Zinc Trifluoroacetate: A New Chapter in Metal-Organic Chemistry
The Roots of Zinc Trifluoroacetate
Zinc trifluoroacetate didn’t pop into existence overnight. Chemists spent years searching for metal salts that could bridge the gap between structural stability and functional creativity. Zinc acetate held the spotlight for a long time. With fluorine’s rise in pharmaceutical and materials chemistry, someone saw potential in swapping the acetates for trifluoroacetates, giving birth to a new class of reagents. The early literature tells a story of methodical synthesizing and careful purification, usually starting with zinc oxide or zinc carbonate and a bit of chemical tenacity. Looking back through journals, I remember stumbling on work describing tricky crystallizations and evaporation steps, all handled by hand — no fancy robotics, no shortcuts. The early researchers probably wore down their fingerprints weighing out reagents, but their persistence paid off.
Getting to Know the Product
Zinc trifluoroacetate comes as a colorless or slightly white crystalline powder. As someone who once spent hours scraping sticky residues out of round-bottom flasks, I appreciate a product that handles as cleanly as this one. The compound dissolves well in water and polar organic solvents, which gives it a leg up in synthesis and formulation laboratories. Heating zinc trifluoroacetate can lead to its breakdown, releasing carbon dioxide and the eye-watering fumes of trifluoroacetic acid, so ventilation matters. Chemists value its strong coordination capability, which opens doors in fields like organometallic chemistry and materials synthesis.
Diving Into the Chemistry
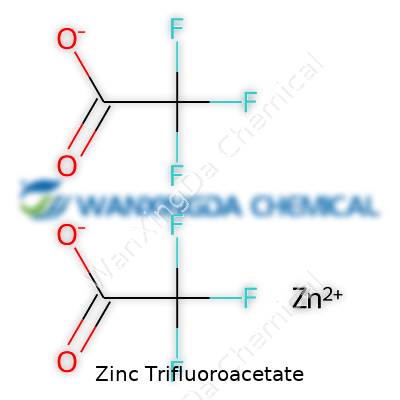
The chemistry community doesn’t hand out gold medals for versatility, but if it did, zinc trifluoroacetate would place high. The trifluoroacetate ligands draw electron density, changing the reactivity compared to its acetate cousins. It coordinates tightly with nitrogen- and oxygen-rich ligands, forming stable complexes used as catalysts or in supramolecular assemblies. I remember trying a palladium-catalyzed cross-coupling reaction that just wouldn’t work with other zinc salts. Swapping in zinc trifluoroacetate moved stubborn intermediates along, likely thanks to its more reactive coordination sphere. The overall molecular formula, Zn(OCOCF3)2, blends the simplicity of a metal salt with the exotic punch of fluorinated organics.
Preparation and Labeling: No Place for Mistakes
Synthesis usually calls for reacting zinc oxide or zinc carbonate with trifluoroacetic acid in a controlled setting. This is a test of patience as much as skill — too much acid, and everything fizzes away, too little, and product yield tanks. The resulting material gets dried under vacuum to remove every trace of volatile acid, which matters for anyone who dreads the smell of strong organic acids in the lab. Accurate labeling tracks lot number, synthesis date, and storage conditions, which helps ensure quality. Regulatory codes require clear hazard statements, since trifluoroacetic acid brings environmental and health concerns. Skipping steps here doesn’t just risk bad batches; it puts users and the environment at risk.
Getting Down to the Properties
Experience tells me that the physical properties of a compound can make or break its value in research and industry. Zinc trifluoroacetate stands out with its solid, crystalline appearance, easy solubility in polar solvents, and fairly high melting point. Chemically, the electron-withdrawing power of fluorine pulls zinc into a different reactivity range. It resists simple oxidation or reduction, which is helpful when working on multi-step syntheses. Exposure to moisture doesn’t destroy it, but it tends to absorb water, which can clump up powders and complicate weighing. The crystal structure, with strong ionic bonds holding zinc and trifluoroacetate together, gives impressive thermal and hydrolytic stability but also means dealing with strong corrosiveness when decomposition sets in.
Chemical Reactions and New Directions
The reactive nature of the trifluoroacetate group leads to interesting chemical transformations. It serves as a leaving group in substitution reactions or helps mediate metal exchange. I’ve seen researchers swap the trifluoroacetate ligands for other organics, using the zinc center as a staging ground for building complex molecular assemblies. In catalysis, zinc trifluoroacetate helps in controlled rearrangements, cross-couplings, and even in forming layered materials with tailored magnetic or conductive properties. Anyone aiming to modify its structure finds a balance between the stubbornness of the zinc-oxygen bond and the lability of the trifluoroacetate groups.
