Triphenylsulfonium Triflate: Beyond the Lab Bench
The Road from Discovery to Daily Use
Triphenylsulfonium triflate didn't just burst onto the scene all at once—its story ties back to the hunt for effective photoacid generators in the 1970s and 80s, as researchers struggled with the limitations of earlier organic sulfonium salts. The rise of microelectronics, especially during the boom in photolithography for semiconductors, really pushed chemists to dig deeper. My time spent paging through old scientific journals revealed just how quickly the adoption of this compound tracked with the leaps in circuit miniaturization. Dirty wafer failures and line-edge blurring in early chip manufacturing forced a shift to more robust chemistry, and triphenylsulfonium triflate showed up with its knack for clean, short-wavelength photoacid generation. You can almost sense the excitement in those old papers—chemists saw a tool that could handle the ultraviolet exposure lines that were becoming the new normal.
Hands-On with the Material
Holding a vial of triphenylsulfonium triflate, you get fine white powder that flows easily under a gentle tap. In the lab, it's stable in dry, cool storage, but moisture or bright light makes it tricky to work with—best practice means dark bottles and desiccators are always close at hand. Chemically, the triflate group tugs electrons away from the sulfonium, lending the compound high thermal stability but still making it snappy to decompose under UV or electron beam exposure. In making up photoresist blends, I've found it dissolves cleanly in most polar organic solvents. The molecule packs a molecular heft of over 400 g/mol, and its structure holds up well under most analytical techniques like NMR or infrared spectroscopy. The triflate anion resists unwanted side reactions, keeping the chemistry tidy during etch processing or thermal cycling steps—a big relief during troubleshooting late at night on a deadline.
Labeling Expectations and Chemist Realities
Labels on triphenylsulfonium triflate flasks stick to the basics: chemical formula, molecular weight, CAS number, and warnings about skin, eye, and respiratory irritation potential. Forgetting gloves or a fume hood is not an option. Tech specs get detailed—purity can matter in tenths of a percent for precision work. I've seen batches called out for 98% purity versus 99.5%, and that scrap of difference led to stubborn photoresist scumming in a series of test wafers, which cost a full day of processing time. Over the years, labeling has pushed closer to global standards, but you want to check the batch analysis sheets every time; the devil is in the details, especially for semiconductor fabs where million-dollar equipment can't tolerate surprises.
The Recipe Book: Preparation and Tweaking the Chemistry
Nothing about making triphenylsulfonium triflate really happens on a whim, at least in my experience. Classical synthesis runs with a sulfonium chloride precursor and silver triflate, usually in acetonitrile. You get precipitation of silver chloride and a clear solution of the sulfonium triflate. That's the lab-scale snapshot, but at commercial scales, process engineers chase yield, easy filtration, and minimum solvent waste. There’s pride in meeting environmental and cost targets without cutting spectral purity. On the bench, the reaction’s exotherm demands careful temperature control—one lazy moment led me to scorch a flask, and the clean-up took the rest of my shift. Once isolated, recrystallization brings out the best properties for photoresist blends, and careful drying beats back any hydrolysis problems.
Chemical Versatility and Modification Power
Triphenylsulfonium triflate has a special spot in the pantheon of photoacid generators thanks to its easy alteration. By swapping the aryl groups or tweaking the anion, chemists can dial in everything from solubility to light sensitivity and decomposition temperature. Custom blends drive breakthroughs in microelectronics, but the same core chemistry shows up in high-resolution printing, optical storage media, and advanced coatings. Over the years at research roundtables, I heard stories of labs adding fluorinated aryls for deeper UV work, or using mixed aryl compositions to beat back yellowing under long exposures. The chemistry never stands still—each modification earns its place only after clearing hurdles in performance, cost, and safety.
What’s in a Name?
In the thick of technical papers and procurement systems, this molecule hides behind several names. Triphenylsulfonium triflate, triphenylsulfonium trifluoromethanesulfonate, and sometimes just TPSOTf. Depending on the supply house or research group, you may run across these variants, but they pinpoint the same powerhouse photoacid generator. I always keep a list handy in the lab notebook to avoid doubling up on orders or confusing a tough customs agent. Those who grew up on older literature still call it by its bench shorthand, but the IUPAC name leads the way in grant applications and patents.
