Zinc Trifluoroacetate Hydrate: A Closer Look at a Modern Chemical Tool
Historical Development
Zinc chemistry has always played a steady role in both industry and research, and trifluoroacetates mark a peculiar turn in that story. Zinc’s early use traces back centuries, yet the fine-tuning of its coordination compounds—like zinc trifluoroacetate hydrate—reflects just how far chemical synthesis has come. Throughout the 20th century, the development of fluorinated organics brought new possibilities: improved solubility in nonpolar solvents, interesting reactivity, and fresh options for molecular design. Zinc trifluoroacetate hydrate did not grab headlines overnight. It grew out of persistent, hands-on work in academic labs, with each iteration spurred by real needs—faster catalysis, better solubility, or gentler reaction conditions. Unlike basic zinc salts, the trifluoroacetate version now lives at the boundaries of organic methodology, thin-film technology, and even niche medical research.
Product Overview
Zinc trifluoroacetate hydrate stands out as an off-white crystalline powder. It owes its particular niche to the trifluoroacetate anion. The fluorines don’t just change the look; they flip conventional zinc chemistry into new territory. Where straight zinc acetate works well in aqueous systems, the trifluoroacetate version brings a talent for dissolving in organic solvents and handling hydrophobic mixtures. The water of hydration stabilizes the salt, making handling a little less tricky than fully anhydrous versions. In the lab, you notice these small details: how easily it scoops, how well it dissolves in your chosen solvent, and how stable it stays even after exposure to air for a short time. Its chemical footprint feels both familiar and unusual, especially for teams used to standard acetates.
Physical & Chemical Properties
Zinc trifluoroacetate hydrate shows a solid, crystalline appearance with reliable stability under ordinary storage conditions. With a moderate melting point and a tendency to absorb moisture from the air, it reminds anyone working at the bench what “hygroscopic” really means. The triple-fluorinated carboxylate groups add notable electron-withdrawing effects—this boosts reactivity but also calls for a bit more care in waste management. It’s not especially volatile, but the chemical still deserves respect for both its acidity and its affinity for organic reactions. Solubility remains an attractive feature: alcohols, esters, and even some less polar solvents all accept this compound where classic zinc salts fall short. This opens doors that tin-based fluorinated salts used to control alone.
Technical Specifications & Labeling
Most commercial batches arrive with a guaranteed purity above 98%, water content clearly listed, and the trifluoroacetate load matched with the expected stoichiometry. One expects labeling to note the hydrate form directly; shelf life and storage instructions are given, too, because shelf-stable does not mean immortal. From a technical standpoint, chemical suppliers recognize that buyers favor batches traceable back to their manufacturing run, with COAs available to back up listed metals content and organic purity. Many in the lab world know that specifications serve as more than just box-checking. They guide safe handling, and they prevent mysterious side reactions that ruin days of work. Every bottle carries these promises, baked into its identity by a discipline that learns from past errors.
Preparation Method
The production of zinc trifluoroacetate hydrate looks straightforward on paper, but the best methods rely on clean, efficient chemistry. Zinc oxide or basic zinc carbonate meets trifluoroacetic acid in a controlled exothermic reaction, with plenty of attention paid to removing excess acid and ensuring the right hydration. Filtering and gentle evaporation coax the product from solution, but technique carries the day. Impurities slip in when haste wins over patience—I've seen, at times, how minor contamination with non-fluorinated organics can defeat the whole point of the trifluoroacetate. Curiosity in greener syntheses has grown, with teams exploring milder acids or solvent-less approaches to cut waste. The discipline here is not just tradition—it reflects a desire to make chemistry cleaner and more precise without sacrificing quality.