Names and Misnomers
Sometimes names confuse more than clarify. You’ll find zinc trifluoroacetate listed as zinc bis(trifluoroacetate), zinc(II) trifluoroacetate, zinc trifluoroethanoate, or even the shorthand Zn(TFA)2. Mislabeling lurks in online catalogs, so researchers cross-check chemical structures before placing any order. Once, I ordered a product under one synonym and ended up with a completely different salt — a mistake that cost valuable time and materials. Synonyms crop up depending on supplier traditions or local nomenclature, but they point back to one pivotal structure: a zinc atom sandwiched between two trifluoroacetate groups.
Safety Matters: Keeping Risks Front and Center
No chemical deserves a cavalier attitude. Zinc compounds often get labeled as “safe,” but trifluoroacetic acid deserves respect. Contact or inhalation brings irritation, and the persistence of fluorinated compounds worries environmental scientists. Operations require proper gloves, goggles, and fume hoods. Lab managers push for training on spills, disposal, and first aid, because mistakes here can have real consequences. Disposal must follow hazardous waste procedures, because the fluorinated ligand does not break down easily. Precaution beats regret, and anyone working with zinc trifluoroacetate takes those warnings seriously.
Applications Breaking New Ground
The demand for advanced materials and precision chemistry pushes zinc trifluoroacetate into the spotlight. Its role as a catalyst in organic synthesis goes beyond simple salt metathesis; it underpins key steps in pharmaceuticals, agrochemicals, and specialty polymers. Materials science teams use it to form thin films for electronics or as a precursor in the vapor-phase deposition of metal-organic frameworks. In coordination chemistry, its unique ligand allows for fine-tuned electronic properties and the assembly of functionalized nanomaterials. I remember grad students discussing how this compound changed the course of their thesis work, taking a stalled reaction and unlocking a novel structure with unexpected properties. Anyone who’s worked late into the night troubleshooting a synthesis can appreciate the value brought by a reliable, versatile zinc compound.
Pushing the Boundaries: Research and R&D Stories
Research groups around the world scan zinc trifluoroacetate for new applications, using it to build up frameworks, catalysts, and even biomolecular mimics. Early experiments in metal-organic frameworks leaned on the unique electron distribution of the trifluoroacetate group. Organic chemists tweak substituents to test new reactivity, while material scientists push for integration in nanostructures for energy storage or molecular electronics. The R&D pipeline buzzes with talk about its role in green chemistry, as fluorinated ligands can sometimes enable milder, more selective processes. My own reading in synthesis journals turned me into a believer: the case studies and patent filings show that zinc trifluoroacetate holds its place as a tool for modern, innovative chemistry.
The Hard Conversations: Toxicity and Environmental Research
Zinc on its own appears in dietary supplements and industry, but adding fluorinated ligands changes the story. Researchers study both acute and chronic toxicity, with data pointing to low-to-moderate biological uptake but high resistance to degradation. Trifluoroacetic acid itself raises flags for persistence in the environment. I’ve attended conferences where toxicologists poured over study results, showing how even trace fluoro-organic compounds linger in waterways and soils. Regulatory authorities have responded by tightening controls and requiring detailed waste management for products and processes using such reagents. In the lab, anyone who’s rinsed glassware containing these chemicals double-checks that wastes head to designated disposal streams, not down the drain.
A Glimpse Ahead: Zinc Trifluoroacetate’s Future
Zinc trifluoroacetate stands poised to play a bigger role in synthetic chemistry and advanced material design. More researchers are exploring its potential in green catalytic cycles and sustainable processes. Teams work to limit environmental impacts by designing ligands that break down more readily or by recycling zinc partners after use. Regulatory shifts and growing public concern about fluorinated compounds push chemists to innovate on both product and process design. I see research groups eyeing this reagent for use in areas like hydrogen storage, smart materials, and even medical diagnostics. As the conversation in science continues to shift toward sustainability and safety, the story of zinc trifluoroacetate will evolve, shaped by researchers’ dedication, creativity, and concern for the world outside the lab.
Zinc trifluoroacetate doesn't pop up in everyday conversation, but for people working in certain corners of chemistry, it often comes with real, down-to-earth value. Learning about this compound feels a bit like finding a tool most folks overlook—a specialized wrench hidden in the toolbox, oddly shaped but irreplaceable once you figure out its true job. I’ve seen research rooms light up when a stubborn problem finally gives way, and in more than one corner, zinc trifluoroacetate has brought that spark.
