Trifluoropropionic Acid: Understanding an Unsung Workhorse in Modern Chemistry
Historical Development
Trifluoropropionic acid doesn’t headline the periodic table, but anywhere that chemistry dives into modern challenges, you’ll find this molecule isn’t far behind. Through the decades, researchers learned that swapping hydrogen atoms for fluorine often changes everything about a compound. Back in the 1950s and 60s, as the chemical industry branched out, folks got hungry for chemicals that brought stability and resistance, particularly for pharmaceuticals and agriculture. Chemists found themselves drawn to the unique features of fluoroalkyl acids. Trifluoropropionic acid showed up in academic papers and research notes as part of a family of perfluorinated compounds that shifted whole chemical landscapes. Scientists didn’t stumble into this molecule by chance; rather, through years of tinkering with organofluorine compounds, the utility of trifluoropropionic acid became clearer. Its development followed the path of advancing synthetic methods, aiming to harness the special qualities provided by those three tightly bound fluorines.
Product Overview
You won’t spot trifluoropropionic acid on grocery store labels, but anyone working in synthesis labs has crossed paths with it. This small molecule carries a character that is both aggressive and strangely reliable. In labs that focus on adding function to molecules or searching for new materials, trifluoropropionic acid sits at a sweet spot: unique enough to transform target compounds but not so exotic that it’s out of reach. Its structure—a three-carbon chain capped with a carboxyl group, each carbon draped with a trifluoromethyl group—makes it far from ordinary. People come to it for its balance. Long enough to give useful properties, compact enough for manageable handling.
Physical and Chemical Properties
Pouring out as a colorless liquid, trifluoropropionic acid has a sharp, biting odor. That’s the fluorine working overtime, giving it a volatility that can’t be ignored in any working space. The boiling point, ranging around 165°C, proves this isn’t your standard organic acid; fluorine atoms tug down volatility and kick up acidity. Its solubility blends well with organic solvents. Acidity stands tall, as you’d expect from a perfluorinated carboxylic acid, and that means strong reactions with bases and metals. The trifluoromethyl groups resist standard degradation, locking in stability even under tough conditions. Anyone who’s handled it knows gloves and eye protection are more than an afterthought.
Technical Specifications and Labeling
Bottles in the lab come marked with hazard symbols for corrosivity and health risks. The IUPAC name—2,2,3-trifluoropropanoic acid—describes every atom’s position, but in practice, people shorten it for convenience. It’s usually offered at purities above 98%, enough for demanding syntheses. Labels also draw attention to handling concerns, keeping it well away from moisture and strong oxidants. In fact, chemical suppliers lay out lots of caution around storage, hinting at both reactivity and the need for good housekeeping. Nobody should let trifluoropropionic acid linger uncontained.
Preparation Method
Making trifluoropropionic acid takes precision. The common routes begin with halogen-exchange reactions, where other perfluorinated species are coaxed into new arrangements through catalysts or specialized equipment. Early approaches drew from hydrolysis of corresponding nitriles or esters (think trifluoropropionitrile, for instance), or from fluorination of precursors using elemental fluorine or more accessible reagents. The process isn’t exactly low-tech. Each step needs to wrangle hazardous reagents, control temperatures, and, above all, prevent runaway side reactions. My own experiences watching skilled synthetic chemists tackle these transformations reveal layers of practice, from monitoring NMR spectra to double-checking every flask for leaks.
Chemical Reactions and Modifications
What sets trifluoropropionic acid apart is how it plays with other molecules. The fluorinated backbone gives it a unique blend of reactivity and stubbornness. Researchers often use it as a starting acid in acylation reactions, introducing the trifluoropropionyl group into more complex structures. The strength of its carbon-fluorine bonds means it resists breakdown, turning up in derivatives meant to survive heat or aggressive pH. Sometimes, chemists modify the acid through esterification, amidation, or reduction, unlocking new materials for analysis or drug discovery. It can form salts that remain stable where more typical carboxylates would stumble. In my time reviewing recent literature, I’ve seen trifluoropropionic acid enable construction of intricate pharmaceuticals and advanced polymers, each project gambling on its quirks and strengths.
