Reflections on Trimethylsilyl Trifluoromethanesulfonate: The Uncelebrated Workhorse of Modern Chemistry
Historical Development
Most folks, even in scientific circles, rarely mention Trimethylsilyl Trifluoromethanesulfonate unless the conversation veers toward serious organic synthesis. The road that shaped this compound traces back to the advancing waves of synthetic chemistry during the late 20th century. Labs were bustling with energy, especially in the 1960s and 1970s, pushed by the search for ever more dependable silylating agents. Chemists needed tools that could work cleanly with alcohols, amines, and sometimes stubborn carboxylic acids to produce protected derivatives, paving the way for longer and more complex synthetic routes. Trimethylsilyl trifluoromethanesulfonate entered the fray as something of a revelation. This reagent didn’t dawdle or linger; it let folks sidestep frustrating protection bottlenecks, making previously daunting syntheses more reachable. It said something about the creativity, tenacity, and sheer problem-solving drive that defined that era.
What This Compound Really Is
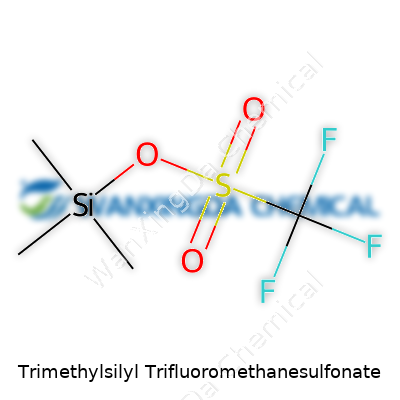
Trimethylsilyl triflate, as it's often called, brings some muscle to the everyday world of organic synthesis. It isn’t fancy or flashy. The formulas show a pairing of a trimethylsilyl group bound to a trifluoromethanesulfonate anion, both of which play distinct roles: the silyl group shields sensitive functional groups, while that electronegative triflate acts as a leaving group, driving all sorts of transformations. In its pure form, this colorless liquid punches well above its weight in reactivity, slicing through barriers others leave untouched. Chemists measure its value in ease of use. Those tiny vials pack a lot of punch, often with only a few drops needed to tip a reaction toward success.
Physical & Chemical Properties
This compound doesn’t seem remarkable on first glance. Pour it out, and you notice a clear, mobile liquid with a sharp, biting aroma. Its boiling point lands well above the lab bench favorite, dichloromethane, a factor that demands some care when using it in large batches. Extreme moisture sensitivity might frustrate those expecting a rugged, forgiving reagent. Trace water turns silyl triflate into a mess of byproducts, often just when the clock and the patience are running thin. As soon as it leaves the bottle, chemists move quickly, cap their flasks, and keep the desiccators close by. Reactivity stems largely from the silicon-oxygen bond, which manages to balance strength and lability in a way that serves as the foundation for countless transformations.
Technical Specs and the Story on Labels
Reading over the technical documentation, it tells you a lot without ever really letting you feel what it means to handle such a reactive fluid. Laboratories ship it in amber bottles, the labels fading after a few months if you let chemicals drip down the side. Concentration sits near ninety-eight or ninety-nine percent purity. Only a fraction of the world’s population gets to see the detailed batch analysis that comes with each shipment, usually a few peaks on a chromatography readout. Regulatory classifications warn of moisture reactivity, with pictograms emphasizing the corrosive and toxic risk. It’s a reminder that making real progress with chemistry often involves a fair bit of risk management.
Pathways to Production
Manufacturing this stuff follows a line etched by earlier experiments with simpler silylating agents. Most commercial processes start from trimethylsilyl chloride and swap out the chloride for the trifluoromethanesulfonate using silver triflate, itself a potent source of trouble if mishandled. The solvent, often dichloromethane or acetonitrile, soaks up the exotherm that comes from mixing two eager reagents. Few would say this process is green or sustainable—silver salts cost dearly and generate waste, and solvents drift out of fume hoods in tiny, persistent clouds. Efficiency often means reclaiming unused starting materials, juggling purity, and coaxing product yields from reluctant mixtures. Success in this business means balancing reliability with safety, never cutting corners where people and the planet might end up paying the price.
