Zinc Trifluoromethanesulfinate: Bridging Chemistry and Practical Application
Historical Development
Zinc trifluoromethanesulfinate has come a long way since the early days of organofluorine chemistry. Decades ago, researchers pushed boundaries to create reagents capable of installing fluorinated groups cleanly and reliably. The drive came from a real need for more stable, scalable methods in pharmaceutical synthesis, as many early approaches struggled with harsh conditions and unpredictable yields. For chemistry labs, especially those venturing into medicinal compounds or imaging agents, the rise of reagents like this one, often called the Langlois reagent, opened up new opportunities. Over the years, small breakthroughs—fine-tuning syntheses, figuring out storage solutions, tweaking procedures to avoid rapid decomposition—have shaped this compound’s presence in the lab toolkits of both academic researchers and industry chemists.
Product Overview
In practice, zinc trifluoromethanesulfinate packs a punch. It’s not the sort of chemical you find in every science classroom—this white or off-white powder shows up more often in advanced settings where chemists want to add a trifluoromethyl group to their molecules. That trifluoromethyl group, tough to handle on its own, brings significant value across drug discovery, agrochemicals, and imaging science. When someone mentions “trifluoromethylation,” this is one of the names that shows up on reaction schemes. Since its introduction to mainstream organic chemistry, the reagent has saved time and unlocked transformations that stumped earlier generations. For my own lab experience, ordering it for a new cross-coupling reaction always felt like exploring a new route in an old city—familiar tools, but a different destination.
Physical and Chemical Properties
Zinc trifluoromethanesulfinate is best recognized for its solid form, usually as a white crystalline powder. It packs a decent shelf-life if stored away from moisture, since water or humid air can degrade it over time. The compound’s solubility profile leans toward polar aprotic solvents, like DMF or DMSO, which often show up in reaction protocols. Thermal stability runs high enough for most bench-top reactions, so daily handling doesn’t demand elaborate precautions. The substance’s chemical backbone features both zinc and trifluoromethylsulfonyl motifs, lending it enough reactivity to play nicely in radical transformations. That stability and reactivity balance is the real trick, and experienced chemists appreciate having a compound that doesn’t require elaborate babysitting or expensive cold storage.
Technical Specifications & Labeling
Most bottles arrive with clear hazard warning symbols, signal words, and, of course, purity ratings. Depending on the supplier, typical grades hover above 95 percent, enough for research and early process development. I’ve seen labels emphasize the need for dry storage and appropriate ventilation during use—reminders that, in chemistry, trust but verify applies double. Instructions highlight personal protection, as fine powders can disperse easily, so solid-phase reactions or careful wet transfers help avoid mess and loss. Though a glove box isn’t essential, a dry atmosphere certainly prolongs the shelf life. Catalog numbers and batch details help with traceability, which keeps quality control front and center for regulated industry settings aiming for reproducibility and audit readiness.
Preparation Method
Most published syntheses of zinc trifluoromethanesulfinate involve reacting zinc dust or powder with trifluoromethanesulfonyl chloride in the presence of a base, usually in anhydrous solvent. Yields can swing depending on the precise stoichiometry and purity of reagents, so most labs run a test batch before scaling up. I’ve found the exothermic nature of the reaction, especially with large charges of sulfonyl chloride, demands patience—slow addition and active cooling go a long way to avoiding runaway reactions. Filtration and recrystallization steps allow purification, with storage under inert gas, like nitrogen, extending usability. Keeping procedures straightforward helps training new lab members, while routine batch quality checks reinforce consistency for those developing protocols for industry or regulatory review.
Chemical Reactions & Modifications
Zinc trifluoromethanesulfinate brings a radical edge to the trifluoromethylation game. It stands out as a solid, easy-to-handle source of the trifluoromethyl group, thriving in reductive and oxidative settings alike. Chemists rely on it to drive transformations with aryl halides, sulfonyl derivatives, or even more exotic scaffolds. In pharmaceutical settings, the compound handles a host of copper-mediated couplings, cleanly installing –CF3 on heterocycles or aromatic rings—steps that often land new compounds on patent applications. Modification with alternative metal salts shifts the reagent’s solubility or reactivity profile, customizing processes for specific starting materials. In my experience, reading the literature uncovers a lively debate about optimal catalysts and ligands, with new tweaks published every year to boost efficiency, reduce byproducts, or streamline purification. The community’s willingness to share hard-won tips builds trust and advances the field one reaction at a time.