Synonyms and Product Names
In research circles, trifluoropropionic acid travels under many aliases. You’ll run into it as 2,2,3-trifluoropropanoic acid, trifluoropropanoic acid, and sometimes as TFP acid in shorthand notes. Older texts might mention trifluorinated propionic acid without specifying exact locations for the fluorines—proving the importance of clear labeling. Sometimes, catalogues reference CAS numbers to avoid confusion, a habit worth keeping for anyone moving between suppliers or regulatory filings.
Safety and Operational Standards
Anyone working with trifluoropropionic acid needs sharp instincts about chemical safety. This isn’t a molecule that forgives sloppy habits. It eats at metals, irritates skin and lungs, and likes to escape into the air. Labs invest heavily in fume hoods, chemical-resistant gloves, and plenty of ventilation. Training focuses on recognizing exposure symptoms—severe discomfort, sometimes persistent skin reddening, and respiratory problems if inhaled. Discussions around this acid stretch beyond routine protocols, moving into strategies for safe disposal and containment. Accidental leaks demand immediate cleanup; environmental agencies watch for releases because of worries about bioaccumulation and long-term persistence. Regular audits force labs to revisit their storage and labeling practices, always chasing tighter standards and safer handling.
Application Area
Despite its dangers, trifluoropropionic acid powers a wide swath of science and industry. Its fingerprints show up in drug design. Medicinal chemists use its acid group to add bioavailability and improve the metabolic profiles of their lead compounds. Agriculture researchers tinker with it to improve pesticide resistance. The electronics field, desperate for stability in harsh conditions, looks at fluoroalkyl derivatives to insulate materials or tweak surfactant properties. Environmental labs analyze water and soil for traces, tracking movement of fluorinated pollutants. My interaction with environmental scientists taught me these molecules can persist, prompting regulatory interest reminiscent of broader PFAS debates in recent years. This dual edge—functionality and environmental impact—mirrors the ongoing struggle between innovation and responsibility in the chemical world.
Research and Development
Chemistry keeps evolving, and so does our relationship with trifluoropropionic acid. Today’s research teams stretch its applications into even narrower targets, like specialty coatings, high-performance adhesives, and advanced liquid crystals. Pharmaceutical scientists keep pushing for new prodrugs and targeted therapies that leverage fluorine’s influence on absorption and metabolic stability. Green chemistry initiatives work on alternative synthesis routes, hoping to leave behind fewer byproducts or lower the risks of environmental release. Working with a multidisciplinary team in a university setting, I saw firsthand the push to design new catalysts for trifluoropropionic acid formation, aiming for more efficiency and lower waste. Every bit of progress here widens our understanding, yet also underscores how much more we still face on the toxicity and disposal fronts.
Toxicity Research
There’s no way around the fact that trifluoropropionic acid brings hazards that demand respect. Studies have shown that acute exposure harms the digestive system, the respiratory tract, and the skin. Chronic exposure raises eyebrows among toxicologists, especially with links to organ damage at certain doses in animal models. Unlike earlier chemical hazards, we’re only beginning to understand how persistent and mobile fluoroalkyl acids can be in the environment. My own background in environmental monitoring impressed on me just how challenging it is to track low-level, long-term accumulation. The current literature keeps looking for links to metabolic disruptions, serving as a warning sign for both regulatory authorities and lab managers.
Future Prospects
It’s not enough to say trifluoropropionic acid will stick around; its future pivots on how we balance feasibility and risk in chemical manufacturing. Demand from the pharmaceutical and specialty materials industries isn’t likely to fade. Yet environmental standards tighten with every new study showing persistence in soil and water. Researchers dig for alternatives, both in process and in molecular design, aiming to reduce unwanted environmental consequences. New catalytic pathways promise more selective transformations and less waste. Some universities and start-ups seek materials that keep fluorine’s valued properties with shorter environmental lifespans. The path forward will rely on a mix of smart regulation, innovative chemistry, and the willingness of the industry to adapt. When new laws or public concern shift priorities, compounds like trifluoropropionic acid get reevaluated in the harsh light of scrutiny, pushing all involved to lead with both caution and creativity.
