can handle these risks. Smaller operations have to weigh whether the benefits justify the expense and the hassle of special handling routines.
We can’t dodge this conversation—the push for greener chemistry calls for real answers about alternatives. Are safer or cheaper reagents catching up? Some new research does point to milder methods and options that rely less on such aggressive chemistry, but for now, triflate keeps its spot in the toolbox for high-stakes synthesis.
Looking Ahead: Chemistry’s Balancing Act
Trifluoromethyl trifluoromethanesulfonate has carved out its place because tough challenges need bold answers. The fact that it powers life-changing pharmaceuticals speaks volumes. Yet, the chemical community owes it to itself to keep searching for safer and more sustainable routes. Until those arrive, we work with respect for what this reagent offers—and with a healthy dose of caution.
Why Respect Science’s Sharper Tools
Science moves ahead using some pretty nasty stuff, and trifluoromethyl trifluoromethanesulfonate sits among the rougher chemicals you’ll meet in the research world. Many folks outside a lab have probably never heard the name, but those who work with it don’t soon forget what it can do. This compound isn’t out for mischief—it’s a necessary reagent in fields hunting for groundbreaking pharmaceutical candidates and advanced materials. The stakes get raised every time someone opens a bottle, since the hazards go way beyond a little skin irritation.
Risk Comes from Real Experience, Not Headlines
Try spilling a drop of this stuff and you'll see how “strong electrophile” translates to eye-watering fumes and a desperate rush to the safety shower. In my years around chemical fume hoods, everyone remembers the time someone reached for gloves and picked the wrong kind. A nitrile slick failed, and that tingling soon turned into pain. Tales like that become training gold, and they underscore why shortcutting protocols belongs nowhere near this chemical.
Concrete Steps — Nothing Fancy, Just Careful
Start with the right gloves: not all lab gloves will shield you. The fluoroelastomer or heavy-duty butyl gives far better protection than the basic disposable kind. Splash goggles prove more than a rule; they block out splatters that attack eyes on contact. Handling happens inside a certified fume hood—desk work or open lab benches invite disaster. The fumes will sting, and even diluted vapor spells trouble.
Breathe freely by checking the airflow. Those in charge of ventilation schedules earn gratitude, not complaints, since an uncompromising fume hood cuts the risk down sharply. The temptation to prop open the sash or work at the edge for “just a moment” never pays off. I’ve learned that tiredness or haste only wins you an extra hazard.
Preparation Trumps Panic
Before work begins, bottles of neutralizing agents and spill kits sit nearby. Relying on memory to track what’s in the kit doesn’t cut it—open it, check, reset as needed. Peer reviews of your prep work aren’t bureaucracy; they’re guardrails. Keeping the work area clean and well-organized keeps elbows, sleeves, and glassware from knocking over what should stay upright.
People rarely talk about practice drills, but running one helps everyone recall what action to take if a spill hits the floor. It’s about knowing routes to the eyewash and safety shower while you can still think straight. This goes double for newcomers, who might freeze up otherwise.
A Culture of Respect Beats Expert Status Every Day
Somewhere along the way, pride can block good habits. I’ve seen seasoned chemists shrug off protective gear, thinking experience grants immunity. No resume shields skin from a corrosive burn. Real mastery means knowing when to say, “Let’s double-check the safety steps.”
Sharing near-miss stories, updating training with each batch of new arrivals, and refusing to cut corners send a message: people matter more than production speed. The best labs build loyalty by putting safety at the front, using both common sense and the lessons passed down from those who learned the painful way.
I remember the early days working in a university lab, people respected chemicals but sometimes underestimated the quirks of the more reactive ones. Trifluoromethyl trifluoromethanesulfonate—a mouthful of a name and even trickier to handle—belongs in that category. Careless storage can quickly turn a promising experiment into an emergency. So why does this particular compound deserve careful attention?
The Chemistry Running the Show
This isn’t your run-of-the-mill solvent or buffer. Trifluoromethyl trifluoromethanesulfonate reacts fast, especially if water or strong bases sneak into the container. Moisture isn’t just a nuisance; it can trigger unpredictable reactions, which brings real safety risks. Once, I saw an improperly capped vial fizz because someone stashed it in a humid cabinet. Nobody wants to mop up that sort of mess.
What’s happening here is simple: strong electrophiles don’t play well with water or air. Exposing them invites breakdown or, worse, dangerous byproducts. Damage doesn’t stop with wasted materials—people can get hurt. That should push anyone to think before tossing the bottle onto an ordinary shelf.