Chemical Reactions & Modifications
Anyone who’s ever tried cross-coupling reactions or worked with organofluorines appreciates the versatility zinc trifluoroacetate hydrate brings to the table. Its reactivity, sparked by the electron-poor trifluoroacetate, can fine-tune metal–organic frameworks, assist as a catalyst in select coupling reactions, or serve as a mediator in polymerizations. Researchers find it useful for introducing fluorine moieties into complex molecules, thanks to its compatibility with sensitive substrates. The salt’s potential for some in-situ modifications has opened up even richer chemistry, such as the exchange of ligands or creating mixed-metal systems. Down at the benchtop, it becomes a bridge—uniting classic zinc reactivity with the unique electron-withdrawing bite of fluorine, without calling down the hazards that pure fluorinated acids or anhydrides bring.
Synonyms & Product Names
Zinc trifluoroacetate hydrate travels by a handful of names depending on context. Some catalogues call it “zinc(II) trifluoroacetate hydrate,” others abbreviate to “Zn(TFA)2·xH2O”, and some old-timers in the lab refer to it as just “zinc TFA”. These names share more than semantics—they point to a shared understanding of the chemistry. No fancy branding or trademarking here—just plain language shaped for practical use.
Safety & Operational Standards
Chemists don’t forget safety because the lessons are written in scars, not just on labels. Zinc trifluoroacetate hydrate doesn’t pack the toxicity punch some organofluorines do, but it does need thoughtful handling. Dust can irritate eyes and mucous membranes. Gloves, goggles, and a fume hood add more than a sense of professionalism; they cut down on accidents. In my experience, those careful with their workspace—clean spills quickly, store containers closed, keep incompatible materials away—avoid the headaches that come from complacency. Waste disposal tracks with concerns about fluorinated residues. Nobody wants these winding up in the general waste stream, with growing scrutiny in the lab about how perfluorinated compounds travel and linger in the environment. For this reason, responsible protocols must become as familiar as basic glassware.
Application Area
Academic and industrial labs have taken up zinc trifluoroacetate hydrate for a surprisingly wide range of projects. It gets a starring role in organic synthesis as a mild Lewis acid, prized for reactions too delicate for harsher catalysts. Material scientists work it into sol-gel processes for transparent conductive films—a field hungry for alternatives to tin and indium in electronics. Biochemists test it as a metal source for enzyme modeling or metalloprotein work, hunting for subtle differences in how fluorinated ligands affect protein–metal interactions. There’s talk in the world of medicinal chemistry, too—finding safer, more robust ways to introduce fluorine into drug candidates, chasing the holy grail of metabolic stability without the byproducts that dog so many synthetic routes. These uses reflect both pragmatism and genuine curiosity; each new project extends its reach a little further.
Research & Development
Bench scientists recognize that the field remains wide open. Novel catalysis, materials for flexible electronics, and even greener organofluorine chemistry pull zinc trifluoroacetate hydrate into new research proposals every year. There’s cutting-edge work around single-source precursors for metal–organic frameworks and advanced battery electrolytes. In meetings, colleagues debate which new combinations offer the best performance—should they stick with tried-and-true protocols or roll the dice on hybrid zinc systems that balance reactivity and sustainability? Careful documentation, peer-reviewed studies, and reproducibility mean everything in this space. Mistakes do not vanish; they linger in the corners of every published method, reminding everyone that real progress comes from rigorous, honest work, not just headline-chasing or chasing the latest trend.
Toxicity Research
Toxicologists have learned not to get complacent with any compound that mixes heavy metals and fluorine. While zinc remains a necessary trace element, overexposure to organic zinc derivatives creates real health concerns: gastrointestinal distress, irritation, and longer-term bioaccumulation issues. Trifluoroacetate brings the added worry of persistent organic pollutants. I’ve seen ongoing work on how these salts break down—what products form, where they linger in soil and water, and how bioavailable they stay to plants and animals. Data so far underscores the need for stricter waste management, but there’s no sign that zinc trifluoroacetate hydrate belongs in the same risk tier as heavier, more bio-persistent fluorinated organics. Still, the combination of vigilance and updated MSDS reviews serves everyone from researcher to janitor.