Opening the Door to New Chemistry
My experience in journalistic research has taught me that the smallest, strangest-sounding chemicals can have outsized effects. Trifluoropropionic acid is a great example. On paper, it reads like an obscure lab supply, but people underestimate how it plugs gaps across fields. Laboratory teams count on trifluoropropionic acid to build fluorinated molecules. These compounds often power the newest pharmaceuticals, agrochemicals, and high-performance materials. The key is the strong influence that fluorine atoms add, giving products better endurance and sometimes allowing entirely new functions.
Making Medicines Smarter
The pharmaceutical industry leans heavily on specialized building blocks. Trifluoropropionic acid steps up in this space because it helps create new drug candidates. Adding fluorine atoms can increase a drug’s absorption or slow its breakdown. For instance, the synthesis of certain antivirals and anti-cancer agents takes advantage of this acid. Big health breakthroughs tend to depend on making small tweaks at the molecular level, and trifluoropropionic acid can turn a promising compound into an actual medicine.
Boosting Crop Science
Ask anyone who has seen the crop chemistry pipeline up close, and they’ll confirm this: new herbicides and pesticides demand ingredients that defy tough environmental conditions. Fluorinated chemicals bring that kind of staying power, helping treatments persist just long enough to be effective, then break down as needed. In practice, research teams use trifluoropropionic acid when developing crop protection molecules that resist rain and bright sun. The end result is that farmers get access to more targeted, longer-lasting tools—fewer wasted applications, better yields, and cost savings down the road.
Engineering Better Plastics and Coatings
The backbone of many modern materials traces to clever chemistry, and here as well, trifluoropropionic acid earns respect. It helps create polymers and specialty coatings for wires, medical devices, and even non-stick cookware. The magic comes from the way fluorinated parts resist water, stains, and heat. In the lab, scientists use trifluoropropionic acid as a starting point to build up these tough molecules, stacking them until they reach just the right performance for demanding uses. Without this acid, the choices for high-performance plastics and coatings would shrink.
Looking at Environmental Challenges
No conversation about chemicals in modern society feels complete without addressing the environment. Having explored environmental reporting, I've watched regulators raise legitimate questions about persistent, bioaccumulative chemicals—fluorinated compounds under this umbrella. Trifluoropropionic acid, though useful, needs careful stewardship. Research is ongoing into biodegradable alternatives and new processes that rely less on persistent molecules. Industry and academic partnerships are testing “greener” pathways for both synthesis and disposal, aiming for progress without compromise.
What Comes Next?
The story of trifluoropropionic acid encapsulates a larger pattern: specialized chemicals paving the way for tomorrow’s treatments, food security, and innovative products. If society wants both innovation and responsibility, ongoing transparency and safer design will remain essential. It takes steady work, frank discussion, and focus on what works best for both industry and the broader public. Trifluoropropionic acid will likely remain influential wherever cutting-edge chemistry is needed, but its place should never overshadow the continuing push for smarter, safer science.
Understanding the Formula and What That Means
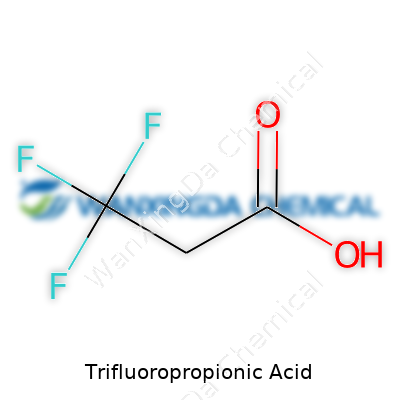
Trifluoropropionic acid goes by the straightforward chemical formula C3H3F3O2. This is more than an arrangement of letters and numbers on a bottle. Each atom in the formula tells a story. You’ve got a three-carbon chain (propionic acid as the backbone) and three fluorine atoms snugged onto the second carbon, turning an ordinary organic acid into a tool with some real bite in modern labs.