Putting Safety Up Front
Working with triphenylsulfonium triflate demands real respect for safe practices. Skin contact leaves irritation, and inhalation brings on coughing, so fume hoods aren't just decoration. At high concentrations or temperatures, the triflate ion can pose risks to health and facilities. Standard operating procedures cover spill control and disposal, with halogenated solvent waste handled as hazardous. I've seen facilities shut down entire bays for a quart spilled outside containment. Lab mates drilling emergency response, fit-testing respirators, and strict solvent inventory—these aren't just bureaucratic routine. They keep exposure rates low, accidents rare, and research moving.
Where the Action Happens: Applications
Photoresist technology for microchip fabrication leads the way in demand, but the reach of triphenylsulfonium triflate runs deep. It drives patterning for printed circuit boards, enables 3D microprinting, and safeguards optical memory formats that have outlasted competitors. The compound enjoys a niche in deep-UV and electron beam lithography, where its clean photodecomposition and strong acid generation set it apart from more sluggish alternatives. I've collaborated with teams working in microfluidics and bio-imprinting, where surface control makes or breaks experimental runs. Here, precise acid release on command means sharper features, fewer defects, and better device yields—key advantages when research budgets run tight.
R&D: The Current Momentum
Every time I attend a conference, R&D labs line up behind the podium with new takes on sulfonium salt photoacid generators. The game right now involves tuning the light sensitivity to shorter and shorter wavelengths, reaching below 193 nm as chip makers face Moore’s Law headwinds. Research groups work on structure-function maps, trading off quantum yield against stability in harsh process chemistries. US and Japanese teams compete to push the limits of environmental compatibility, green chemistry, and recyclability, knowing public scrutiny and cost-conscious customers don’t take shortcuts lightly. We've even started seeing crossover into organic solar cells and lightweight, flexible electronics, where traditional photolithography chemistry feels out of place.
Toxicity and Environmental Considerations
Long-term toxicity data on triphenylsulfonium triflate stays limited, but acute studies flag moderate irritation and some aquatic toxicity. Labs invoke strict containment, and waste streams route for incineration or advanced chemical treatment. I once joined a project that used advanced LC-MS to chase down trace levels in wastewater; the public demand for greener supply chains and cleaner final products leaves no margin for sloppiness. As industry tightens up, regulatory agencies in Europe and Asia have started calling for detailed lifecycle assessments and explicit emissions tracking, making environmental stewardship a core part of triphenylsulfonium triflate’s story.
Glimpsing What's Next
Microelectronics advances keep triphenylsulfonium triflate in the league of must-have specialty chemicals. The pressure to go smaller, faster, and more efficient means pushing this molecule’s capabilities right to the edge. There's growing excitement around hybrid materials, green recyclability, and new end-uses in photonic and energy-harvesting devices. The next few years look set for even tougher purity standards, fresh molecular tweaks, and tighter integration with automated process controls on the factory floor. Based on everything I’ve seen in the lab and on the conference circuit, chemists and engineers are hardly done rewriting the playbook for this staple. This chemistry holds plenty of mileage for years of discovery and transformation.
Why Triphenylsulfonium Triflate Matters Beyond the Lab
Some molecules change the world by making things around us faster, sharper, and more reliable. Triphenylsulfonium triflate fits into that category. Many people outside of specialized chemical fields have never even heard of this tongue-twister, but it quietly plays a crucial role in how we make microchips and other modern devices.
Big Role in Microchip Manufacturing
Anyone holding a smartphone, driving a hybrid car, or swiping a credit card benefits from technology shaped by advanced photolithography. The key challenge in chip fabrication comes down to carving impossibly tiny patterns onto silicon wafers. Triphenylsulfonium triflate does not grab headlines, but it acts as a photoacid generator (PAG). When ultraviolet light shines on it during the chip-making process, triphenylsulfonium triflate releases acid right where needed. The acid triggers a chemical reaction in specialized light-sensitive coatings (photoresists). As a result, technicians can draw lines and patterns at nearly atomic scales. Precision on this level keeps Moore’s Law alive, packing more transistors into every chip generation.
Why This Chemical Won’t Disappear Anytime Soon
Shrinking the components inside semiconductors is hard work. Every year, the world asks for greater speed, less energy use, and new applications from electronics. Chemical solutions that keep up with the demand must perform consistently without destroying expensive equipment or introducing toxic byproducts. Triphenylsulfonium triflate sets the gold standard for this job, because it offers high sensitivity to light and strong acid release—exactly what microfabrication demands. Without it, chip makers would spend more money on development and face new safety or environmental headaches.