Reactions and Chemical Tinkering
Trimethylsilyl triflate plays a pivotal role in modern chemistry. Protecting groups shield alcohols or amines from unwanted sidetracking: no one wants to waste weeks fixing a scrambled intermediate. This silylating agent achieves rapid conversion under conditions that other reagents shy away from. It’ll transform enolizable ketones, promoting their conversion into silyl enol ethers, the unsung heroes of aldol chemistry and other carbon–carbon bond-forming feats. Using this reagent, I’ve seen graduate students, myself included, breathe easier after it rescued a sluggish reaction from becoming a teachable failure. Triflate leaves so cleanly that purification rarely turns into a slog. But beyond protection, trimethylsilyl triflate finds a place adding silyl groups to all sorts of reactive centers, forming what those in the know call “super silyl electrophiles.” It catalyzes glycosylation and boosts sluggish acylation, driving modern methods that cut across medicinal, materials, and even agrochemical chemistry.
People Call It Many Names
Ask around, and most chemists cut the formalities, calling it “TMSOTf” with a quick roll of the tongue. Purists cite the full title: trimethylsilyl trifluoromethanesulfonate. Trade catalogs sometimes call it “TMS triflate,” “TMSOTf,” or just “silyl triflate.” Each name points to the same clear liquid, the same careful handling, the same reliable shield for vulnerable molecules.
Safety, Rules, and Realities
For all its chemical finesse, this compound deserves respect. Given the nastiness of triflate residues and the explosive reactivity with water, fume hoods remain the only sensible workspace. Standard safety protocols aren't optional; every bottle comes with reminders for gloves, goggles, lab coats, and competent waste handling, but none of these labels conveys the caution that slips into every movement when you’ve seen someone ignore the rules. You learn quickly to double-check your glassware for moisture, to use septa and syringes, and to keep spill kits within reach. Waste disposal remains an issue: silylated waste meets harsh treatment to make it safe for landfill or incineration, and accident reports from hurried handling show that even seasoned chemists pay the price for shortcuts.
Where It Makes a Difference
Trimethylsilyl triflate isn’t just another bottle on the shelf. In academia, it stands as a keystone for the protection and activation of functional groups, allowing long routes to reach the finish line. Pharmaceutical labs use it to steer glycosylation reactions, where sugars attach to a molecule in just the right spot to change how a drug behaves. The materials world finds uses in semiconductor microfabrication, where this compound silylates surfaces, prepping them for layer-by-layer buildup. Every field that relies on precise modification of molecules owes something to the versatility of this single tool. My own work on advanced intermediates saw this reagent flatten timelines and turn potential dead-ends into new products.
Research, Development, and the Long Road Forward
Open any chemistry journal, and you’ll find research that draws heavily from what trimethylsilyl triflate can do. Every tweak to the reagent, every effort to swap out the triflate for something safer or greener, comes straight from the growing demands of a world less forgiving of waste and risk. Some researchers hunt for alternatives that leave less of an environmental footprint; others work to design processes that recycle or regenerate byproducts, slashing waste while holding on to the unique reactivity. The innovation here isn’t about making old reactions work better; it’s about rethinking what’s possible when the basic tools aim higher for safety, cost, and accessibility.
What We Know About Toxicity
Risks attached to trimethylsilyl triflate don’t just fade after the reaction. Toxicology studies underline trouble for skin, eyes, and lungs. Short-term exposure can set off severe irritation, and long-term risks stay murky for those outside the academic and industrial safety offices. Animal studies hint at acute toxicity if inhaled or misapplied. Watching for unexpected symptoms, providing training, and documenting near-misses all play into a culture that values safety as much as new discoveries. I’ve seen colleagues suffer minor scrapes, a reminder that simple mistakes can still cost dearly. Anyone handling this agent ought to enforce strict controls, never letting routine dull the critical edge that good lab work demands.