In structural terms, chemists usually write it as CF3CH2COOH. The “CF3” at the start stands for a trifluoromethyl group—three fluorines on a single carbon. Next comes a two-carbon chain before you hit the carboxylic acid group. This setup isn’t just for show. Those fluorines don’t play nice: they drag electron density away from the rest of the molecule, making the acid much stronger than its plain old cousin, propionic acid.
Real-World Uses: Not Just a Whiteboard Curiosity
Many folks picture acids in classrooms or labs, bubbling in beakers. The reality cuts deeper. Trifluoropropionic acid makes things happen in pharmaceutical chemistry. Adding fluorine often boosts how drugs move through the body, improves stability, and blocks the enzymes that would chew these drugs up. This little acid shows up as a building block for medicines that handle inflammation, diabetes, and more.
Fluorinated chemicals don’t stop with medicine. Agricultural scientists use them for new pest control options. Electronics makers build them into special plastics and advanced coatings. The backbone stays the same, but that row of fluorines on the molecule opens doors that regular organic acids can’t unlock.
Why Pay Attention?
Chemicals like this bring both promise and warning. On the one hand, trifluoropropionic acid does jobs that non-fluorinated acids just can’t touch. Its strength and reactivity push science forward—right into our medicines and modern gadgets. I’ve seen how a tiny tweak, like adding a few fluorines, can either make or break a whole research project. These tweaks change everything, from solubility to how a compound resists breaking down in harsh conditions.
But those same features also spell trouble if mishandled. Strong acids that resist breaking down make disposal an issue. Fluorinated compounds tend to stick around in the environment. Plenty of scientists are worried that, as these chemicals spread through industry, runoff or mishaps could spark long-term problems. We’ve watched other “forever chemicals” like PFAS turn from scientific marvels into global headaches. It’s important to learn from that.
Solutions Need Level Heads
More and more research groups are experimenting with greener ways to make and break down fluorinated acids. Industry leaders focus on recycling, closed processes, and tighter regulations at every stage. Medicines built from these acids come with special studies to track what happens in the environment after they serve their purpose.
Society benefits from chemicals like trifluoropropionic acid, no doubt. The challenge sits in using them carefully, making sure strong tools don’t create new problems faster than they solve old ones. Care, transparency, and smarter design matter at every step—from lab bench to manufacturing plant.
Why Chemical Safety Hits Home
Trifluoropropionic acid doesn’t make headlines outside research labs or chemical plants, but people who work with it know there’s not much room for error. I’ve been around labs enough to see what happens when routine turns into risky business. People usually think of big accidents—spills that shut down a building, thick vapors and alarms. Day-to-day mishaps, though, can be just as dangerous over time.
Treat Hazardous Chemicals Like Live Wires
This acid isn’t as forgiving as table salt or vinegar. It’s corrosive and volatile, and it won’t wait for someone’s attention span to return. Everyone who’s ever knocked over an unlabeled bottle can tell you the panic gets worse if you’re not sure what’s inside. This acid eats through metal and skin. It stings eyes, nose, and lungs almost at once. OSHA and similar watchdogs worldwide keep close tabs on substances like this for good reason.
Storage Starts With Respect—And the Right Containers
Anyone setting up a lab shelf shouldn’t just reach for any glass bottle or plastic jug. Trifluoropropionic acid reacts with metals; storing it in something like stainless steel lays out an invitation for trouble. Only containers designed for corrosive, volatile acids belong in the picture: heavy-walled glass with leak-proof caps, or fluoropolymer bottles that shrug off the acid’s punch. I’ve seen storage rooms where the difference between a safe week and a leak on the floor came down to good labeling and the right bottle type. Anything less is just a gamble.
Temperature and Ventilation: Little Details, Big Difference
Chemicals sometimes remind us of moody pets: the slightest shift in temperature and they change behavior. Store this acid cool and out of sunlight. Lock it in a chemical safety cabinet if possible. These cabinets are built to keep fumes from escaping, and they’re fire-resistant. Forgetting to close the cap or letting bottles crowd together can speed up accidents nobody wants to explain. Proper ventilation, like working beneath a fume hood, is a must. One whiff of this stuff makes clear why—irritation starts almost right away and doesn’t let up easily.