Synonyms & Product Names
In literature and supply catalogs, this compound hides behind several aliases: trifluoromethanesulfinic acid zinc salt, Langlois reagent, and zinc trifluoromethanesulfonate crop up often. Knowing the synonyms prevents ordering the wrong bottle. I’ve had conversations with colleagues where two teams realized they’d been using the same reagent under different names all along, which always led to a few laughs and some updated spreadsheets. Such naming quirks call for vigilance, especially for international teams or collaborations across academia and industry.
Safety & Operational Standards
Working with zinc trifluoromethanesulfinate doesn’t carry the same hazards as strong oxidizers or volatile organics, but nobody likes accidental exposure. Dust masks and eye protection handle most bench-scale risks, and industry standards stress the importance of proper ventilation. Spills are best handled with dry cleaning agents, since water can start decomposition or react with traces of acid. The compound displays low acute toxicity, though the literature encourages treating it with respect and minimizing unnecessary contact. I’ve watched experienced chemists drill newcomers on double-checking scale calculations, verifying reagent compatibilities, and running small test batches. These routines prevent avoidable mistakes and keep projects on track. For waste management, segregating fluorinated byproducts from regular organics simplifies disposal and minimizes environmental footprints.
Application Area
The real-world value shines brightest in synthetic chemistry, pharmaceutical development, and agrochemical design. Drug makers often chase trifluoromethyl-substituted building blocks, which resist metabolic breakdown in the body and enhance efficacy at lower dosages. In crop protection, similar logic applies—fluorinated groups bring resistance to degradation, keeping active ingredients potent in the field. Some imaging agents for medical diagnostics rely on high fluorine density, where stable reagents like this make labeling less of a headache. I’ve even seen research teams in material science probe new uses in polymer cross-linking or surface treatments. Each of these domains benefits from reagents that balance stability and selective reactivity, especially ones that avoid elaborate safety infrastructure or hard-to-access supply chains.
Research & Development
Ongoing research delves into greener processes, alternative starting materials, and expanded scope across both small and large molecules. Green chemistry initiatives have spurred tweaks in reagent design, with some labs experimenting with aqueous solvents or flow chemistry setups to cut down on hazardous waste. Collaborative studies with analytical teams reveal subtle effects of impurities or alternate metal centers, driving refinements in catalyst selection or workup strategies. Looking back at group meetings and conference talks, it’s clear that direct communication among process chemists, analysts, and bench scientists powers most major advances—feedback loops that shrink development timelines and flag hurdles before scale-up. Funding agencies and industry sponsors keep the pressure high to deliver new variants tailored for safer, faster, or more cost-effective syntheses.
Toxicity Research
Toxicological data on zinc trifluoromethanesulfinate paints a conservative picture: minimal acute risk at typical exposure levels, but real caution around inhalation of powders or chronic skin contact. Animal studies show limited systemic absorption, yet detailed studies lag behind those for more common zinc or fluorine compounds. In the lab, the guiding principle lands on “use the right precautions every time,” a habit drilled into young researchers by mentors and safety officers. For large-scale operations, institutional safety audits and regular air monitoring catch issues before they escalate. Despite its relative safety profile, nobody takes shortcuts with personal protective equipment or good ventilation—practice built from experience and reinforced by every incident report that circles through the organization’s email.