Future Prospects
Industry and research share a hope that zinc trifluoroacetate hydrate stands to solve problems old and new. As regulatory pressures shift toward cleaner, less persistent chemicals, this compound occupies a middle ground—powerful enough for cutting-edge science, familiar enough to trace, and potentially friendlier by design than heavier fluorinated alternatives. Upcoming years hold promise for further green chemistry improvements: less solvent waste, modular syntheses, and new downstream products in pharmaceuticals and materials. Students enter the field with sharper awareness of environmental stewardship and a willingness to pressure companies to innovate right alongside academic labs. If all goes well and the chemical community keeps pushing for both robust science and real-world safety, zinc trifluoroacetate hydrate will keep its niche—expanding not because it sounds exotic, but because it earns its keep experiment by experiment.
In a lab, the tools you pick shape your day. Zinc trifluoroacetate hydrate looks intimidating, but it’s quietly important in the work of chemists and researchers. The compound pops up most often in the stories of synthetic chemistry. I remember watching a colleague use it while puzzling through a reaction that needed a gentle, reliable nudge — not a hammer, but a subtle push. She chose zinc trifluoroacetate hydrate to help shift the odds, turning a stubborn chemical mix into something new and useful.
At the heart of its usefulness is its role as a zinc source. In my own experience, getting pure zinc into a reaction isn’t always simple. Zinc powder, zinc chloride — each lands differently in a flask. Trifluoroacetate as a partner brings a twist: the trifluoro groups do more than just balance charges; they help dissolve stubbornly insoluble chemicals, and they don’t bring in much baggage for the next steps in a reaction. That makes the hydrate version handy, especially when you don’t want water-sensitive drama.
Researchers have leaned on this compound in making new organic molecules, especially those that play by tricky rules. For example, organic chemists use it to install fluorine atoms in certain pharmaceutical building blocks. The world of medicine loves a smartly placed fluorine, since it adds stability and helps new drugs last longer in the body. Beyond fluorination, zinc trifluoroacetate hydrate serves as a mild Lewis acid. That means it can gently coax electrons to take new paths, forming or breaking bonds in ways more aggressive catalysts would ruin.
Universities and research labs invest plenty of time and money into finding better catalysts and reagents for the drug pipeline. I’ve seen professors push for new zinc-based compounds because they often work under milder conditions and produce fewer harmful byproducts. Industrial labs notice as well. Cleaner chemistry saves on waste disposal and keeps projects on the right side of environmental regulations, which isn’t just good citizenship — it’s just plain practical.
The future may see more from zinc trifluoroacetate hydrate as green chemistry becomes the standard, not the exception. Sourcing pure chemicals and minimizing waste usually head up the checklist of lab managers, especially as research gets more complex. Companies and universities can work together, sharing what works and what flops. In my view, outreach between academia and industry helps spread best practices, like choosing a gentler zinc source that’s also effective.
One persistent challenge with such specialized compounds is access and cost. Not every lab has the budget for a cabinet full of options. Academic groups often stretch funds, making tough decisions about which reagents to keep on hand. Sharing resources with nearby labs, or pooling orders, drops the price. Open communication about what actually works — through conferences, preprints, and informal chats — keeps the field honest and helps young researchers avoid wasted effort.
Like many specialty reagents, zinc trifluoroacetate hydrate makes a bigger difference than its closet-sized footprint would suggest. Beneath the mouthful of a name, it's another example of how the right molecule, at the right moment, can push science a few steps forward without much noise.
Living with a Reactive Chemical
Anyone who’s ever cracked open a new reagent bottle in a college lab knows the quiet anxiety of keeping things uncontaminated. Some compounds shrug off the odds, but others like Zinc Trifluoroacetate Hydrate set higher expectations. One wrong move, and you find your research sidetracked or lab safety compromised.
Sensitive Chemistry, Real-World Lessons
Zinc Trifluoroacetate Hydrate demands respect for a simple reason: it doesn’t like moisture. This isn’t some textbook warning. I recall working with a cousin compound as a grad student. Ignore the loose cap on a humid day, and the next time you weigh out a sample, it’s a clumpy mess. Water gets in fast and changes what you’re working with. This isn’t just annoying—it throws off your percent yield, disrupts crystal formation, and can send an entire week’s work out the window.