Gear Up—Because Gloves Alone Aren’t Enough
Latex gloves won’t cut it here. Nitrile or neoprene gloves work better. Lab coats, safety goggles, maybe even a face shield if there’s any chance of splashing. People working in labs sometimes skip full protection for quick tasks—years of stories from ER doctors show how short cuts lead straight to burns or worse. Even if you’re just transferring a few milliliters, every bit of skin covered counts. Eye wash stations and safety showers nearby have saved real lives. They aren’t just boxes to check on a safety list.
The Human Factor: Training and Clear Rules
No one expects every worker or researcher to memorize chemical encyclopedias. Ongoing training, not just a yearly refresher, makes the real difference. Without practice, even smart people make dumb mistakes. Supervisors and senior staff should show—not just tell—new colleagues how to store and handle acids like trifluoropropionic acid. Repeat reminders matter. Posting clear rules and organizing regular drills keeps good habits alive and stops complacency from creeping in.
Finding Smarter Solutions
Long term, some labs are moving to less hazardous chemicals, especially where students or new staff do a lot of the hands-on work. While that’s ideal, it isn’t always possible. Using digital inventory systems with real-time tracking, double-checking labels at each step, and sticking to written procedures keeps things moving smoothly. Mistakes love disorganized shelves and guesswork—order keeps everyone safer, every single day.
What Trifluoropropionic Acid Means for Safety and Health
Trifluoropropionic acid (TFPA) sits in the group of fluorinated organic acids, so it grabs attention both in the chemistry lab and among environmental watchdogs. A small, sharp-smelling liquid, its chemistry brings plenty of practical uses. Whether tweaking pharmaceuticals or crafting specialty coatings, people value its properties. But as history keeps repeating, chemicals with big advantages sometimes leave invisible tracks that come back to haunt us. Trifluoropropionic acid, like many fluorinated compounds, triggers nerves over toxicity and environmental persistence.
Speaking Plainly: What Are The Hazards?
Anybody who's spent time around strong organic acids can tell you: respect is key. TFPA will cause severe irritation if you breathe its vapors or if it lands on your skin. Even in diluted form, repeated contact dries out the skin. Inhalation brings a tightly wound feeling in your throat and nose, and the eyes burn. I once watched a graduate student accidentally spill a few milliliters on her glove—her surprise showed just how fast it chewed through.
The bigger worry, though, goes deeper. TFPA’s relatives, including trifluoroacetic acid, don’t just evaporate and disappear. They drift into water, soil, even into living things, and they often break down at a snail’s pace or not at all. Anything that lingers in our environment that long means more people and more wildlife bump into it, with exposure accumulating over years. That’s the pattern behind a lot of PFAS-related health scares—cancers, thyroid issues, immune disruptions. While TFPA has not been as widely studied as its heavier cousins, its structure gives plenty of reasons to take it seriously.
Why Trifluoropropionic Acid Needs Scrutiny
Science demands judgment calls. Sometimes we get lucky, and short tests reveal the main risks. With TFPA, the evidence is still emerging, but we know the trifluoro group grants unusual stability. What won’t break down quickly in nature often builds up in groundwater and can pass through current filtration systems. I’ve seen labs spending fortunes trying to get rid of trace fluorinated acids, only to see persistent readings pop up month after month. This is why environmental agencies now pay sharp attention to any trifluorinated chemical, even if it hasn’t hit the headlines yet.
Acute toxicity, on the occupational side, reminds anyone using TFPA to tighten protocols. Respirators, gloves, and fume hoods aren’t luxury items—they’re baseline precautions. Spills must be contained fast, because TFPA’s spread into drains or onto benchtops creates more risk later. For companies and research centers, training sessions need more than box-checking. I remember one year a younger scientist forgot the acid neutralizer when cleaning up a small TFPA spill; it cost a week of extra cleaning, and a lot of tension about lab safety culture.