A Helper in Organic Synthesis
Organic chemistry sometimes feels like a jigsaw puzzle with infinite pieces. Catalysts help snap those pieces into place, setting the stage for reactions without getting used up or worn out. In this field, zinc trifluoroacetate often steps in. Its knack for encouraging certain reactions—especially those involving carbon-fluorine bonds—helps chemists build complex molecules for pharmaceuticals, agrochemicals, and advanced materials. Real progress in medicine or crop protection depends on finding new, precise ways to assemble molecules, and this compound gives researchers that extra edge.
People ask, “What’s the big deal about trifluoroacetate?” It’s all about control. Fluorine brings stability and reactivity both, and pairing it with zinc opens doors in synthetic work. Sometimes, a scientist’s whole thesis finds its backbone because this compound trims weeks off a project or turns a tricky step into something repeatable.
Clean Reactions, Cleaner Waste
If you’ve spent time in a chemistry lab, you know well how waste piles up fast. Some catalysts make things work, only to leave a mess behind—heavy metals, toxic byproducts, sludge that nobody wants to deal with. Zinc offers less environmental pushback compared to tougher metals like mercury or lead. You don’t just get a reaction; you also face fewer headaches when it comes to cleaning up the byproducts, an issue much on the minds of regulators and environmentalists.
Chemistry often forgets the people sweeping up at the end of the day. Less waste means a safer workplace. Cleaner processes mean less stress about what seeps into the ground or escapes into the air. That slow, steady shift toward more responsible lab practices has a champion in zinc trifluoroacetate.
Room for Further Discovery
For all the things zinc trifluoroacetate does well, innovation doesn’t stay still. New ways to use it spring up as researchers push the limits—looking for faster reactions, greater selectivity, friendlier byproducts. In my years around labs, I’ve noticed the appetite for compounds that don’t just “work,” but solve problems chemists didn’t even know they had. This compound’s unique structure makes it a go-to candidate for new research in catalysis and material science.
Industry and academia both keep an eye on synthetic methods that combine effectiveness, price, and impact. That’s a tough mix, but zinc trifluoroacetate has a track record as a useful building block and a patient helper behind the scenes. If more researchers put these kinds of practical solutions front and center, everybody benefits—from scientists at the bench to the communities living downstream.
Getting to Know the Formula
Zinc trifluoroacetate draws attention in labs and research settings for more reasons than its tongue-twister of a name. Its chemical formula, Zn(CF3COO)2, reveals much about its structure. Breaking it down, you have one zinc ion paired with two trifluoroacetate groups. These groups each combine a carbon backbone with three tightly bound fluorine atoms, showing off the electronegative flair that fluorine always brings.
Its molecular weight sits at about 325.43 grams per mole, which might seem oddly heavy for a compound made from fairly common elements. Those fluorine atoms tip the scale, adding mass and increasing the volatility and reactivity compared to simpler zinc salts. This explains why zinc trifluoroacetate behaves so differently from its close cousins like zinc acetate.
Why the Formula Matters
Zinc compounds find daily use in chemistry. In my own lab days, I found that subtleties in a compound’s structure could make or break an experiment. For zinc trifluoroacetate, the trifluoroacetate group acts as more than just a spectator. It drastically changes the solubility, reactivity, and even the way the zinc center interacts with other molecules or metals. This becomes a big deal in organic synthesis or when making specialized materials, where the tiniest change in reaction partners alters outcomes.
Researchers who look for new catalytic processes often land on zinc trifluoroacetate thanks to its unique mix of properties. Compared to the usual go-to reagents, it offers new reactivity. A trifluoromethyl group makes anything it touches harder to reduce, harder to oxidize, and a lot less willing to play nice with water. So, the formula isn’t just trivia, but a key to why this compound works as it does.
Safety and Environmental Notes
Handling trifluorinated chemicals brings extra responsibility. The same features that make them valuable in synthesis can cause headaches for waste disposal. Trifluoroacetate groups resist breakdown, persisting through typical treatment systems. In my own experience, waste management and careful planning mattered as much as the reaction itself. Labs and industry both have to consider alternatives or better ways to contain and recycle what’s left after experiments finish.
Where Science Goes Next
Innovation doesn’t sit still. Researchers and graduate students spend late nights looking into greener routes and alternatives to zinc trifluoroacetate, hoping to capture its benefits without the baggage. The unique mix of reactivity, volatility, and stability keeps this compound popular, especially for chemists pushing the frontiers in fluorination or advanced materials.