Future Prospects
Zinc trifluoromethanesulfinate’s story continues to evolve, driven by the hunger for smarter, safer, and more effective molecular tools. With the pharmaceutical and materials industries turning over every stone for incremental gains, demand for stable trifluoromethylating reagents stays high. The reagent’s versatility and safety make it an attractive candidate for flow chemistry platforms and automated synthesis, both of which aim to shrink timelines and cut operational risks. As regulatory scrutiny grows, especially concerning environmental releases of fluorinated byproducts, R&D teams face new obstacles and incentives to develop recyclable or less persistent alternatives—pushing for creative blends of chemistry and engineering. From my own experience, chemistry never sleeps; each better reagent or cleaner process shapes the field and opens doors nobody imagined a decade ago. Zinc trifluoromethanesulfinate, building on its solid track record, stands ready for new chapters as both a workhorse and a testbed for the next generation of synthetic chemistry.
Demand Grows for Reliable Reagents
Chemistry pulls its weight by giving us the tools to build medicines, materials, and technology. In the modern lab, a handful of reagents spark more innovation than others. Zinc trifluoromethanesulfinate is one of those odd-sounding powders that’s quietly reshaping organic chemistry and drug development.
The Appeal of Simple Approaches
Zinc trifluoromethanesulfinate, often called the Langlois reagent, gets attention for its ability to add a trifluoromethyl group (CF3) to organic molecules. This isn’t a throwaway process. Trifluoromethyl groups show up in a string of drugs and agrochemicals because they can boost a compound’s stability, help it pass through cell membranes, or even ease dosing concerns. You won’t hear about this outside of chemistry circles, yet without these modifications, many blockbuster medicines would never reach pharmacy shelves.
The Shortcut Chemists Appreciate
A big draw is that zinc trifluoromethanesulfinate lets chemists attach a trifluoromethyl group without fussing over tricky reaction conditions. In the past, people had to use harsh chemicals or risk explosive byproducts when chasing these fluorinated building blocks. This reagent allows work under gentler conditions, even with oxygen and moisture in the air. Fewer headaches, less fuss, safer for everyone in the lab. As someone who spent long hours in graduate research juggling risky fluorinating agents, I would have appreciated these safer alternatives years ago.
Impact on Drug Discovery
Medicinal chemistry keeps hunting for new ways to tune drug molecules. When a trifluoromethyl group lands on a drug candidate, it can make the compound last longer in the body or skew its interaction with enzymes. More than thirty percent of new drugs contain a fluorine atom somewhere in their structure. Adding these groups remains pivotal for companies chasing next-generation treatments for cancer, infection, and even mental health. Zinc trifluoromethanesulfinate fits right into workflows thanks to its reliability. Labs aren’t just using it once or twice—they rely on it across entire drug discovery programs, from the first sketch to late-stage optimization.
Challenges and Solutions in Sourcing
There’s a catch, though. The cost and stability of specialty reagents can put a dent in budgets, especially in small startups or cash-strapped university groups. Access depends on manufacturing routes and supply chain quirks. To better support research, the chemical industry would do well to explore greener production methods and simplify purification steps. The more open suppliers make these reagents, the faster scientists can develop safer and more effective drugs.
Thinking Beyond the Laboratory
Making chemical research safer and more accessible doesn’t stop with the right reagents. Training matters. Not every chemist gets to work in a world-class lab, and not every country has equal access to the latest tools. Open sharing of protocols and results, better funding for chemical education, and streamlined regulatory approvals all help science serve communities. I remember seeing students struggle with outdated materials; a steady flow of safer, more user-friendly reagents could lift their work—and their careers.
The Practical Value of Tiny Innovations
It’s easy to look at a jar of chemical powder and overlook its broader impact. But reagents like zinc trifluoromethanesulfinate remind us that genuine progress builds from small, thoughtful changes. Each time a chemist safely creates a promising molecule, the world edges a little closer to new cures and technologies. Easy-to-use, accessible chemistry keeps that momentum alive.