Beyond moisture, exposure to atmospheric carbon dioxide becomes a stealthy problem. Tons of chemicals plod along just fine in the open. Zinc Trifluoroacetate Hydrate doesn’t. It starts to break down and leave behind unwanted byproducts. For research chemists or anyone using this chemical in synthesis, purity is everything.
Storage Practices: It’s More Than Just a Cabinet
Let’s get specific—what helps? Airtight containers matter. A heavy screw-cap glass bottle holds up better than a plastic tub. Every seasoned bench scientist I know keeps a stash of desiccators and silica gel packs. It’s not about playing it safe; it’s about keeping reactivity in check so experiments return usable answers.
Temperature control is just as important. Uncontrolled warmth disturbs the hydrate’s structure and brings on caking or color changes. Room temperature works for short stretches, but for the long game, a cool, dark storage spot builds confidence. Forgetting this means buying new chemical even if the old container isn’t technically “expired.”
Labeling and Tracking—Not Just for Show
It’s easy to treat hazard labels and batch logs as bureaucratic hurdles. Over the years, though, I’ve seen mix-ups turn minor headaches into lab-wide scrambles. If Zinc Trifluoroacetate Hydrate moves between students or projects, proper labeling prevents unwanted reactions and keeps contamination at bay. That record-keeping also helps when regulators swing by or if something ever goes wrong.
Practical Solutions from Day One
Small tweaks keep risks down. I make sure to return chemicals to their original containers, using clean utensils every time. Swapping powder between bottles with reused spatulas can seed contamination that grows invisible at first, then roars to life on the day of a crucial test. Keeping chemicals in limited access areas stops casual traffic and respects the seriousness of the material.
Training matters, too. I learned early not to take a veteran’s grumbling about “fussy” chemicals as mere venting. Years later, I insist on running new staff through storage drills with compounds like this, showing them what a hydrated salt feels like in the hand, and what a compromised one looks like. It isn’t overkill. It builds habits that lower risks.
The Broader Picture
Storing Zinc Trifluoroacetate Hydrate goes beyond simple compliance. Every careful step helps safeguard experiments, protects colleagues, and keeps research budgets from ballooning with avoidable replacements. The details matter. Sturdy bottles, dry cabinets, careful handling, and lots of respect for the compound’s quirks add up to safe, reliable science.
Understanding the Formula: A Matter of Precision
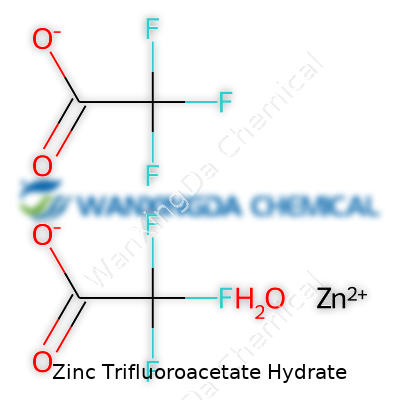
Zinc trifluoroacetate hydrate brings a certain curiosity in any chemistry lab. Anybody working with fluorinated reagents or complex zinc salts knows why: trifluoroacetate stands out for its electron-withdrawing power and stability. The chemical formula for the basic, anhydrous form of zinc trifluoroacetate is Zn(CF3COO)2, but science rarely stays that simple indoors. The common laboratory version almost always comes with water molecules attached, leading to zinc trifluoroacetate hydrate. The most encountered hydrate in labs, at least in published literature, is the tetrahydrate: Zn(CF3COO)2·4H2O. This detail matters since extra water in the compound changes the actual amount of zinc and trifluoroacetate you end up using, which throws off calculations if you miss it.
Molecular Weight: How Numbers Matter in Real Reactions
Skeptical eyes in any chemistry group will calculate the molecular weight more than once. Anhydrous zinc trifluoroacetate measures in at about 317.5 g/mol. Add the four water molecules in the tetrahydrate, and the total weight rises to roughly 389.5 g/mol. Skipping over these details while making up a reaction mixture leads to lower yields or skewed data, so clear knowledge saves more than just time—it preserves the integrity of an entire experiment.