Anyone working regularly with this compound needs more than a formula scribbled on paper. They need practical understanding, real-life experience, and access to up-to-date data. Plain facts and common sense matter as much as high-tech instrumentation. Chemistry is a hands-on science—knowing your materials, not just their numbers, helps you innovate while keeping a careful eye on safety and sustainability.
I remember the first time I opened a small bottle of zinc trifluoroacetate in the lab, I felt a mix of curiosity and responsibility. Chemical safety isn’t about following a checklist for its own sake—it’s often about learning from people who’ve worked with different compounds before and understanding those lessons matter even for experienced hands.
Simple Choices Can Avoid Big Problems
Zinc trifluoroacetate isn’t flashy, but it asks for respect. This salt finds its way into organic synthesis, specialized coatings, and even pharmaceutical research. It picks up moisture from the air far more eagerly than most everyday chemicals, so giving it a dry, snug home isn’t just a suggestion. Storing containers in a desiccator or a tightly sealed jar works better than most fancier options I’ve tried. Exposing it to room air for more than a few minutes leaves you with clumpy waste, and that’s an expensive way to ruin a batch. Dry cabinets are worth their shelf space, especially if you’re storing a few grams for weeks or months.
I learned early to label everything clearly—date received, who opened it, and any hint that the powder has changed texture. These extra notes have saved many people from accidental missteps. Fresh zinc trifluoroacetate has a distinct, sharp odor. A dull or sour smell can mean the compound has absorbed water or decomposed.
Think About Heat, Light, and Reactions
Zinc trifluoroacetate won’t burst into flames at room temperature, but ignoring its appetite for moisture gets risky. Heat speeds up decomposition, especially if you leave the cap loose on a warm day. I always store it away from light sources, because chemical changes often creep in faster with exposure. Keeping it in a lower cabinet, away from direct sunlight and any heat-producing equipment, has kept my material cleaner longer than alternative setups.
Combining it with strong bases or acids without proper planning tends to generate more than the desired reaction. I’ve seen test tubes bubble and smell sharp because someone rushed past the safety notes. Even simple gloves and goggles are more than decoration here, since trifluoroacetates can irritate skin and eyes. It makes sense to keep water nearby to quickly address any spills.
People Matter More Than Policies
Good ventilation in a storage space helps everyone, not just at the benchtop. I’ve worked in places where chemical odors drifted around because ventilation wasn’t checked regularly. Closing a container tightly, checking for cracks, and keeping bottles stored away from incompatible materials—these small habits cut down risks far more than posters on the wall. The best labs I’ve worked in ran monthly cleanouts, where people shared stories of what almost went wrong. These moments built a culture of safety that no written protocol could match.
For disposal, following established hazardous waste guidelines keeps you and your coworkers out of trouble. Many university and industrial facilities have on-site hazardous waste pickups—don’t pour it down the drain. If you’re not sure, someone else has likely faced the same question, so ask. In my experience, a few cautious questions can keep an entire lab safe.
The Value of Experience and Vigilance
Reading the fine print goes hand in hand with checking in with colleagues and other chemists. I rely on trusted chemical safety resources and databases, and still learn something new every year. Safety with zinc trifluoroacetate means treating it with the same care you’d want if you were the next person reaching for that container. Respect for the material, and the people around you, benefits everyone down the line.
Everyday Hazards in a Specialized Chemical
Zinc trifluoroacetate isn't a chemical most folks keep at home, but it pops up in certain research settings and industry labs. The stuff packs that sharp, sour smell familiar to those who've worked with other fluoroacetate compounds. As someone who’s spent time around chemical stores and seen plenty of safety training slides, I can tell you: strange-sounding chemicals always draw wary glances, and for good reason.
Toxicity: What the Science Shows
Digging into the literature, the data on zinc trifluoroacetate is thin compared to better-known substances. But here’s what jumps out. Trifluoroacetates belong to a group tied to corrosiveness and metabolic disruption. They stem from trifluoroacetic acid, a material notorious for strong acidity and persistent residue. Touching the pure acid irritates skin and eyes, and its vapor stings the lungs. Swapping in zinc changes some properties but not the general rule: caution wins the day.
Studies show zinc as an element supports growth in trace amounts but gets risky beyond that. High doses harm the stomach, provoke vomiting, and might meddle with copper absorption in the body. In zinc trifluoroacetate, you get both the metal ion and the aggressive trifluoroacetate group, raising red flags for toxicity and environmental impact if it spills in running water or soil. There’s evidence similar compounds can disrupt aquatic organisms and linger in groundwater, which spells serious trouble outside a lab.