Better Practices, Less Harm
No one can wish away chemicals like TFPA. The right path means investing in closed systems for handling, and keeping storage tightly secured. For labs, switching to less persistent alternatives where possible reduces risk. Water treatment utilities should work on advanced filtration, like activated carbon or high-pressure membranes, which snag more of these persistent acids. Open reporting and tracking—something the European Chemicals Agency has started for similar acids—helps keep public focus and regulatory scrutiny sharp.
Staying ahead of toxicity concerns isn’t about knee-jerk bans or blaming one industry. It’s about honest assessment, research, and solutions that match the scale of the issue. TFPA reminds us that every breakthrough in the lab needs a partner in safety, or we risk poisoning our future water, soil, and health for some short-term gains. Knowledge and vigilance are our best tools.
Understanding the Market
Trifluoropropionic acid usually comes up in conversations among scientists working in fields like pharmaceuticals, agrochemicals, and organic synthesis. Word travels quickly in these circles when a reliable vendor pops up, mostly because supplies can be inconsistent and purity demands run high. Availability shifts depending on global supply chains and regulations. Some compounds flow easy through catalogs and websites, but others, like trifluoropropionic acid, often turn into a scavenger hunt.
Where to Buy
In my experience, most researchers start with specialty chemical suppliers. Companies with a reputation for handling fluorinated intermediates pop up as strong contenders. Sigma-Aldrich, TCI, and Alfa Aesar stand out in the minds of many lab managers. Typically, these suppliers hold enough inventory to ship to academic labs and mid-size R&D facilities. It’s not common to see this acid in general laboratory or hardware stores, since storage and shipping require tight controls due to corrosive properties.
Online marketplaces focused on science—like Fisher Scientific or VWR—sometimes carry trifluoropropionic acid, though regional availability swings with regulatory rules. Before ordering, buyers often jump through several registration and verification steps. I recall colleagues facing back-and-forth emails just to confirm institutional status and permissible usage, especially in countries with tight chemical import rules.
Packaging Sizes On Offer
Most suppliers pack trifluoropropionic acid in small glass bottles for lab use, with the common sizes ranging between 5 grams and 250 grams. Large containers run into issues with handling and shipping, so bulk buyers—mainly chemical manufacturers—usually coordinate directly for drum shipments or larger packages. In the lab, breakage and exposure risk push buyers to avoid bigger bottles, unless experiments demand scale-up quantities.
The real world’s lesson: For anything beyond a 250-gram bottle, discussions with a sales representative start early. They want to know intended use, lab accreditation, and who signs off on hazardous shipments. For researchers, this step turns into a ritual. Documentation flows back and forth—purchase requests, end-use declarations, safety acknowledgements. It slows down spontaneous research, especially for newer labs still learning the process.
Why Traceability Matters
Trifluoropropionic acid qualifies as more than just another building block. Its handling carries health and environmental risks. The industry has moved toward traceable sourcing. Regulatory pressure means suppliers need to show where materials originate. The customer is responsible, too: once you buy the acid, you need a plan for handling, storage, and waste treatment. Auditors and internal compliance teams check this regularly, adding another layer of paperwork and oversight. I’ve seen labs run into trouble over missing manifests or mislabeled bottles.
Pathways Toward Smoother Procurement
The maze of regulations and documentation will not shrink anytime soon. Digital procurement platforms, with integration to institutional compliance dashboards, could cut down the red tape. As more research goes remote, secure online portals that show real-time inventory, track regulatory paperwork, and automate approvals could make life easier for scientists. At the same time, strong partnerships with trusted suppliers save both time and headaches, as repeat business brings better communication and understanding of requirements.
Safety deserves the last word. Every bottle of trifluoropropionic acid ordered needs proper storage, robust inventory tracking, and a plan for spills or accidental exposure. Old habits—such as hoarding mystery bottles in shared refrigerators—don’t fly anymore. The stakes rise along with the art of chemistry, making careful purchasing and robust documentation an integral part of every lab’s daily routine.