What Comes Next
Looking beyond the bench, the conversation around trimethylsilyl triflate grows. There’s pressure to adopt greener solvents, safer and simpler procedures, and to track every gram that leaves the waste stream. Future directions point toward finding better substitutes for costly starting materials, engineering catalysts that mimic or surpass what TMSOTf can do without the same baggage, and linking lab practices to a larger sustainability effort. Whether these changes unfold in academic or commercial labs, they spring from a need to balance progress, safety, and stewardship. As much as we’ve leaned on Trimethylsilyl Trifluoromethanesulfonate so far, the road ahead calls for new ideas, fresh methods, and a willingness to let go of old habits. The story of this silylating agent runs parallel to the broader arc of chemistry—problem-solving through innovation, guided by facts, grounded in real-world needs, and sharpened by an unrelenting drive to do better for people and the planet.
The Real Value Behind a Difficult Name
The first time I saw a bottle labeled “Trimethylsilyl Trifluoromethanesulfonate,” it looked more like a password than a chemical. But beneath that unwieldy name sits something that serious synthetic chemists count on almost daily. Often shortened to TMSOTf, this compound earns its keep in organic labs where making new molecules quickly — and safely — pulls real results.
Why Chemists Reach for TMSOTf
Making new medicines, materials, or even flavors usually means building molecules from smaller pieces. This process gets tricky when moisture or stray acids ruin careful work. TMSOTf steps in as a water scavenger, a tool that helps chemists keep reactions dry so sensitive compounds won’t decompose. It pulls away unwanted water, letting the main reaction run clean and strong.
But there’s more. Chemists also use TMSOTf to add the “trimethylsilyl” protective group to alcohols — a move that shields the alcohol from dangerous reactions until it’s time to remove the protection and finish the job. For instance, someone building a complicated cancer drug may want to keep a certain alcohol group safe through ten or fifteen steps. TMSOTf gives them that shield. The fact that it reacts quickly and selectively — especially compared to other similar chemicals — often means fewer byproducts to clean up later.
Why Safety and Reliability Matter Here
Doing real chemistry outside textbooks showcases all the ways a process can fail. Minor traces of water, leftover acid, or weak reaction conditions sometimes flip a whole project upside down. TMSOTf brings reliability. In my own experience, switching from less effective alternatives to TMSOTf boosted reaction yields from frustrating puddles to healthy piles of powder. This difference can mean the world, especially in projects with tight timelines and tight budgets.
Chemists aren’t just chasing numbers; they’re often tying up student hours, grant funds, and company dollars in every flask. Knowing a chemical will deliver saves everyone time and money. Patents, new treatments, and better efficiency often ride on that kind of reliability.
Room for Caution—and Smarter Use
Effective as it is, TMSOTf isn’t something you use carelessly. The same features that make it powerful create risks. It reacts aggressively with moisture — a blessing in the flask, a curse on exposed skin or in open air. Labs working with it need airtight handling and proper training. Even after nearly a decade of lab work, I’ve seen enough accidents to respect what good habits — like working in a fume hood — can prevent. Industrial and academic labs have plenty of safety guidelines that acknowledge these dangers.
Lab life also feels the pressure to replace hazardous chemicals where possible. Researchers have begun exploring safer, greener alternatives for some of TMSOTf’s roles, especially in undergraduate teaching labs and large-scale production. Finding replacements that work just as well remains a work in progress.
Worth the Complexity
TMSOTf brings muscle and precision to synthetic chemistry. For those working on real-world problems — cancer treatments, advanced materials, specialty chemicals — it doesn’t just add convenience. It’s become a staple tool, trusted for the control and consistency it provides. Until someone figures out something safer and just as effective, bottles of Trimethylsilyl Trifluoromethanesulfonate won’t be gathering dust on the back shelf anytime soon.