Unpacking a Zinc Compound’s Role in Chemistry
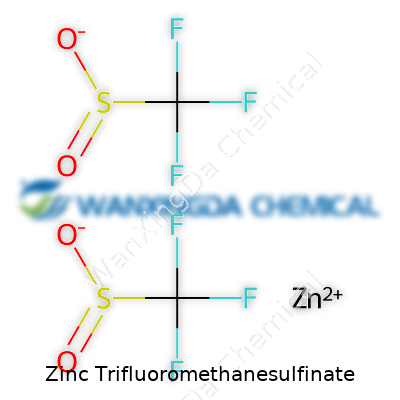
Now and then, a mouthful like “zinc trifluoromethanesulfinate” pops up in a conversation or a scientific journal, and, for most of us, it’s a head-scratcher. Still, this isn't just another chemical lost in a lab manual. Zinc trifluoromethanesulfinate—often referred to as ZnTFMS or Langlois reagent—holds a pretty useful spot in organic synthesis. Its formula is Zn(SO2CF3)2. Imagine two trifluoromethanesulfinate groups (SO2CF3) dangling off a zinc center, with the zinc carrying a +2 charge to balance each negatively charged sulfinic acid fragment. Visualizing it gets tricky, sure, but picture zinc at the center flanked by two SO2CF3 arms, and you’re close enough for most purposes.
Why the Structure Matters Beyond the Lab
Chemistry, for me, truly started clicking only once I realized how formulas connect to the workbench. Each atom in this compound shapes how it behaves when mixed with other stuff. The fluorine atoms in those CF3 groups give a punch of electronegativity, which means they pull electron density hard. This ends up making the compound act as a gentle, controllable source of trifluoromethyl radicals—handy little units that chemists use to tweak pharmaceuticals and agrochemicals.
Over the years, I’ve watched researchers lean on reagents like ZnTFMS when other methods fail, especially for attaching trifluoromethyl groups to target molecules. This kind of structural editing improves the stability and uptake of medications by the body or lengthens the shelf life of some crop treatments. Such tweaks can take years off a development timeline.
The Broader Stakes: Why Such Chemistry Matters
Quiet breakthroughs like those involving ZnTFMS rarely make headlines, yet they shape the medicines and food we rely on. Fluorinated molecules turn up everywhere from asthma inhalers to flea collars, thanks to compounds like this. I’ve sat through lectures where smart use of a single zinc salt meant the difference between a successful synthesis and a failed experiment.
Concerns emerge about accumulation of fluorinated chemicals in soil and water. People deserve transparency, so I dig into published scientific studies and environmental assessments before celebrating any new chemistry. Current research shows that using reagents with such strong fluorine character must follow careful handling and waste protocols. Problems arise mostly when shortcuts are taken—a lesson I’ve learned from seeing rushed benches and poorly managed waste streams come back to haunt labs.
Dealing With Risks and Moving Forward
Instead of calling for blanket bans on compounds like ZnTFMS, I focus on practical improvements. More researchers are building methods that either use less persistent fluorinated reagents or recycle and contain them better. Universities and industry labs share protocols for greener hazard management, including strict waste segregation and regular solvent recycling, which cuts down on unnecessary discharge.
Pushing for open publications of environmental impact data can nudge the conversation from scare tactics to responsible discussion. People who do the daily work in chemistry, from grad students to older professionals, know that responsibility comes with knowledge. So I keep advocating—both in my community and online—for transparent science and strong stewardship, even when it complicates things. Doing chemistry right matters, and it starts one molecule at a time.
Taking Stock of Lab Realities
A lot of folks outside the lab think chemistry is all breakthroughs and sparks, but some of the most important work is as simple as getting storage right. Zinc trifluoromethanesulfinate doesn’t make headlines like lithium or ammonia, but for anyone who’s spent hours chasing purity or cleaning up messes, the nuts and bolts of its storage matter day to day.
Why Storage Habits Shape Outcomes
Having handled a range of organofluorine compounds, I’ve learned the real trouble often comes in through overlooked details. Some compounds can weather poor storage, but zinc trifluoromethanesulfinate isn’t one you want to shove on a crowded shelf next to every other reagent. Over time, you see that dry, cool conditions aren’t just a line in a manual, they save your budget and reputation.