Looking at Safety and Environmental Considerations
Working with chemicals like triphenylsulfonium triflate requires thoughtful handling. Those of us who have spent time around photoresist labs know well the smell of solvents and the importance of proper protective gear. Laboratories and fabs maintain strict dust control and waste treatment. The use of materials like triphenylsulfonium triflate sharpens discussion about safe waste disposal, possible exposure risks, and minimizing airborne contaminants. Companies invest heavily in closed-loop systems and best practices, but there’s always room to lower risks. Independent watchdogs and internal audits help keep operators focused on limiting accidental releases.
Innovation and Future Directions
Anyone who has watched the evolution of chip manufacturing sees that every technical leap sparks new research. Some scientists test greener alternatives to current PAGs, while others push triphenylsulfonium triflate’s performance to new heights. Research groups from top universities publish studies on tweaking the chemical’s structure for sharper imaging or faster reactions. Broader industry goals—like achieving ever-smaller transistor dimensions or using extreme ultraviolet (EUV) light—demand continuous improvement. New forms of triphenylsulfonium triflate, hybrid PAGs, or entirely new classes of chemicals may eventually appear, but its value and influence in today’s electronic landscape remain strong.
What We Can Learn from Its Story
People rarely see the hidden chemistry inside their gadgets, but these compounds make modern life possible. By demanding safer, more sustainable, and even more precise processes, we encourage progress both on the factory floor and in everyday life. As consumers and professionals, asking questions about the parts no one talks about—like triphenylsulfonium triflate—can drive better transparency, smarter design, and a healthier tech ecosystem for everyone.
Unlocking Its Structure
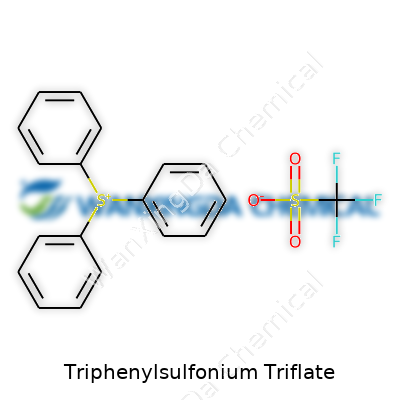
Triphenylsulfonium triflate has a reputation in scientific circles for its role in photolithography and photoacid generation. Its name points to two major components: triphenylsulfonium, a cation, and triflate, an anion. Looking at the chemical structure, the cation forms when sulfur bonds to three phenyl groups, making [(C6H5)3S]+. The triflate anion—trifluoromethanesulfonate—sports the formula CF3SO3-. Together, the full chemical formula is C18H15S·CF3SO3.
At first glance, the compound seems simple: bulky aromatic rings surround sulfur, and the triflate balances the charge and brings its own benefits to the table. In the real world, I’ve seen lab benches splattered with all sorts of chemicals, but this one draws attention for the way it can unleash acidity with the right push from light.
Significance in Scientific and Industrial Research
I remember watching a wafer processing setup where triphenylsulfonium triflate played a quiet though crucial role. When exposed to UV light, it releases strong acid—a trick that shapes circuits at sizes no human hand could etch. This talent supports the electronics we carry every day. The molecule isn’t just about its looks; it opens the way for innovation in microchip manufacturing and flexible displays.
The driving force here is the stability of the triflate anion. Unlike weaker acids or less stable partners, this one won’t decompose or interfere easily with what’s happening in the reaction flask. Chemists value compounds that stay put until they’re called upon. I learned to appreciate this quality after a few messy decompositions during grad school that left me cleaning up glassware for hours.
The Push for Safer and Greener Solutions
There’s no ignoring the fact that chemicals like triphenylsulfonium triflate walk the line between progress and safety hazards. Triflate salts, due to their high reactivity, pose health and environmental concerns if mishandled. I once spoke to an industrial chemist who stressed the importance of waste management—disposal and storage demand attention to keep byproducts from contaminating water and soil.
Researchers are working to shrink the environmental impact of photoacid generators. Newer molecules aim to break down safely or generate less hazardous byproducts. The focus turns to green chemistry—fewer toxic releases, better recyclability, and reduced waste. Success requires teamwork from chemists, environmental experts, and engineers. If there’s a roadblock, it usually involves finding a substitute that matches the performance without major compromises.
Where Next?