Why Extra Care Around Strong Chemicals Isn't Up for Debate
Work in any lab long enough and you’ll pick up a kind of respect for the more “ambitious” reagents on the shelf. Trimethylsilyl trifluoromethanesulfonate (TMSOTf) counts among that group. Its strong reactivity in organic synthesis makes it a go-to for silylation and protecting group strategies, but ignoring safe handling here isn’t a job hazard—it’s a gamble.
What Makes TMSOTf So Demanding?
TMSOTf reacts aggressively with water, releasing triflic acid, a substance that eats through skin, eyes, and anything organic like a hot knife through butter. I learned that lesson back in grad school when a careless pipetting slip sent a few drops sizzling through the bench paper—luckily, not through someone’s hand. Sharply irritating vapors roll off even slow leaks, making it easy to forget this clear liquid demands real caution the moment the cap comes off.
Concrete Steps For Safety: No Shortcuts, No Excuses
Folks getting started with TMSOTf need to think about protection from the very start. Too many stories circulate about folks skipping goggles or gloves because they “never spill.” Full-wrap safety glasses, chemical-resistant gloves (think nitrile at least, double upped), and a solid lab coat become your daily wardrobe. I’ve seen careless handling create emergency eye wash epics for no good reason.
Chemists keep their workspace as dry as possible before setting up. Water in the air, leftover in pipettes, or a fingerprint in a flask sets off dangerous reactions. Dryboxes, nitrogen-filled glove bags, and those old-school calcium chloride tubes might feel excessive, but anyone who’s dealt with the clouds of acid smoke knows better.
In public or shared labs, fume hoods earn their reputation. The glass sash blocks splashes and shields anyone nearby from the acidic vapors that drift out if you lose concentration. TMSOTf doesn’t give a second chance if you catch it in the eye or lungs. I’ve seen lab-mates who didn’t take that fume hood warning seriously; they got a much-too-close tour of the campus health center.
What To Do If Things Go South
Every bottle of TMSOTf carries an ugly burn hazard, so keeping calcium carbonate handy for spills helps snuff out rogue drops fast. Paper towels alone won’t cut it once the stuff hits the benchtop. For major spills, contain the area, alert your team, and reach out for professional cleanup. Contact with skin or eyes? No hesitation—run to the emergency shower or eyewash and flush for a solid 15 minutes.
Keeping Risk in Check—For Yourself and Your Team
Nothing builds a safety culture like watching out for each other. Before working with TMSOTf, everyone deserves a walk-through, pointing out where to find spill kits, PPE, first aid stations, and exits. Emergency drills save brainpower for the real thing, so folks don’t panic if a bottle tips or a drop lands where it shouldn’t.
Looking Forward: Safer Chemistry and Smarter Labs
Some teams have swapped in less hazardous alternatives for certain reactions, but there’s no replacement for basic vigilance. Double-checking—instead of trusting “good enough”—saves both injuries and careers. Every time I reach for a bottle of TMSOTf, I remember stories from folks who thought they’d “never be that person.” Best practice boils down to treating every milliliter like it’s been waiting for a mistake, so nobody winds up a cautionary tale.
Trimethylsilyl Trifluoromethanesulfonate, or TMSOTf to those of us working in the lab, isn’t just some bottle you shove on a shelf and forget about. This reagent, prized for activating reactions, asks for some respect because of how feisty it gets around water and air. I’ve seen a few bottles go from clear to cloudy in no time, just from casual handling. Corrosive fumes and ruined samples, all from skipping a few steps, aren’t stories anyone likes repeating.
Aiming For Longevity, Not Mayhem
“Keep it dry and keep it cold.” That saying gets thrown around a lot, and with TMSOTf, it’s not just tradition, it’s pure necessity. Set this compound loose at room temperature with any hint of humidity, and you’ll smell trouble before you see it: strong acid forms, glassware etches, and projects derail. This isn’t just a headache for your benchwork; you’re gambling with both your safety and investment. Making the effort to store it at low temperatures, like inside a chemical fridge or freezer, isn’t overkill—it protects potency and avoids nasty surprises.