Zinc Trifluoroacetate Hydrate’s Significance in Research
Some people might wonder, "Why the fuss over one chemical’s formula and weight?" It connects to something fundamental: in chemical synthesis, small errors at the base level multiply into bigger ones during scale-up or repeat runs. I recall the look on my mentor’s face when a talented but distracted grad student ignored that “hydrate” in the recipe, using the wrong molecular weight for a catalyst preparation. The whole week’s progress evaporated because the reaction never reached the temperature or concentration needed to proceed.
Researchers who work on organic fluorine chemistry, advanced materials, or even new battery technologies encounter compounds like this—not just as academic curiosities but as building blocks and catalysts. Zinc without the right counter-ions can’t coordinate as desired, and the trifluoroacetate group adds something unique by pulling electron density and changing reactivity. Water molecules, often seen as impurities, do more than pad the weight: they stabilize the salt and sometimes serve a role in crystal structure or solubility. That’s not a side note; it shapes the whole workflow, from storage to solvent choices to the details of crystallography.
Improving Clarity in Chemical Communication
Some labs still gloss over details, listing "zinc trifluoroacetate" in procedures without clarifying the hydrate level. Open disclosure should become the norm, not an afterthought. Accurate labeling in published protocols and clear communication from suppliers cuts confusion. Sharing knowledge from your own mishaps or lessons learned makes peer review more meaningful and chemistry more reliable.
Moving forward, investing a little extra care upfront—double-checking the hydrate form, recalculating weights, and clearly labeling compounds—avoids wasted resources and prevents cascading failures. These small steps shape not just the success of a synthesis but also the reputation and integrity of the entire scientific community.
Understanding the Compound
Zinc trifluoroacetate hydrate isn’t something you’d stumble across on a grocery shelf. This chemical pops up more among scientists and industry folks, used in labs for special chemical reactions and as a catalyst for certain syntheses. What sets it apart is the trifluoroacetate group, which introduces fluorine atoms—a detail that always catches the eyes of chemists and toxicologists, since certain fluorinated compounds can pack a punch both chemically and biologically.
The Hazards Beneath the Surface
Anyone handling zinc trifluoroacetate hydrate must treat it with real care. Breathing in its dust or vapor can irritate the nose, throat, and lungs. Get it on your skin, and you may see redness or a rash. Eyes react badly to dust or splashes, potentially leading to burning or stinging that lingers. Ingesting it isn't smart, as it could upset your stomach or even provoke systemic effects if enough gets absorbed.
Trifluoroacetate shows up in scientific discussions due to its stability and reactivity. Populating certain pharmaceuticals, fluorinated chemicals can hang around in the environment longer than many other compounds. That slow breakdown poses a new set of problems for workers and the planet. Synthetic molecules that resist natural breakdown accumulate over time. Using them in a tightly-controlled lab setting reduces risks, but spills and improper disposal multiply dangers, both to workers and to communities downstream.
Damage Isn’t Just Theoretical
Look at reports of fluorinated substances in groundwater. Regions with heavy industry or chemical manufacturing have faced trouble from “forever chemicals,” including PFAS, which share those tightly-bound fluorine atoms. Though zinc trifluoroacetate hydrate hasn’t triggered public health crises as large as some other fluorinated compounds, its structure fits the general pattern of chemicals that require close oversight. Years ago, few thought common lab chemicals would end up dogging water supplies and health systems. Yet here we are, uncovering health connections tied to these persistent, bioaccumulative molecules.
Research Gaps and Worker Safety
A frustrating reality surrounds many specialty chemicals: the research spotlight shines bright on major contaminants, yet leaves those like zinc trifluoroacetate hydrate in the shadows. The available hazard data usually describes irritation potential and points toward risk when handled sloppily, but long-term, low-level exposure studies are rare. That creates uncertainty. Most employers, faced with uncertainty, lean on best practices drawn from related substances—gloves, goggles, fume hoods, training that emphasizes good habits and quick spill cleanup.