Moisture Is the Quiet Enemy
Every chemist with experience knows water sneaks in if you let your guard down. With zinc trifluoromethanesulfinate, exposure to moisture can start problems you might not notice until your next synthesis fizzles. If a compound clumps, changes color, or even sticks inside the bottle—these are all signals that humidity is interfering. Around here, I store similar reagents with fresh desiccant in tightly sealed containers, tucked away from the reach of humid air. Fancier labs with gloveboxes solve the problem more elegantly, but even a double layer of zip bags and silica gel in a fridge can go a long way.
Heat: An Unseen Threat
Keeping the bottle away from direct sunlight or heat sources feels obvious, but too many custodial slip-ups happen with bottles left next to vents or radiators. Chemistry classrooms and research spaces tend to run warm, especially in summer. I’ve seen folks try to offset this by stashing everything in the cold room. That works, as long as you remember to let the bottle reach room temperature before opening it, to avoid condensation sneaking in as the bottle “sweats.”
The Safety Layer
Zinc trifluoromethanesulfinate isn’t the most hazardous, but dust and powder always warrant respect. Sealed containers should be labeled with storage dates and hazards. Someone’s always finding old bottles tucked behind newer supplies. Without a clear label, it’s easy to forget what’s inside, and forgotten or degraded chemicals are at the root of far too many minor lab accidents.
Real Solutions: Support from Protocols and Practice
Years ago, my lab picked one central shelf for all sulfur-based reagents, away from acids and bases, under lock and key to keep visitors from grabbing bottles out of curiosity. Having rules everyone knows—no food nearby, clean spills immediately, double-check seals before putting anything away—prevents many headaches. I encourage documenting storage temperatures in your inventory, so changes don’t catch you by surprise.
The Value of Small Steps
A bit of caution saves hours of rework and keeps your work accessible to collaborators. Safe storage isn’t glamorous. Still, the credibility built from attention to these gritty details will last longer than any single experiment. I’ve found that rookie mistakes usually happen not out of ignorance, but from rushing through the “boring stuff.” Slowing down pays dividends, and it shows real respect for the science—and everyone sharing the lab bench.
Zinc trifluoromethanesulfinate isn't a household name—unless you spend your days in a chemistry lab. This compound comes up in organic synthesis, prized for its ability to help attach trifluoromethyl groups to molecules. These groups change how medicines break down in the body or how agrochemicals last in the field. That's a big deal if you're designing new drugs or crop protection formulas.
The question of safety hangs over every specialty chemical. Most people outside the lab don’t realize just how cautious chemists can be. With zinc trifluoromethanesulfinate, what risks are we really talking about?
What’s Actually Known
The research community hasn’t raised many alarms about acute toxicity. Zinc as a metal isn’t high on the hazard list for humans. Fluorinated organics sometimes get side-eye, based on the dangerous reputation of cousins like PFAS (per- and polyfluoroalkyl substances), but not all fluorine chemicals behave the same way, biologically or environmentally. In the case of zinc trifluoromethanesulfinate, recognized commercial suppliers highlight “harmful if swallowed” on their documents, along with typical recommendations for chemical gloves, goggles, and fume hood work. That doesn’t mean a single exposure leads straight to the ER, but there’s a solid reason for care—especially since inhaling powders or getting them in your eyes is always unpleasant in lab life.
Let’s face it: a mistake can have lasting effects. Chronic exposures, which can creep up over time, are often trickier to pin down. My own lab training drilled into me how repeated unnoticed spills or unwelcome dusty air could provoke allergic skin reactions, eye problems, or breathing issues. With specialty chemicals, many research groups lean on animal testing data or hazard analogies with similar compounds, simply because no long-term epidemiological studies exist for every obscure reagent. That leaves a margin for uncertainty nobody should ignore. Safety Data Sheets tend to err on the side of caution, and I’ve seen workers develop asthma or rashes from what seemed like low-hazard chemicals—usually because something slipped through the cracks on proper handling. Zinc trifluoromethanesulfinate may never make headlines, but complacency doesn’t pay off.