Sealing is non-negotiable. Air sneaks in, and soon the reagent degrades or, worse, pressure builds. I’ve used glass ampoules or tightly capped bottles under a dry inert gas like nitrogen or argon. A desiccator helps, but pairing both low temperature and inert gas guards the contents from almost anything your lab’s atmosphere throws at it.
Storing for Safety, Not Just Shelf Life
Ignoring proper storage doesn’t just eat into yield. Health and safety folks have to think about more than the bottom line. Corrosive vapors from poorly sealed containers invite damage to metal shelving, lab electronics, and, most critically, skin, eyes, and lungs. Local fire marshals and safety officers grew their rules from real disasters, so labeling and segregating TMSOTf with other water-sensitive reagents makes everyone’s life easier. I’ve dodged more than one “close call” by making sure nothing gets shelved above strong bases or stacked near oxidizers—these chemicals don’t mix with TMSOTf’s temperament.
Reducing Waste and Risk With Hands-On Habits
Small changes in storage practice ripple outward. Avoiding condensation by letting bottles warm to room temperature while still sealed before opening, for example, keeps reactions reliable and waste low. Instead of storing large, half-used bottles, I transfer smaller amounts into ampoules—less air exposure, less risk. Keeping labs organized and post-it notes off bottle labels also helps trained and new folks alike steer clear of careless mistakes.
If you manage a teaching lab, running drills on proper handling isn’t just academic—students remember a minor “whoosh” from accidental contact far longer than a slide in their safety binder. As the Institute for Safe Medication Practices points out, the habits you build today prevent serious incidents tomorrow.
Everyday Vigilance Delivers Peace Of Mind
Safeguarding TMSOTf isn’t just about preserving a bottle’s worth. Proper storage builds a safety culture, keeps valuable chemicals potent, and protects anyone walking through the door. After years at the bench, I’ve seen that a few extra seconds sealing, labeling, and segregating chemicals brings peace of mind that beats any fire drill.
In the world of organic chemistry, few reagents spark caution quite like Trimethylsilyl Trifluoromethanesulfonate. Folks in the field often call it TMSOTf, and plenty of chemists rely on it for its power to trigger reactions that other materials can’t. Its value is real, but the risks it brings shouldn’t fade into the background. I’ve watched colleagues get burned—sometimes literally—by treating TMSOTf as just another bottle on the shelf.
What Really Makes TMSOTf a Danger?
The worst thing about TMSOTf isn’t how it looks or smells. You barely notice it until it meets water. The reaction hits fast, releasing toxic and corrosive fumes. In my own experience, even a small spill in a fume hood fills the air with acrid vapors, which means anyone nearby breathes them in before they think to hold their breath. That gas isn’t just a throat-tickler—it can cause damage deeper in the lungs and eyes. Medical journals and safety records point to the risk of permanent eye and respiratory harm if someone isn’t quick with the right first aid. Gloves and eye protection, by themselves, only slow the risk. Any exposed skin stings and reacts quickly, so double layers and splash shields become more than suggestions.
TMSOTf doesn’t just react with water. Even a trace of moisture in the air can start an exothermic chain, raising temperatures to points where plastics melt and glass can crack. I’ve seen researchers underestimate ventilation, storing their bottle on a crowded bench instead of back in a dry, dedicated cabinet. Do this long enough, and leaks or broken seals turn into fire hazards. The trifluoromethanesulfonate group releases acidic byproducts, eating through surfaces and leaving surprises for whoever cleans the lab next.
Pressure, Waste, and the Shadow of Environmental Impact
There’s another hazard people skip: the pressure inside containers. TMSOTf can build up vapor pressure, especially if mishandled or stored in warm areas. Over time, this fits the definition of a bomb, with risks to anyone handling storage or disposal. Old vials left unchecked sometimes pop their tops, splashing liquid around. Even trained chemists forget this under deadline pressure.