Hands-On Storage: Not a Job for Guesswork
Dry, airtight, cool. Those three rules saved more than one project during my stints in both research and small industry labs. A desiccator—ideally one containing fresh silica gel or similar drying agents—provides a strong line of defense against stray moisture. I’ve trusted glass containers with screw-top caps and extra parafilm wrap as back-up. Leaving triflate reagents in plastic risks slow leaching or breakage. The difference isn’t just academic. Longevity drops when light or air gets into the mix.
Temperature plays its own role. Most researchers and manufacturers hold to fridge or cold-room conditions, well below room temperature. Besides slowing reactions, the cold keeps vapor pressure down. This means less chance for vapors to escape and cause breathing hazards or contamination nearby. I always labeled bottles clearly—confusion about contents has no place here. Storage outside the lab fridge led to mixed-up bottles more than once, and that’s never safe.
Human Factor: One Slip Changes Everything
I’ve seen labs pay dearly for shortcuts. Trifluoromethyl trifluoromethanesulfonate offers zero forgiveness if handled lazily. Anyone working with it should wear gloves, goggles, and a lab coat. One splash or whiff lingers far longer than a simple risk assessment might suggest. Just last year, a colleague slipped and bumped into a poorly stored vial; they spent hours sorting out exposure protocols and decontamination. Lesson learned: store the chemical right, or brace for the fallout.
Building Good Habits and Systems
The best solutions go beyond checking boxes on a safety form. Strong lab culture means every user understands why airtight glass storage, low temperatures, and dry conditions matter. Regular checks on seals and desiccant levels prevent surprises. Smart labs post clear signage, offer routine refresher training, and set up inventory logs for high-risk reagents. Involving everyone, not just lab managers, helps keep vigilance high. Modern digital inventory software can send reminders to check containers or replace desiccant, a tool I wish we’d had years ago.
Trifluoromethyl trifluoromethanesulfonate isn’t evil, but it demands respect. The rules protect not just the bottle, but every person who comes near it. Every person with hands-on experience knows: quality storage separates a productive day from something far messier—and sometimes much, much worse.
Diving Into the Structure: Science Meets Sophistication
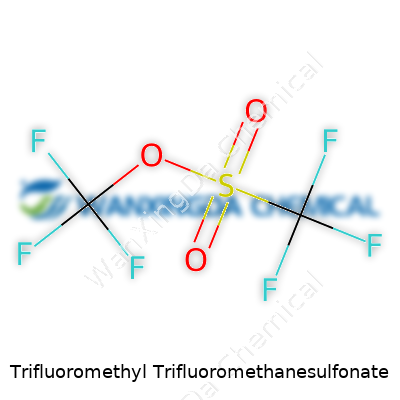
Trifluoromethyl trifluoromethanesulfonate, more commonly known among chemists as the “triflate” ester with a mouthful of a name, captures the imagination for a reason. Its chemical formula is CF3SO2OCF3. This isn’t just an arrangement of letters and numbers—it speaks to the way fluorine atoms dominate the molecule. Each of the trifluoromethyl (CF3) groups brings a unique blend of reactivity and stability that comes in handy during complex molecule construction. It’s one of those compounds you hear mentioned in labs, always lurking in discussions about creating elegant shortcuts in organic synthesis.
The Power Lies in Its Reactivity—and Unforgiving Nature
What I find striking about trifluoromethyl trifluoromethanesulfonate is how it balances the scales between being a gentle helper and a demanding partner. It acts as a powerful electrophilic trifluoromethylating agent, meaning it drops that electronegative CF3 group into other molecular frameworks. The cooled-down excitement among researchers isn’t just for show; using this stuff takes craft. Touch a little water or basic conditions, and you’re liable to lose all the expensive reagent in a blink.
Calling the Shots in Drug Design
Drug discovery can feel like searching for a singular tree in a forest full of lookalikes. Trifluoromethyl groups can tip the balance by tweaking natural properties—solubility, metabolic resistance, and, more often than not, biological activity. I’ve watched medicinal chemists stack these CF3 groups on molecules, betting they’ll stick around longer in the body or sneak a drug past the liver’s defenses. Trifluoromethyl trifluoromethanesulfonate moves this dream along by opening more efficient routes to those prized structures.