Environmental Questions and Responsible Handling
The fluorinated part of the molecule stirs up discussion. Nobody wants a new source of persistent or bioaccumulative pollution in our water or soil. EPA regulatory attention focuses sharply on chemicals with unclear breakdown products, especially if they might linger in ecosystems. So far, there’s not enough public data showing that zinc trifluoromethanesulfinate builds up in the environment the way some notorious PFAS do, but the prudent move is to collect chemical waste and not pour unused materials down the drain. Treat every spill as a problem, not an irritation. If you care about your local environment, the less you put unknowns into your wastewater, the better.
Where Solutions Start
Lab safety isn’t magic—it’s practice and awareness. Nobody expects perfection every moment, but skipping the basics only piles up trouble. Gloves, lab coats, and a good fume hood represent old school habits that have saved many careers and, in some cases, lives. Educators and lab managers set the tone. People learn safety culture from their first supervisors: if disposing of zinc trifluoromethanesulfinate neatly and logging each bottle seems normal, younger researchers pick that up.
It helps to keep the safety discussion going. Sharing stories—good and bad experiences—matters more than dry paperwork. If an accident occurs, talk about it. No shame, just lessons for the next shift. Push for transparency with chemical suppliers and regulators: ask for more data, not less. Uncertainty about unknowns doesn’t mean ignorance is bliss; it just means the community has more work to do.
Few tools make as much difference in the lab as a reagent that opens new shortcuts in building molecules. Zinc trifluoromethanesulfinate, widely known as the Langlois reagent, is one of those unsung heroes. The chemistry community loves to focus on glamorous reactions, but if you need to pop a trifluoromethyl (CF3) group onto a molecule, the conversation often comes around to this humble zinc salt.
Unlocking Trifluoromethylation
CF3 groups show up all over the place in pharmaceuticals and advanced materials. Adding them can boost metabolic stability and often helps a drug sneak past the liver’s enzymes, so it sticks around longer and works better. Big pharma keeps looking for new ways to install this group. A few years ago, many labs leaned on more hazardous options — things like Ruppert–Prakash reagent or expensive fluoroalkyl iodides. Both bring their own headaches, including high costs and serious handling hazards.
Zinc trifluoromethanesulfinate entered the scene as a less aggressive, more practical choice. It delivers that coveted CF3 group under milder, friendlier conditions. That matters, especially for larger-scale work or in labs where safety and budget always need attention. And the method doesn’t just help chemists; patients who rely on modern medicines benefit, too, since safer drug synthesis can speed up timelines and reduce side effects tied to impurities.
Pushing the Boundaries of Aromatic Chemistry
For making aryl trifluoromethyl compounds, this reagent became a game-changer. Take a look at how many FDA-approved drugs now include trifluoromethyl arenes. Many wouldn’t exist without reliable methods for attaching that group to rings. In the past, chemists struggled with limited options, but with zinc trifluoromethanesulfinate, the world of electron-rich and even moderately electron-deficient arenes opened up to CF3 chemistry.
And it works: simple oxidants like Selectfluor or copper salts can get the reaction rolling. You don’t need high temperatures or intense light. There’s less mess with by-products, fewer strong acids or bases in the mix, and usually a good yield — so less wasted material, fewer headaches during purification.
Strengthening Diversity in Fine-Chemical Synthesis
Drug discovery can get stuck if every step needs harsh conditions or rare chemicals. The Langlois reagent has made alkene and heterocycle functionalization less of a grind. It has found use in the creation of agrochemicals and specialty materials, not just blockbuster drugs. Everyday researchers get another shot at designing new products without raising the risk profile or production costs through the roof.
What’s Next for Safer, Smarter Chemistry?
Accessibility counts for a lot. A reagent that checks safety, cost, and grunt work off the list means chemists spend less time dodging explosions in the fume hood and more time thinking creatively. The new push is to build on what we have — maybe tweak the zinc reagent to deliver other functional groups, maybe find even more earth-friendly oxidants.
Better understanding the mechanism also matters. Curious minds with access to better tools (like flow chemistry setups or electrochemical cells) can tinker at the intersection of speed, efficiency, and environmental care. The real win? Making the toolkit broader, so the next generation of medicines, smart materials, and crop protectants are just a reaction or two away.