Over the years, I’ve spent time in teaching labs and chemical stockrooms. Each new material brings a script: respect it, stay alert, and clean up after yourself. Guidance about fluorinated materials has grown stricter as the science has advanced. I learned early on that companies and universities often update their safety rules to keep pace. It’s far easier to stay safe in a culture that expects questions about every bottle on the shelf than in those that treat unlabeled jars as afterthoughts.
Paths Toward Safer Practice
The solution isn’t complicated, but it does require a bit of discipline. Stricter labeling, clear instructions, accessible training, and proper disposal keep these chemicals from turning into headlines. Companies should conduct regular audits of both their stockrooms and their policies. Transparency about what goes out the door prevents headaches later for both people and ecosystems. Where possible, seeking alternatives that do the same job without the stack of risks makes a real difference, both for folks on shift and for everyone else who calls the area home.
Chemistry labs intimidate plenty of people, and with good reason. Complex names lead many to underestimate risk, but some compounds carry hazards that stick around long after the beakers get scrubbed. Zinc trifluoroacetate hydrate belongs to this club. People might see “zinc” and think supplements or sunscreen, but toss in “trifluoroacetate” and the safety calculus changes. Handling this chemical safely demands attention, not routine.
The Real Hazards: Beyond the Label
Trifluoroacetates aren’t run-of-the-mill laboratory salts. Fluorinated compounds bring persistence and reactivity. I’ve seen chemists with decades under their belt stop and double-check their gloves and goggles. Zinc trifluoroacetate hydrate doesn’t ask for respect; it requires it.
Skin contact is not just unpleasant — it risks burns and systemic toxicity. Vapors or dust carry threats to lungs and eyes. If a spill happens, you want that lab coat and face shield on, not hanging on the back of a chair.
No Room for Sloppy Storage
Storing this compound in the open, where ambient moisture or heat acts unchecked, lands chemicals where you don’t want them: in the air, on surfaces, with people who haven’t reviewed the latest hazard data. Sealed containers, dry environments, clear labeling, and secured shelves don’t just look good in inspection reports — they keep accidents (and exposures) out of daily operations.
Personal experience says: label everything, store away from acids (to avoid gas release), and keep incompatible chemicals at a safe distance. Too many stories circulate of near-misses due to careless habits and outdated systems.
Waste Isn’t Garbage
Once a reaction wraps up, no bottle of zinc trifluoroacetate hydrate becomes just “trash.” Pouring anything related down the drain or tossing in general garbage equals pollution. Trifluoroacetates have a stubborn nature. They linger in water and soil, and the effects don’t just disappear. I’ve watched lab managers flag improper disposal as both a personal and community risk.
The EPA and similar agencies frown on dumping fluorinated compounds — fines and liability aren’t theoretical. Bagging contaminated material, using clearly designated hazardous waste containers, and documenting transfer for hazardous waste pickup stops these chemicals from slipping back into the environment. Waste contractors handle the destruction process, using high-temperature incineration or chemical methods that neutralize toxicity.
Training Isn’t Fluff
People sometimes grumble about safety talks and annual refreshers. My own training included real-world case studies: what it meant for a lab to misuse chemicals, the lifelong health consequences when someone skipped a safety step. The best-run labs run drills, inspect PPE stock, and expect questions from newcomers. Shortcuts and guesses lead to misery.
Safe procedures protect workers and communities. A forgotten hazard today can end up in the news years later, long after the original users leave. Handling zinc trifluoroacetate hydrate safely isn’t just about ticking boxes. It’s about recognizing the power these compounds hold and refusing to risk people and the planet.
Moving Forward: Vigilance and Accountability
The most effective solution stays stubbornly simple: treat every session with this chemical as a new one, double-check equipment, follow institutional protocols, and ensure waste never mingles with regular trash. Only then does chemistry become not just a pursuit of new discoveries but a responsibility to those who share your workspaces, your water sources, and your neighborhood. Training, documentation, and proper waste streams — these habits build trust and safety, not just for your lab, but for everyone downwind and downstream.