Safety Behind the Bench: Not Just for Show
This reagent doesn’t hand out second chances. Breathing vapor or skin contact can mean serious trouble, and it reacts with water even in small traces. Most of the chemists I’ve known insist on working with it inside a glovebox or fume hood. Proper training looks less like bureaucracy and more like keeping your health and research on track. As someone who’s seen close calls, I can’t stress enough the respect this compound demands.
Environmental and Ethical Reflections
The world has developed sharper eyes for sustainability and safety. While trifluoromethyl trifluoromethanesulfonate unlocks possibilities, it also highlights chemistry's crossroads: progress meets responsibility. Fluorinated compounds tend to hang around in the environment, and that sparks questions. Just because we can use a chemical doesn’t always mean we should do it without thinking ahead. Green chemistry efforts push for safer alternatives or improved processes to minimize waste and exposure. Laboratories everywhere are rethinking procedures—not simply out of regulation, but out of respect for both researchers and the wider community.
The Way Forward: Smarter Solutions, Not Just New Reagents
Instead of doubling down on brute-force chemical tools, the best labs now aim for creativity. That might mean exploring catalytic amounts, recycling methods, or shifting to other trifluoromethylating agents with safer profiles. Sharing results and honest feedback helps, since not every solution needs to reinvent the wheel. Success isn’t just about achieving new molecular bonds; it’s about doing so with craft, care, and an eye on the bigger picture. In my experience, it’s this blend of curiosity and caution that moves science forward.
Why Purity Always Matters
Every synthetic chemist knows the value of a clean reagent. Trifluoromethyl trifluoromethanesulfonate—often called triflic anhydride—isn’t much different. High purity carries more weight than just a fancy label; it draws the line between successful reactions and wasted materials. Impurities in a reagent don’t just dilute the outcome—they can crank out misfires and side-products that muddy up downstream efforts.
At a time when many labs watch budgets as closely as yields, chasing after the purest form can seem like fussing over a detail. The truth pops up during workup and analysis: cutting costs on purity invites headaches. For instance, even a tenth of a percent of moisture or chloride contamination triggers side-reactions that throw off selectivity, reduce product stability, or kill yields outright. In some runs, that means starting over. Sometimes, the hidden costs creep into the next stage when trouble crops up with spectral analysis or purification.
The Numbers Behind Specification
It usually makes sense to look for product above 99% purity. This keeps unwanted byproducts low and gives consistent results batch to batch. It's not paranoia; it’s just practical, especially while pushing for high-value or regulated end products. Spectral analysis—NMR or GC/MS—backs up those purity claims, so anyone shopping for a batch can double-check what they’re about to use.
In the usual certificate, water content runs under 0.05%. Acidic impurities, which kickstart decomposition, tend to be held below 0.01%. If that sounds like splitting hairs, consider what even tiny acidic residues do to a moisture-sensitive sulfur trioxide group. Extra acid turns a well-behaved reagent into a problematic one, particularly in scale-up scenarios.
Density (1.6 – 1.7 g/cm³ at 20°C) and a clear, colorless liquid appearance show up in the standard checks. A yellow tinge, or a drop-off in measured density, should serve as red flags. Some suppliers even add further breakdowns—conductivity, specific isomer content, or chloride traces. Much of this gets lost in cheaper products, which show up in organofluorine reaction mishaps or oddball TLC spots.
Stability, Handling, and Real-World Lessons
Stable storage changes everything. I’ve seen what happens when reagent bottles fight humidity—results get unpredictable fast. These compounds react quickly with traces of water, releasing acid and throwing off stoichiometry. Dry, cool, and inert gas-purged storage isn’t a laboratory luxury, but a baseline requirement. Some products even come sealed under argon just for this reason. Once broken, plan to use it soon or risk losing both purity and cash.
Moving Toward Cleaner Reagents
There’s a place for less demanding grades, but not in precision work. Drug manufacturers and electronics development lean on the highest spec versions—mishaps get expensive there. Routine organofluorine chemistry can use a budget batch, but only with thorough pre-checks and purification. Quality assurance isn’t just for bean counters—it helps young and old hands avoid late-night troubleshooting and wasted prep time.
Labs have nudged suppliers for better testing, tighter specs, and transparency. Good suppliers routinely publish full spectra and impurity profiles. The best even flag lot-to-lot consistency. End users can help push the industry in the right direction—request data, share experiences, and return poor quality shipments. No one likes paying more, but in sensitive chemistry, purity isn’t optional. It’s a basic ingredient in every good result.