How to Work with It Safely
Veteran lab workers never downplay chemical hazards just because material isn’t famous. I’ve watched new researchers skip gloves or ignore fume hoods, chalking up their caution to “overkill.” Soon enough, they realize chemical safety rules build up for good reason, not just paperwork. The right gloves, splash-proof goggles, and good ventilation aren’t extras—they’re basics. Many fluoroacetate compounds give off fumes that stick to the throat and eyes, so a quality fume hood matters for more than just comfort.
Regulation and Responsible Use
No big regulatory agencies, like the EPA or OSHA, list zinc trifluoroacetate among their most wanted toxins, mainly because its industrial uses stay limited. That doesn’t mean it’s safe to treat it lightly. Chemical waste disposal rules make it clear: trifluoro compounds need special handling and can’t go down the drain. Responsible users put spent materials in dedicated waste streams for fluorinated organics, and treat everything as potentially hazardous to the environment. Taking care with these details shows respect for both coworkers and the neighborhoods beyond the lab walls.
Finding a Smarter Path Forward
In every teaching lab and workplace, the culture around safety starts with honest discussions. Training shouldn’t feel like a dry recap of rules or just a checklist. Stories matter: folks remember hearing about real injuries or near-misses more than they remember warnings written down on a safety sheet. Strong leadership and open talk about hazards set the tone for everyone coming through the door.
The bottom line with zinc trifluoroacetate: nobody benefits by downplaying real risks tied to chemistry’s less familiar corners. Being aware, staying careful, and asking questions beats working in the dark—every single time.
More than Just a Zinc Salt
Zinc trifluoroacetate doesn’t make headlines the way lithium batteries or printed electronics do. Still, folks in a lab coat know it by smell, use, and even reputation. Years in the lab have shown me that plenty of progress in chemistry and materials science leans on reagents that never show up in glossy magazines or on the evening news. Here’s where this underrated compound tells a bigger story about how research works today.
The Real Strength: Organofluorine Chemistry
Those who have spent time working with fluorinated molecules recognize zinc trifluoroacetate as a go-to starting material. The trifluoroacetate group gives it a unique ability to transfer zinc into reactions where water and oxygen tend to make a mess of things. I’ve found it especially helpful in synthesizing specialty organofluorine compounds, the class that powers everything from some pharmaceuticals to advanced polymers designed to withstand the worst that solvents and weather can throw at them.
Catalysis and Coordination Chemistry
Researchers pursue simple answers by running countless small reactions. Catalysts play a big role here. Zinc trifluoroacetate often enters the fray as a mild Lewis acid. It helps gently coax molecules into new configurations without tearing them apart. Journal articles have shown its use in cyclization reactions, where the backbone of a drug or polymer is bent into shape by coordination to the zinc ion. This kind of control is something I struggled to get just right using harsher zinc or copper salts.
Tech That Turns Out to Matter
Looking outside synthetic chemistry, zinc trifluoroacetate has uses where clean delivery of zinc is key. Semiconductors and optoelectronics benefit, as it brings together high zinc content and organic compatibility. Colleagues in thin film deposition often tell me that this compound’s volatility and solubility make it work like few others. I have seen research gear up to create transparent conducting oxides using this salt as a starting source. Such coatings sprinkle across touchscreens, solar cells, and LED tech.
Pharmaceuticals and Material Development
Drug design keeps witnessing a push for new ways to build molecules that biological systems don’t already break down. Fluorine atoms, including those in the trifluoroacetate group, play a role in boosting drug stability and tweaking how a compound interacts in the body. Zinc trifluoroacetate helps introduce these groups or acts as a gentle zinc donor in processes that protect delicate, reactive samples from decomposing. Its reliability in producing high-purity intermediates speaks to the effort chemists put in to control every step.
The Catch: Hazards, Costs, and Solutions
Regular lab users also recognize the headaches: trifluoroacetate isn’t environmentally benign, and disposal needs good oversight. Large-scale use in industry gets tangled with fluorine waste rules, which add time and cost. I’ve seen efforts within university and industrial labs to switch to greener processes or develop better recycling methods for fluorinated byproducts. Alternative zinc compounds sometimes step in, but the loss in selectivity or ease of handling raises its own costs.
Where We Go Next
Zinc trifluoroacetate keeps a seat in the chemistry world not because it’s perfect, but because it brings a rare mix of reactivity, selectivity, and compatibility. Its applications spark advances in diagnostics, electronics, and pharmaceuticals. Even as more labs hunt for greener replacements, the lessons learned from compounds like this one push us to keep building safer and smarter chemistry. The hope is that future research can keep the benefits, while giving up the environmental costs that have trailed these chemicals for decades.