This molecule’s structure doesn’t just belong on diagrams or in textbooks. It stands as a marker of how chemistry solves real-world problems while bringing its own set of responsibilities. Schools and labs should emphasize not only how these chemicals perform, but how to handle them safely and responsibly. My experience has taught me that small decisions in the lab ripple out into bigger changes across industries and society.
Triphenylsulfonium triflate may not become a household name, but the way we work with it—and how we innovate beyond it—makes all the difference for both technology and our planet.
Triphenylsulfonium triflate isn’t something you’d want to leave lying around on a cluttered shelf. In labs and cleanrooms, it plays a role in advanced applications like photolithography—almost nobody bumps into this stuff outside high-tech workspaces. The risks that come with it aren’t well known to the general public, but chemists and people in electronics manufacturing know how critical it is to keep respect for safety front and center.
A Close Look at Storage Realities
In my time handling specialty chemicals, forgetting real-world constraints never ends well. Sturdy labeling and secure containers come first. Triphenylsulfonium triflate can break down under light or moisture, so airtight amber glass bottles are a staple. These keep out water and UV rays, reducing the chance of chemical changes that can turn a useful photoacid generator into a waste problem or, worse, a hazard.
Every lab technician knows storage room temperature matters. Manufacturers often recommend keeping triphenylsulfonium triflate under 25°C, because heat can speed up decomposition and lead to pressure build-up. Sudden release can force out particles or vapor, and breathing that in is nobody’s idea of a good day at work.
Chemicals like this never belong near acids, bases, or anything reactive. I’ve seen mistakes: misplaced bottles, improperly closed stoppers, even drips pooling on shelves after bad capping. Cross-contamination with any reactive substance can create dangerous fumes or unintended reactions. Carelessness doesn’t just cost money; it can trigger alarms, or worse, endanger people downwind of an unexpected release.
The Personal Side of Safe Handling
Personal protective equipment always comes before curiosity in any lab, but I remember colleagues who skipped gloves or goggles “just for a minute.” That minute sometimes ends with skin irritation, stained bench tops, and, in a few cases, medical reports for chemical burns. Chemical-resistant gloves, goggles, and lab coats should be second nature when pouring, weighing, or transferring triphenylsulfonium triflate. Fume hoods should hum away so vapors don’t leave the bench area.
One overlooked step is checking for fine dust after handling powders. Electrostatic charge attracts particles, and even a wipedown can leave residue behind. Proper spill control—absorbent pads, neutralizing agents, quick disposal—is the responsibility of everyone who steps up to the workbench. Walking through these procedures with new hires or interns doesn’t just build trust, but also sets habits that stick long after training.
Building Habits, Not Just Rules
Safety isn’t about obscure regulatory compliance; it’s about stopping preventable injuries and keeping the people making technology safer. Technical data can only take us so far—real learning comes from shared experience and two-way communication in the workplace. If you see something off, say something. Ask co-workers if they’ve double-checked bottle seals or reviewed spill cleanup steps this week.
Shared respect for dangerous substances comes from taking even “simple” steps seriously. In the world of specialty chemicals, shortcuts wind up costing time and, sometimes, lives. Thoughtful storage and handling protect more than laboratory results; they protect the people doing the work.
A Quiet Workhorse in Modern Manufacturing
Many people outside of chemistry circles have never heard of triphenylsulfonium triflate, but this salt has quietly shaped entire branches of technology. I’ve seen its impact in areas from chip fabrication to coatings. Its special feature? On its own, it's unremarkable, but when it hits UV light, it gets to work, setting off reactions that wouldn’t happen otherwise. This photoinitiation has given people incredible control over polymer chemistry.
Microchips and Photolithography
Nowhere is this chemical’s quiet effectiveness more obvious than in semiconductor factories. Workers layer silicon wafers with light-sensitive films called photoresists, and triphenylsulfonium triflate makes these layers “developable” by light. UV beams strike patterns onto the wafer, and the initiator turns light energy into acid, which triggers changes in the resist. That means better circuit lines, sharper patterns, and ultimately, smaller, more powerful chips.
I once watched a line engineer explain how switching to stronger photoacid generators like triphenylsulfonium triflate helped them shrink features down to well below a micron. That coming together of chemistry and engineering sits at the core of all our modern electronics: smartphones, computers, and beyond. Without this molecule, Moore’s Law might not look the same today.