On disposal, waste containing TMSOTf turns into a nightmare for hazmat crews. Water-reactive and corrosive waste sits in a class by itself at most universities. Disposal mistakes have led to drains getting eaten away, fumes in public hallways, and significant cleanup costs. Waste contractors charge a premium to handle this material, and with good reason. Down the chain, there’s concern about triflate residues in water systems, which standard treatment may not handle.
Reducing Risk Isn’t Impossible
No reagent deserves the label “necessary evil,” but respect has to match power. The right steps start simple: only pull enough TMSOTf for the day, keep it under dry nitrogen, and label everything with the start and end dates. Freshness counts. I’ve made a habit of double-checking storage every week, and insisting others in the lab report even the smallest spills immediately.
Training matters more than endless paperwork. New lab members should practice basic drills—glove changes, emergency eye-wash use, and fast spill response—until muscle memory takes over. Safety culture grows across years, not days, and TMSOTf rewards that investment. No reaction is worth risking long-term health or the safety of coworkers for short-term gains.
Trimethylsilyl trifluoromethanesulfonate, often called TMSOTf, packs a punch for anyone working in organic synthesis. This reagent turns up in transformations that call for finesse and reliability. Purity, the chemical’s quiet partner, shapes every step. Chemists eye purity like a hawk because a contaminant can wreck a reaction or chew up valuable starting materials. Sometimes, only pure TMSOTf means the difference between a clean conversion and a frustrating mess.
The Numbers Behind Purity
Makers of TMSOTf use tight specs—often demanding purities above 98%. That number doesn’t come from nowhere. Even a percent or two of water, alcohols, or leftover starting compounds can tank a whole batch. Water especially causes headaches, since TMSOTf reacts fast and furiously in the presence of moisture. For a reagent designed to scrub up hydroxyl groups or protect functional groups with its silyl moiety, stray drops of water can force unwanted side reactions or ruin the yield. Most commercial bottles guarantee moisture levels below 100 parts per million. This number speaks volumes to anyone working late in a chemistry lab, hoping their new project survives to see morning.
Impurities Make a Difference
A trace impurity does more than pad the weight of the bottle. Acidic leftovers can drive the wrong reaction or leave tough-to-remove residue embedded in the product. Solvents like dichloromethane, common during manufacture, must be kept out or risk skewing results or fouling a purification. Some folks deal with color variation; true TMSOTf ranges from colorless to pale yellow. A dark tinge might mean age, exposure, or the presence of stubborn byproducts.
Precision matters for scientists scaling up reactions. Someone running a ten-milligram test might struggle through the noise, but a kilo-scale run can expose problems quickly and expensively. Inconsistent purity also messes with experiment reproducibility. If a chem lab can’t trust that new bottle, researchers face detours through dry-downs, titrations, or in-house testing, all of which cost time and money.
Taking Quality Seriously
Top manufacturers invest real effort in distillation and careful packaging. No reagent survives humidity, so companies ship TMSOTf in tightly sealed glass with secure liners. Labs take the hint and keep bottles closed, sometimes torching the air out with inert gas before sealing again. Every time the bottle sits out, the risk of contamination climbs; a careless step blocks progress for hours or days.
Modern labs rely on third-party purity checks. NMR and Karl Fischer titration show water and solvent content, giving chemists a true snapshot. Chromatographic methods flag unexpected contaminants and help ensure every drop added to a reaction flask brings the same strength and selectivity.
Finding Solutions on the Bench
While better controls from makers help, leading researchers double-check before use. Some labs go so far as to rebottle or redistill their own TMSOTf. High-end research often lives or dies by reliability, and no one likes a failed synthesis turning into a week’s cleanup.
Pushing for stronger standards benefits everyone. Sharing experience—down to which supplier, lot number, or storage trick worked—keeps the science honest. Purity doesn’t just show up in the bottle; it shows up in every result a chemist delivers.