Coatings and Inks That Harden in a Flash
Beyond electronics, the process has changed the printing and coatings business. UV-curing technology uses these photoinitiators to solidify inks and coatings with a flash of light, locking color and texture onto surfaces almost instantly. Printers run faster, energy costs drop, and the final product looks sharper. I’ve heard packaging manufacturers explain how this shift cut down on solvents and production time.
Dental materials also benefit from this chemistry. Dentists use UV light to harden resins in seconds. Those fillings last longer and are easier for patients to get. Here again, triphenylsulfonium triflate’s role stays mostly invisible but no less important.
Driving Cleaner, Greener Manufacturing
Manufacturers these days worry about energy use and environmental impact more than ever. Replacing heat-curing or solvent-based systems with UV and photoinitiators supports that shift. Lower temperatures, less waste, and faster production means a smaller footprint for every item made.
Of course, challenges remain. Some people in the field flag the toxicity and handling risks involved in using strong acids and sulfonium salts. Good research teams address these problems by improving ventilation, updating protocols, and exploring safer alternatives. The hunt for eco-friendlier substitutes moves forward, but right now, triphenylsulfonium triflate delivers unmatched speed and reliability.
Looking Ahead in Material Science
For me, triphenylsulfonium triflate stands as a reminder that science thrives in the details. Behind every new phone or medical device lies a network of hidden but essential chemicals and processes. We rely on them daily, even if most of us never see their names or shake their makers’ hands. The challenge moving forward is making these technologies safer, smarter, and even better for the world we share.
Triphenylsulfonium triflate brings back memories of long days spent in photolithography labs. The compound’s role in the fabrication of microelectronics means it often ends up on the workbench, usually next to a stack of lab gloves and a bottle of isopropanol. It’s appreciated for its ability to generate acids under UV light, which qualifies it as a strong photoacid generator. Progress in microfabrication leans heavily on these types of chemicals, yet the daily routine often skips past a serious conversation: what actually happens when these substances interact with the people handling them?
Exposure Isn’t Just a Numbers Game
Most researchers know the bare minimum: avoid direct skin contact, wear gloves, don’t eat in the lab. It all sounds sensible until someone spends a late night cleaning up a spill with only latex gloves and the faint hope that nothing soaks through. The biggest concern with triphenylsulfonium triflate lies in its ability to irritate skin, eyes, and respiratory tract. Dust finds its way into awkward places. Breathing problems or skin rashes show up even after “small” incidents. Chronic exposure is the part most overlook because the symptoms build quietly over time. Headaches, coughing, and persistent skin irritation start to take away from the job’s excitement, especially after seeing colleagues deal with those issues firsthand.
Regulatory Oversight, Real-World Gaps
OSHA and other safety agencies include many chemicals in their exposure limits lists, but triphenylsulfonium triflate doesn’t always enjoy such detailed regulation. The lack of explicit exposure limits doesn’t mean it’s safe. The European Chemicals Agency lists this compound as irritating, and the material safety data sheets from reputable suppliers do not sugarcoat its hazards. Working with triphenylsulfonium triflate without a lab coat or goggles means inviting trouble. In places where ventilation systems aren’t up to par, the risk doubles.
Common Sense Trumps Formality
Knowing the technical specs or reading a safety data sheet isn’t enough. My own experience shows that near-misses often traced back to simple shortcuts — working outside the hood or skipping goggles for “just a quick test.” The aftermath usually included frantic washing and monitoring for skin changes. Practicality must win over convenience, especially if you want to avoid the slow creep of chronic symptoms from repeated exposure. Substituting nitrile gloves for latex means fewer breakouts and a better chemical barrier. Double-gloving for spill-prone tasks saved more than one coworker from a nasty day. Washing hands before stepping out of the lab quickly becomes a habit rather than protocol after seeing what lingering residues can do.
Solutions That Actually Work
Effective change starts with layout. Keep a functional fume hood close to where work with photoacid generators happens. Remind folks that personal protective equipment needs regular checks — not just at the start of the semester. Develop spill procedures that take speed and clarity into account, then drill them until they’re muscle memory. Create an environment where colleagues look out for one another, calling out lapses without embarrassment. People learn from each others’ close calls and mistakes more than from a laminated chart on the wall.
Advanced materials work thrives on precision and control. The same approach belongs in safety. Triphenylsulfonium triflate doesn’t forgive shortcuts. Common sense, simple precautions, and real-time communication usually make the difference between a productive lab and a regrettable incident.